A technical reference for QA managers and validation engineers on the integration of consumable traceability into the site Contamination Control Strategy (CCS).

In a GMP-regulated environment, the cleanroom mop is no longer viewed as a generic utility but as a critical controlled input. Batch traceability cleanroom mop systems serve three primary functions:
Product-level traceability identifies a SKU; batch-level traceability identifies the specific manufacturing event. For cleanroom mops, this requires a "genealogy" that links the finished product back to the specific lot of polyester yarn or microfiber fabric used, the cleanroom laundry shift, and the specific sterilization cycle.
When mops are treated as controlled inputs, every individual package is a data point. This data allows QA teams to verify that the product in their hand is identical in performance and purity to the product validated during the initial facility startup or PQ (Performance Qualification).
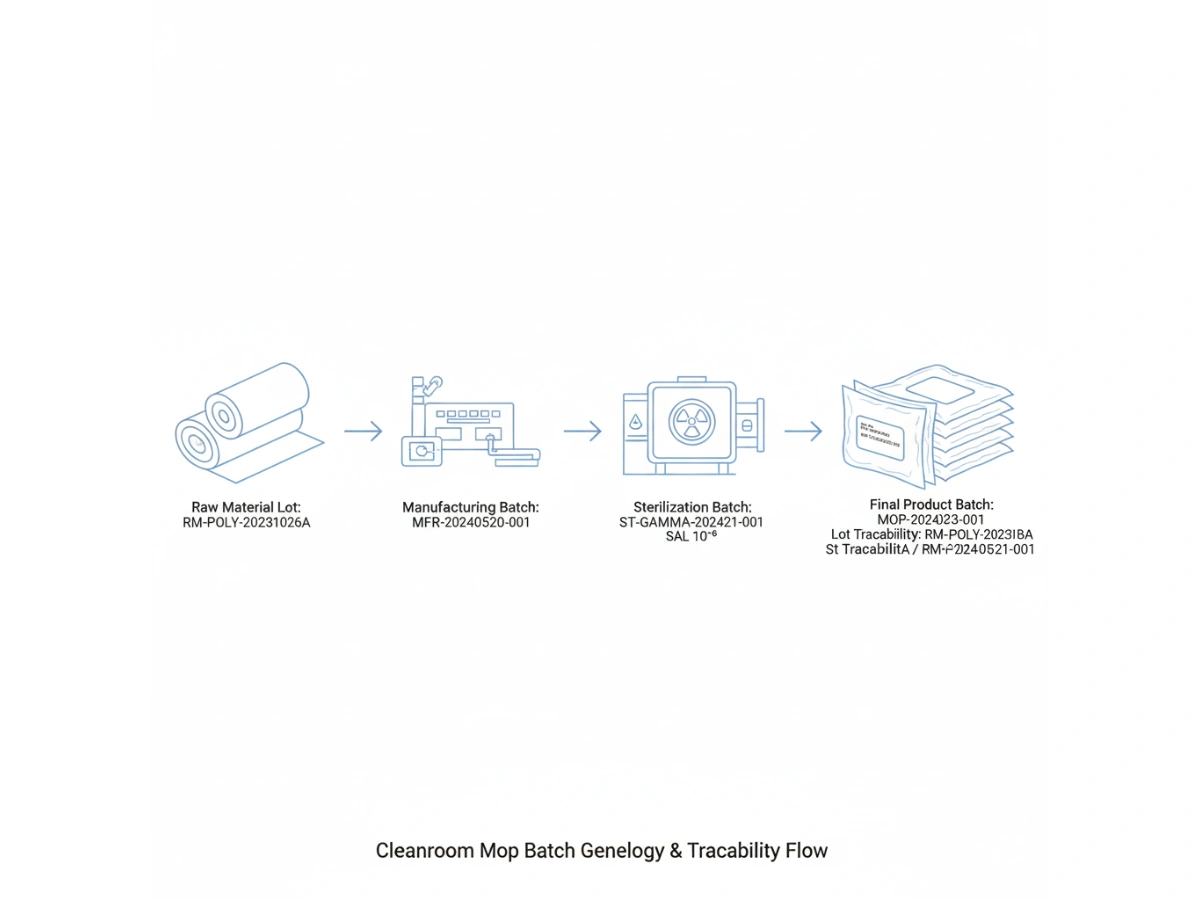
Modern GMP principles require that all materials coming into contact with sensitive surfaces be fully accounted for. During an inspection, an auditor may select a date of production and ask for the lot traceability of the cleaning materials used on that day.
Expectations focus on the "Linkage of Records." An auditor will look for a seamless transition from the facility's cleaning logs to the batch-level documentation provided by the supplier. If a mop batch cannot be linked to a Certificate of Analysis (COA) or a sterilization record, the sterility and cleanliness of the entire cleanroom suite may be called into question.

A robust traceability system for cleanroom consumables GMP compliance must include:
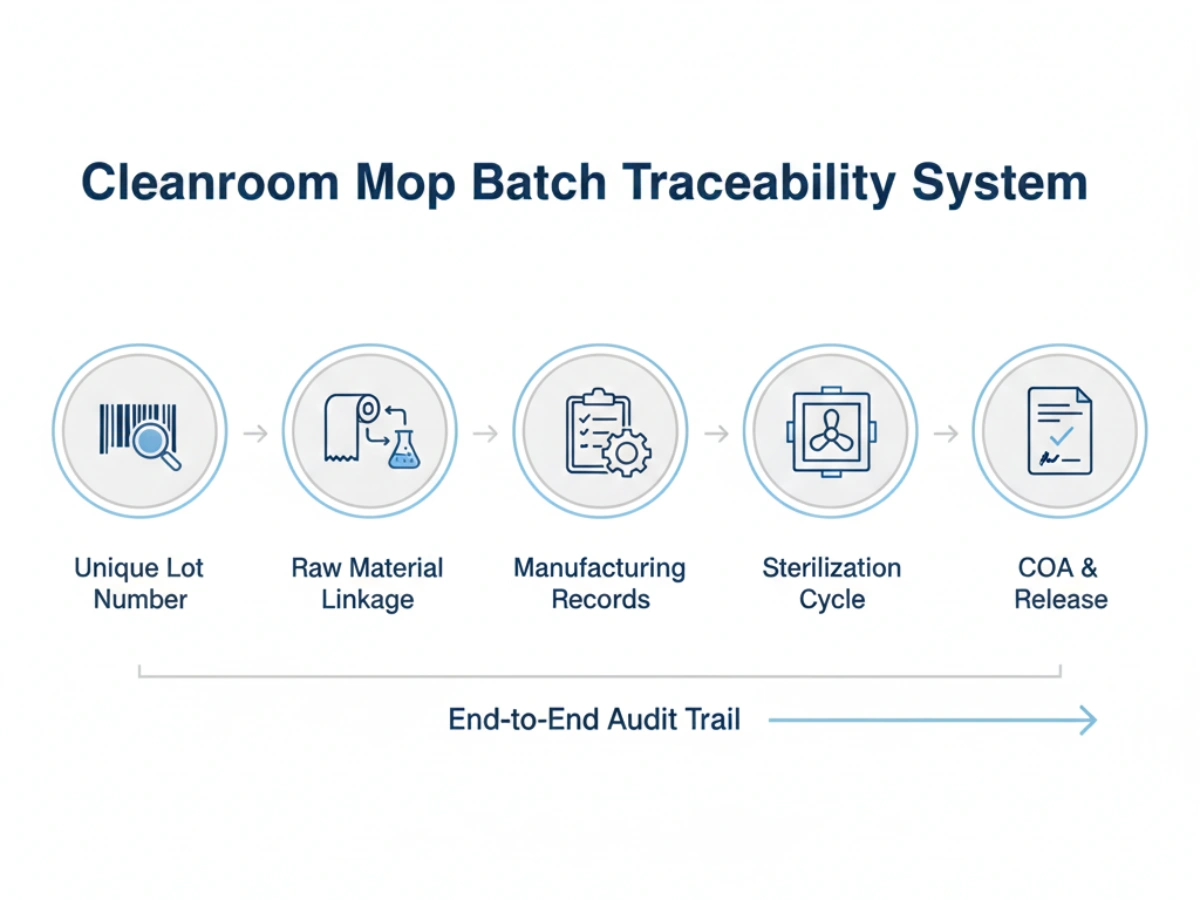
Supply stability is often threatened by "drift"—small, undocumented changes in manufacturing that accumulate over years. Batch traceability prevents this by establishing a baseline for lot-to-lot consistency.
In the event of a CAPA (Corrective and Preventive Action) involving environmental monitoring (EM) spikes, traceability allows QA to determine if a specific batch of mops contributed to the event. Without this, the facility may be forced to discard all inventory and halt production, posing a massive operational risk. Traceability transforms a potential facility-wide crisis into a localized material quarantine.

A pharmaceutical mop manufacturer / OEM acts as an extension of the pharma site’s quality system. Their responsibilities include:
For a qualified manufacturer, the ability to produce these records on demand is the hallmark of GMP maturity.
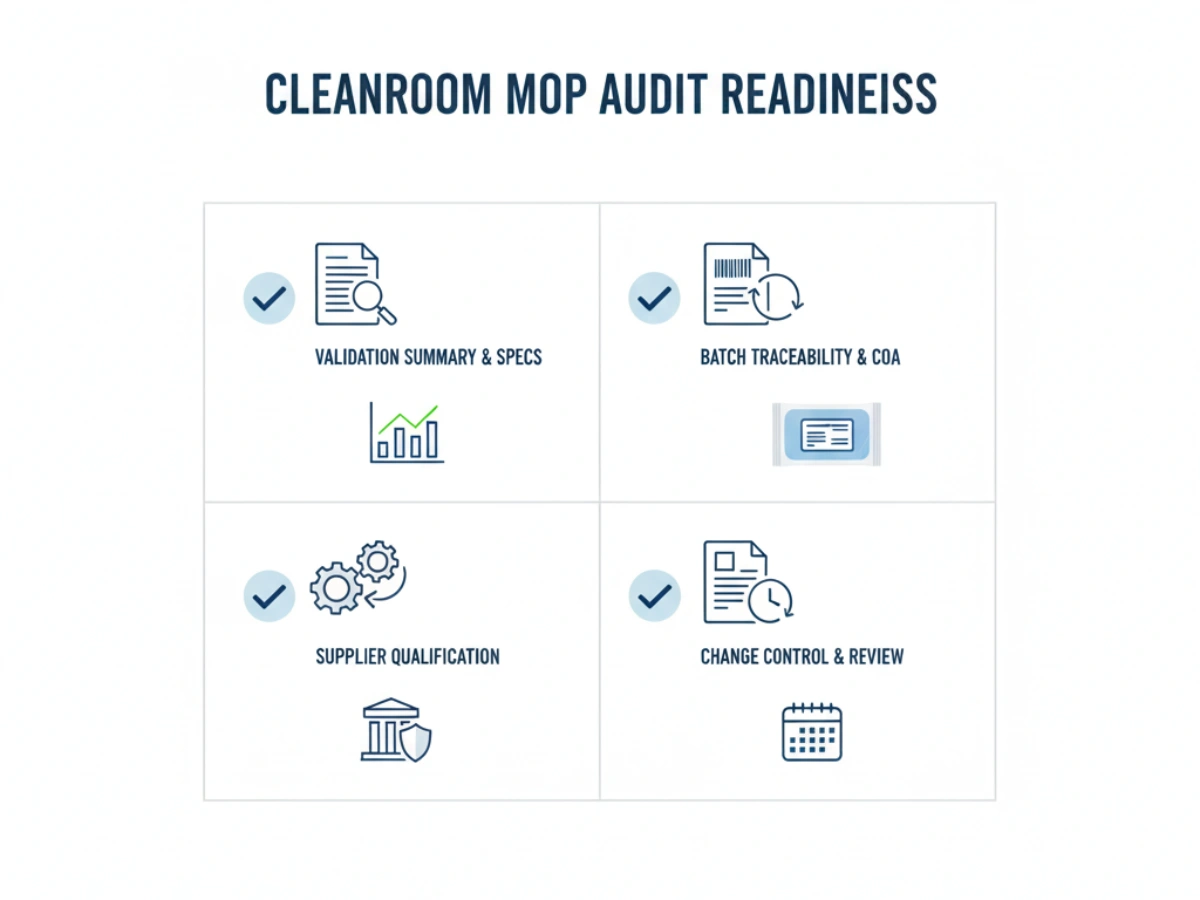
During OEM supplier qualification, QA teams should utilize the following checklist:
| Checkpoint | Requirement |
|---|---|
| Batch Definition | Is the batch size clearly defined and manageable? |
| Sample Retention | Does the supplier keep retain samples for every lot? |
| Audit Access | Can the supplier provide full raw material logs within 48 hours? |
| Physical Labeling | Is the batch number resistant to cleanroom disinfectants (IPA)? |
Batch traceability is not merely a documentation exercise; it is the backbone of a defensible quality system. In the context of Grade A and B environments, the ability to track every fiber and sterilization cycle ensures that the cleaning process remains a controlled variable rather than an unknown risk.
Mature pharmaceutical QA teams prioritize suppliers who demonstrate total transparency in their batch records, viewing them as long-term partners in patient safety and regulatory compliance.
Ask for lot genealogy, batch-specific COAs, sterilization linkage, and change-control readiness to strengthen your audit narrative.
Request Documentation Package

We will contact you within 1 working day, please pay attention to the email with the suffix “*@midposi.com”.