Batch traceability for cleanroom consumables is not just a documentation exercise. It is the operational backbone that allows manufacturers, sterile processing teams, and regulated facilities to link raw materials, production, sterilization, distribution, and end use into one verifiable chain of evidence.
In pharmaceutical, biotech, semiconductor, and medical device environments, incomplete traceability creates risk far beyond paperwork gaps. It delays investigations, weakens recall response, complicates audits, and undermines confidence in contamination control programs. A robust system should help quality teams move from reactive record collection to proactive control.
Batch traceability for cleanroom consumables means maintaining a complete, retrievable record of each batch from incoming material through manufacturing, sterilization, distribution, and final use. In practice, an effective system should identify what was made, from which source materials, under which process conditions, where it was shipped, and which customers or internal users were affected if a deviation or recall occurs.
For regulated facilities, the best systems are not just compliant on paper. They are fast to search, easy to verify, audit-ready, and capable of supporting a targeted recall within hours instead of days.
Strong batch traceability links raw material lots, production batches, sterilization records, shipment data, and end-user consumption into a single chain of evidence.
Audit-ready systems must support rapid investigation and targeted recall, not just passive storage of disconnected records.
A clear batch numbering logic reduces operator confusion and improves downstream reporting, labeling, release, and complaint handling.
Forward traceability, backward traceability, and recall simulation should be periodically tested to prove the system actually works under pressure.
Many facilities already record batch numbers on cartons, labels, or sterilization documents. The problem is that these records are often fragmented across purchasing, warehouse, production, sterilization, logistics, and end-user logs. When a complaint, contamination event, or audit request occurs, the team then has to reconstruct the history manually.
That approach is slow, error-prone, and expensive. A mature traceability program reduces investigation time, improves release confidence, and limits the scope of recall actions. It also supports trend analysis, supplier management, CAPA, and customer communication.

A cleanroom consumables traceability system should be designed around how regulated organizations investigate deviations and prove control. For many users, the most relevant references include EU GMP Annex 1, FDA cGMP recordkeeping expectations, and ISO 13485 traceability requirements for medical-device-related quality systems.
| Regulation / Standard | Focus Area | Operational Meaning |
|---|---|---|
| EU GMP Annex 1 | Traceability, batch records, recall capability | Facilities should be able to identify affected materials and finished goods rapidly during investigations or market actions. |
| FDA 21 CFR 211.180 / 211.198 | Distribution records, complaint files, retention | Records should support complaint evaluation, lot impact review, and controlled recall execution. |
| ISO 13485 Section 7.5.8 | Unique identification and retrieval | The organization should maintain documented traceability procedures and be able to retrieve linked records when needed. |
For buyers and QA teams, this means traceability should not be treated as a warehouse-only function. It is a cross-functional quality requirement touching supplier control, production documentation, sterilization release, shipping history, and end-user accountability.
A useful way to structure batch traceability is to divide the system into five connected layers: raw materials, manufacturing, sterilization, distribution, and consumption. This framework helps teams define ownership, data fields, and verification checkpoints without losing end-to-end visibility.
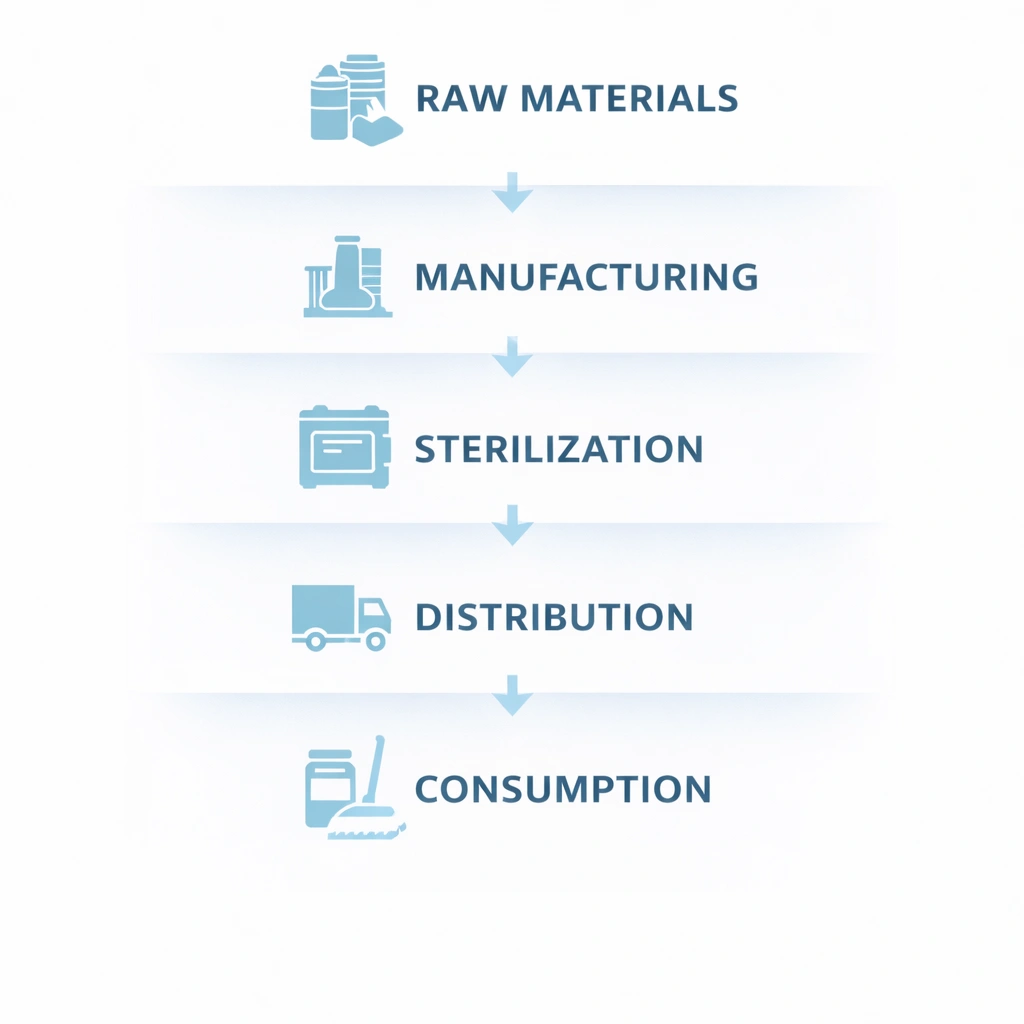
Capture supplier identity, supplier lot number, receipt date, incoming inspection status, and relevant certificates such as COA or CoC. This is the starting point for upstream control.
Record which materials were used, which line produced the batch, who operated or released the process, and which in-process or final QC checks were performed.
For sterile consumables, link the production batch to the sterilization batch, cycle parameters, sterilization method, release status, and any validation evidence.
Link batch numbers to customers, shipment quantities, shipment dates, carrier information, and destination details. This is essential for complaint triage and recall containment.
Where the use model requires it, maintain user-side records of batch number, date used, location, and disposal route. This is particularly valuable in high-risk GMP environments.
The batch number should be more than a serial stamp. It should support quick interpretation, accurate storage, clean label presentation, and low-error manual handling. A structured format also makes digital search and reporting more reliable.

| Batch Element | Example | Why It Matters |
|---|---|---|
| Supplier / Manufacturer Code | MIDPOSI | Identifies manufacturing origin and separates brands, sites, or supply streams. |
| Date Segment | 2026-04-06 | Helps operators quickly identify production chronology and retention logic. |
| Serial Number | 001 | Differentiates multiple batches produced within the same day or cycle window. |
| Suffix / Process Marker | A / B / C | Useful for production, sterilization, re-sterilization, or QA workflow distinctions. |
Even strong physical labeling is not enough if supporting records are incomplete. A defensible traceability file should allow a reviewer to move from a finished batch back to source materials, and from a source material forward to every affected finished batch and customer shipment.
Supplier lot, received quantity, inspection result, document check, release status.
Batch number, operator, line, process date, material usage, QC checkpoints, deviations.
Sterilization lot, cycle data, release decision, customer shipped quantity, destination, complaint linkage.
The documentation set should also be easy to retrieve during supplier audits, customer qualification, complaint handling, and CAPA reviews. If locating the right file takes too long, the system is functionally weaker than it appears on paper.
A traceability system should be tested, not assumed. The most practical approach is to run three types of exercises: forward traceability, backward traceability, and recall simulation. These tests reveal whether data fields are truly linked, whether teams know where to look, and whether response time meets internal expectations.
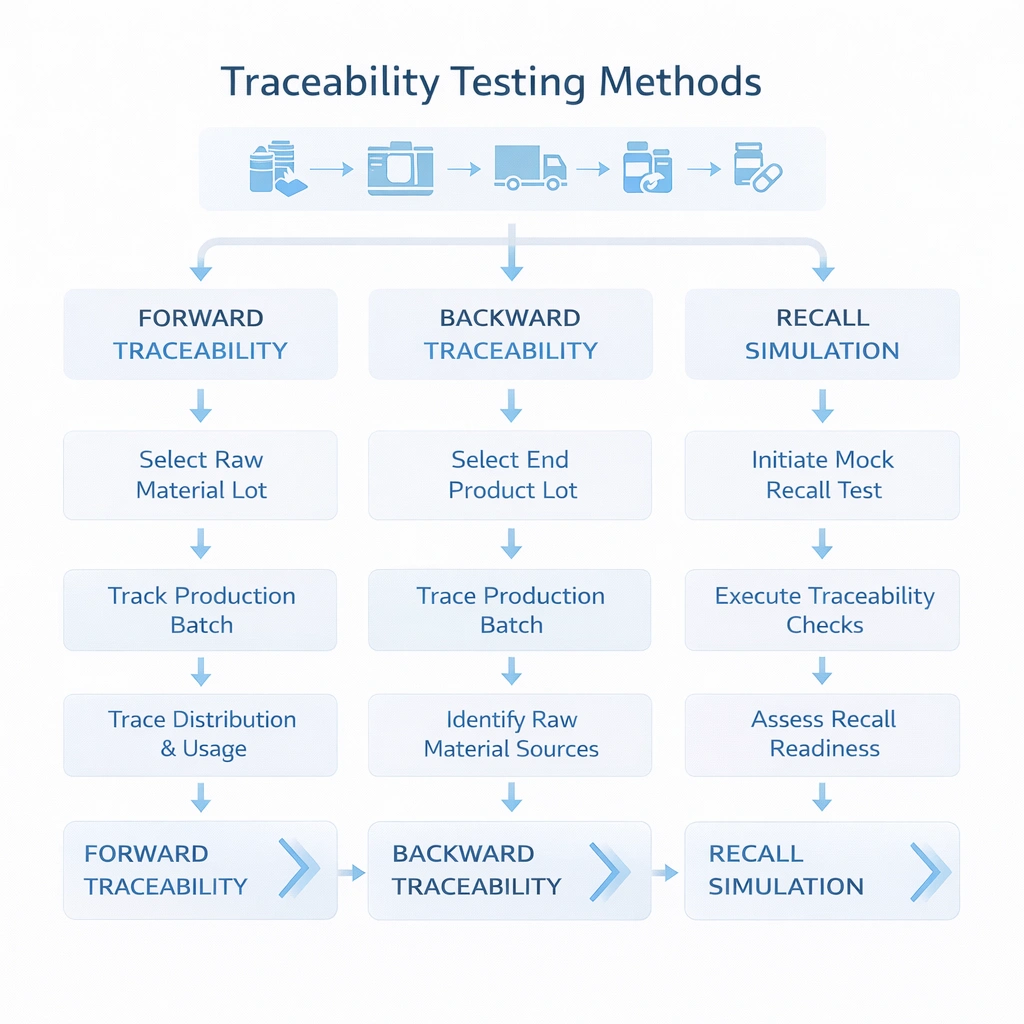
Start with an incoming material lot and determine which production batches, sterilization cycles, and customer shipments were affected.
Start with a complaint or customer-used batch and trace back to the source materials, process records, and release status.
Select a batch and simulate a targeted recall: identify every affected customer, quantity, shipment route, and contact owner. Record the total time needed and the data gaps found. In high-control environments, this exercise is one of the fastest ways to assess whether the system is genuinely recall-ready.
| Test Type | Main Question | Expected Output |
|---|---|---|
| Forward Traceability | Where did this input go? | Affected production, sterilization, and shipment records |
| Backward Traceability | What created this result? | Source material, process history, release evidence |
| Recall Simulation | Can we act fast and accurately? | Customer list, quantity map, response timing, data gap log |
A recall workflow should be predetermined rather than improvised. When a quality issue appears, teams should already know how to classify the issue, identify affected batches, notify stakeholders, stop further distribution, and document root cause and corrective actions.
Spreadsheet-based systems can work at low complexity, but as product range, customer volume, and sterility workflows expand, electronic traceability usually becomes more practical. Barcode or RFID-supported systems reduce manual entry risk and make recall simulation faster and more repeatable.

Define data fields, ownership, batch code logic, retention rules, and traceability scope for sterile and non-sterile lines.
Standardize forms, supplier lot capture, production records, sterilization linkage, and shipment records.
Run forward traceability, backward traceability, and recall simulation tests and document the gaps found.
At a minimum, the system should connect incoming material lots, production batches, release or sterilization records where applicable, shipment history, and the ability to identify affected customers or users during an investigation.
Batch labeling identifies a product group. Full traceability links that label to upstream materials, process records, release decisions, and downstream distribution or use records.
Many quality teams perform it at least annually, while higher-risk or more complex operations may test more often, especially after system changes, supplier shifts, or major CAPA actions.
Yes, for low-volume operations. But once product variation, sterility control, or customer complexity increases, digital systems usually provide better retrieval speed, error reduction, and audit control.
Because sterile products require tighter control of batch history, sterilization linkage, release evidence, and downstream visibility if a complaint, sterility concern, or recall decision arises.
Midposi supports regulated buyers with cleanroom consumables designed for controlled environments, including documentation-oriented communication for qualification, sterility status review, and batch traceability discussions.


We will contact you within 1 working day, please pay attention to the email with the suffix “*@midposi.com”.