In the evolving pharmaceutical landscape, quality assurance and compliance officers face increasing pressure to develop sophisticated risk-based cleanroom environmental monitoring SOPs that align with 2026 regulatory expectations. This guide facilitates the shift toward using data and risk assessment to focus resources where they provide the most protection. Integrating high-performance cleanroom mops into your contamination control strategy is a baseline requirement for these dynamic environments.
Who Should Use This Guide
- Quality Assurance Specialists: Responsible for EM program design and oversight.
- Compliance Officers: Ensuring adherence to FDA and EMA requirements.
- Validation Engineers: Leading cleanroom qualification activities.
- Facility Managers: Implementing monitoring programs.
- Regulatory Affairs: Navigating evolving compliance requirements.
2026 Regulatory Landscape

FDA's Risk-Based Approach Evolution
- Enhanced Quality Risk Management (QRM): Integration of ICH Q9 principles.
- Data Integrity 2.0: Real-time data collection and automated trend analysis.
- Continuous Monitoring: Requirement for continuous environmental monitoring (CEM) in critical areas.
- Predictive Analytics: Implementation of early warning systems for contamination risks.
- Supply Chain Integration: Risk assessment including supply chain impacts.
EMA GMP Annex 1 (2026 Implementation)
- Risk-Based Classification: Dynamic cleanroom classification based on risk assessment.
- Contamination Control Strategy (CCS): Comprehensive approach to persistent contamination.
- EM Program: Risk-adjusted monitoring frequencies and locations.
- Investigation Protocols: Structured approach to monitoring excursions.
- Continuous Improvement: Mandated requirement for ongoing program optimization.
Transition Timeline for Compliance
| Timeline | Requirements | Actions Required |
|---|---|---|
| Q1 2024 - Q2 2024 | Gap analysis and risk assessment development | Develop risk assessment framework; identify high-risk areas. |
| Q3 2024 - Q1 2025 | SOP revision and validation | Update EM procedures; validate new monitoring approaches. |
| Q2 2025 - Q4 2025 | Implementation and pilot testing | Roll out risk-based program; collect performance data. |
| Q1 2026 - Full Compliance | Complete implementation and optimization | Finalize program; establish continuous improvement. |
Risk Assessment Framework Development
Step 1: Define Risk Assessment Criteria
Establish comprehensive criteria for evaluating cleanroom areas and processes.
- Product Risk: Sterile vs. non-sterile, therapeutic area, route of administration.
- Process Risk: Process complexity, automation level, intervention frequency.
- Facility Risk: Air changes, pressure differentials, personnel traffic.
- Historical Data: Previous monitoring results and contamination trends.
- Change Impact: Recent changes, deviation history, CAPA effectiveness.
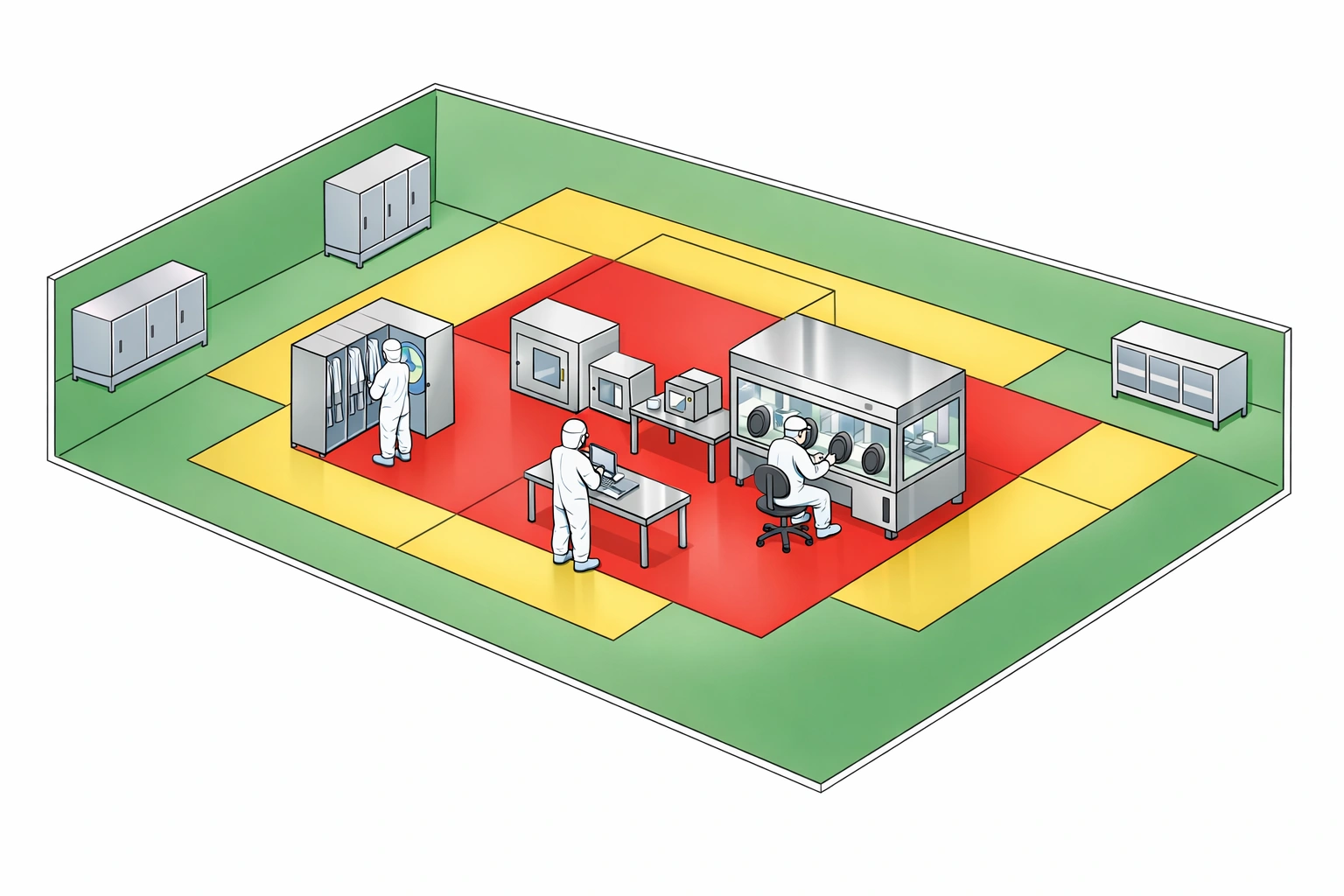
Step 2: Develop Risk Matrix
Categorize areas and processes based on predefined criteria into a structured matrix.
Continuous Monitoring
Immediate Investigation
Increased Frequency
Timely Investigation
Standard Frequency
Routine Investigation
Implementation Tip: Use a scoring system (1-5) for each risk factor. Areas scoring ≥15 require high-risk monitoring strategies.
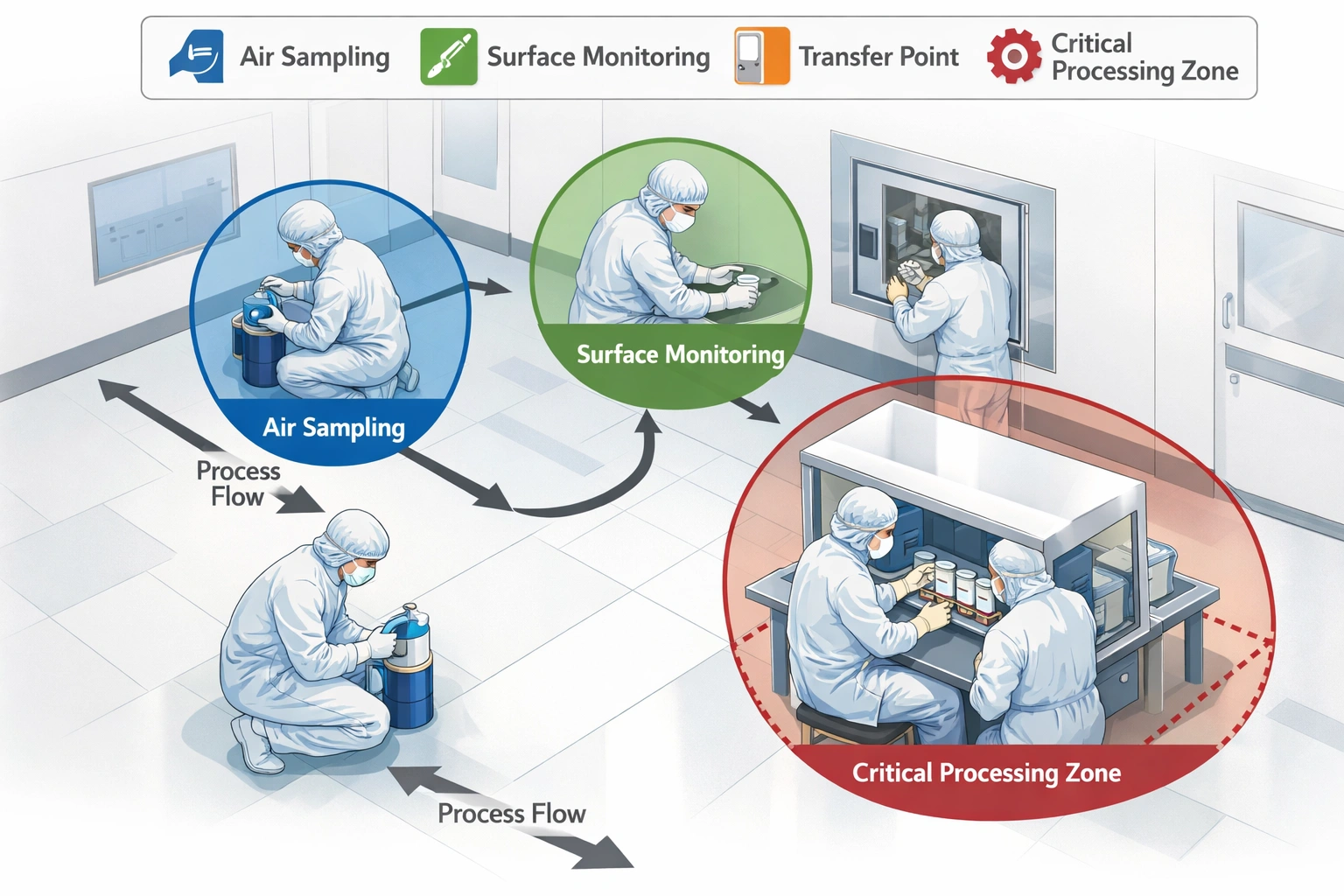
Step 3: Establish Monitoring Locations
Strategic placement of equipment is vital. Use cleanroom validation insights to pinpoint critical locations.
- Aseptic Processing: Critical zones, fill-finish interfaces.
- Transfer Points: Airlocks, gowning rooms.
- High Traffic Areas: Main corridors, intervention points.
Step 4: Define Monitoring Frequencies
| Risk Level | Viable | Non-viable | Surface |
|---|---|---|---|
| High | Continuous + Weekly | Continuous | Daily. |
| Medium | 2-3 times/week | 2 times/week | 2-3 times/week. |
| Low | 1-2 times/week | 1 time/week | 1 time/week. |
Step 5: Set Alert and Action Levels
Establish scientifically justified levels based on historical data and process vulnerability. Apply statistical process control methods for precision.
SOP Development Structure
Template: ENV-MON-001 (Version 2.0)
1.0 Purpose: Establish a risk-based EM program that ensures product quality while optimizing resources.
4.0 Procedure: Risk assessment protocol, monitoring location strategy, frequency determination, alert/action level management.
| SOP Element | Traditional Approach | Risk-Based 2026 |
|---|---|---|
| Monitoring Locations | Fixed grid pattern | Risk-based strategic placement. |
| Frequency | Standardized schedule | Risk-adjusted with dynamic review. |
| Alert Levels | Static numerical values | Statistical + contextual analysis. |
Implementation Best Practices
Effective pharmaceutical facility management involves moving from static to dynamic procedures.
Technology Recommendation: Implement CEM systems with real-time data analytics. These provide essential predictive capabilities for 2026 compliance.
Training and Competency Matrix
| Audience | Training Topics | Assessment |
|---|---|---|
| QA Staff | Risk methodology, Data analytics | Risk assessment simulation. |
| Microbiology | Microbial risk, Trend analysis | Case study analysis. |
| Operations | Alert response, Documentation | Practical demonstration. |
Case Study: Global Implementation
Global Pharmaceutical Success Story
- 35% reduction in monitoring costs.
- 40% faster contamination detection.
- 50% reduction in false positive alerts.
- Enhanced operator understanding of risk concepts.
"The risk-based approach transformed our environmental monitoring program from a compliance burden to a valuable quality assurance tool".
Performance Metrics and KPIs
| Metric Category | Specific KPI | Target |
|---|---|---|
| Effectiveness | Contamination detection time | < 2 hours. |
| Efficiency | Monitoring cost per unit | ↓ 30% baseline. |
| Compliance | Regulatory inspection score | ≥ 95%. |
| Quality | Alert accuracy rate | ≥ 90%. |
Ready to Transform Your EM Program?
Don't wait for 2026 mandates to act. Contact our expert team for a comprehensive assessment and custom implementation plan tailored to your facility.
Additional Essential Resources
- FDA Guidance for Industry: Sterile Drug Products (2021).
- EU GMP Annex 1: Manufacture of Sterile Medicinal Products (2026).
- ISPE Baseline Guide: Environmental Monitoring (2024).
- ICH Q9 Quality Risk Management.
- ISO 14644 Series: Cleanrooms and associated environments.
Disclaimer: This guide is for informational purposes only. Consult with regulatory experts and qualified professionals for your specific facility requirements.





Expert Commentary: The Evolution of Monitoring
"The shift from traditional to risk-based environmental monitoring represents the most significant evolution in cleanroom management in decades. It's about using data and risk assessment to focus resources where they matter most".
"By 2026, regulatory agencies will mandate risk-based approaches. Companies that proactively implement these strategies will gain competitive advantages through improved efficiency and better contamination control".