Why Environmental Monitoring Matters in GMP Cleanrooms
Even well-designed cleanrooms with strong HVAC systems, validated cleaning programs, and trained personnel cannot be assumed to remain under control without evidence. Environmental monitoring provides that evidence by showing whether contamination levels stay within defined expectations during routine operation.
An effective EM program supports microbial control, particulate control, investigation readiness, process understanding, and continuous improvement. It also creates a direct connection between room classification, personnel gowning, personnel training, airflow visualization, validation lifecycle, and cleaning effectiveness.
| EM Component | What It Measures | Why It Is Important |
|---|---|---|
| Particle monitoring | Non-viable airborne particulate levels | Helps verify air cleanliness and HVAC performance |
| Active air sampling | Viable airborne microorganisms | Detects microbial contamination risk in room air |
| Settle plates | Passive viable fallout over time | Useful for assessing exposure during operations |
| Contact plates / swabs | Surface microbial contamination | Shows effectiveness of cleaning and touch discipline |
| Trend review | Recurring patterns across time and zones | Supports risk detection before larger failures occur |
Viable vs Non-Viable Monitoring
Environmental monitoring usually includes both viable and non-viable methods. These are related, but they are not interchangeable. A room can meet particulate expectations and still show microbial risk, particularly if personnel behavior, cleaning practices, or intervention controls are weak.
| Monitoring Type | Examples | Main Use |
|---|---|---|
| Non-viable monitoring | Particle counters, airborne particle testing | Verifies cleanroom air classification and dynamic control |
| Viable monitoring | Active air samplers, settle plates, contact plates, swabs | Detects microbiological contamination risks |
Need GMP-ready cleanroom support?
Midposi supports contamination control programs with cleanroom garments and GMP-focused product solutions designed for pharmaceutical and controlled environments.
Common EM Sampling Methods in Cleanrooms
Sampling methods should be selected based on room grade, activity level, product exposure, intervention frequency, and investigation needs. Good programs define not only what is sampled, but also why each method is appropriate.
Typical methods include:
- Particle counters for real-time airborne non-viable monitoring
- Active air samplers for viable airborne monitoring
- Settle plates for passive viable monitoring during operations
- Contact plates for equipment, walls, work surfaces, and gloves
- Swabbing for irregular or hard-to-contact surfaces
Visual Explanation: EM Sampling Strategy
A strong environmental monitoring map should show which methods are used in critical zones, support areas, personnel touchpoints, and dynamic intervention locations.
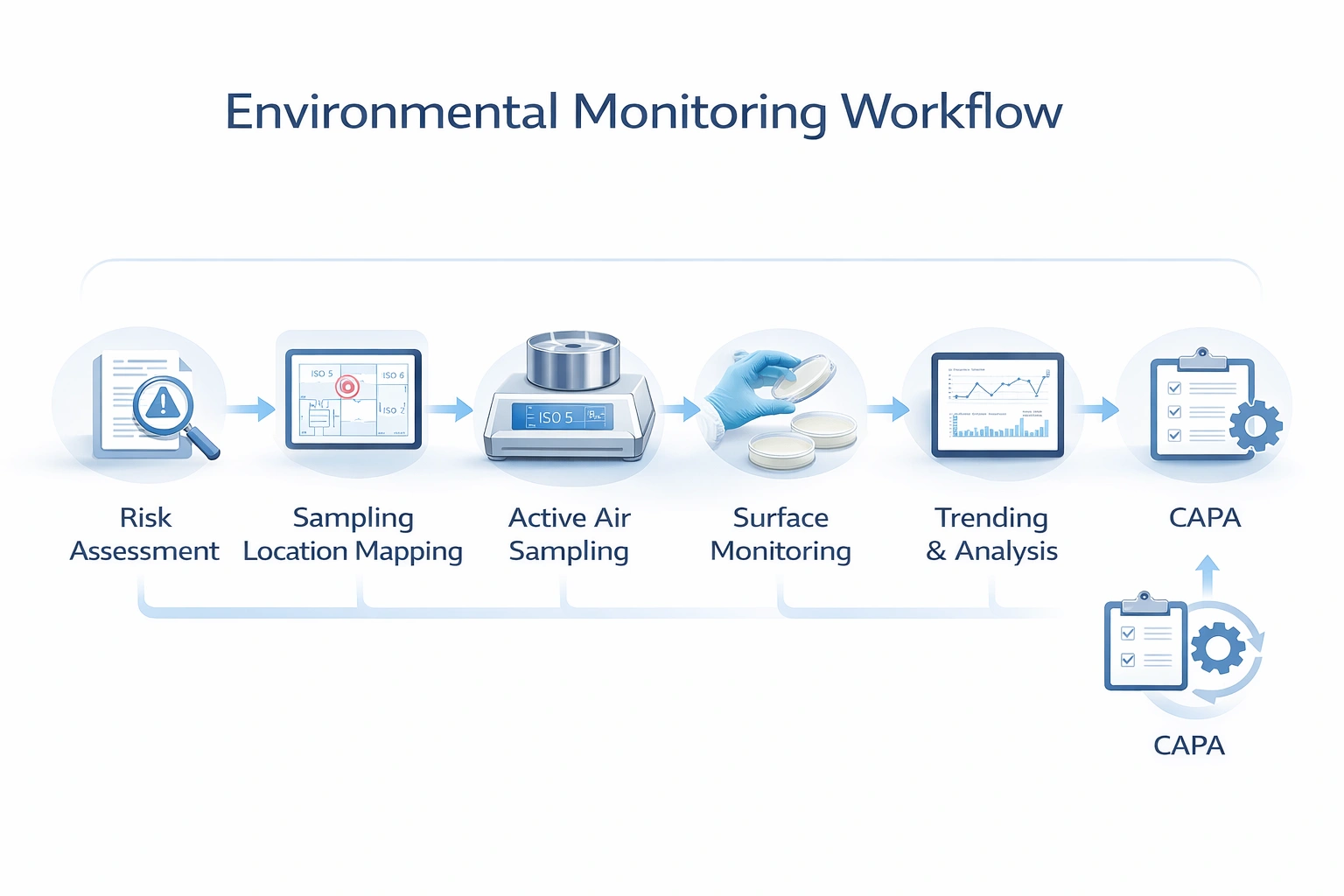
How to Define EM Locations and Sampling Frequency
Monitoring locations should never be random. They should reflect contamination risk, critical airflow paths, intervention zones, operator presence, material flow, and historical event patterns.
High-value sampling locations often include:
- Critical processing points
- Open product exposure zones
- Glove fingertips and sleeves
- Equipment surfaces near intervention sites
- Transfer hatches and material entry points
- Areas with previous excursion history
- Define rationale for each fixed location
- Differentiate between at-rest and operational monitoring
- Use higher sampling intensity in critical or high-intervention areas
- Review location relevance after layout, equipment, or process changes
Alert Limits vs Action Limits
One of the most misunderstood parts of environmental monitoring is the difference between alert limits and action limits. Alert limits are early warnings that conditions may be drifting. Action limits indicate a more serious deviation that requires defined investigation and response.
| Limit Type | Purpose | Typical Response |
|---|---|---|
| Alert limit | Signals an unusual shift or adverse trend | Review recent data, confirm repeatability, assess contributing factors |
| Action limit | Indicates significant excursion or control failure | Initiate formal investigation, define CAPA, assess product and process impact |
Well-designed limits should be risk-based and supported by historical data, room classification, process criticality, and regulatory expectations. Limits copied from another site without justification often create weak control logic.
Trending, Investigation, and CAPA
Collecting EM data is not enough. The true value of a monitoring system comes from its ability to identify patterns, trigger timely investigation, and support CAPA decisions. Trend review should look beyond single excursions and ask whether repeated signals point to structural weaknesses.
Trend review often evaluates:
- Repeated events by location
- Shift-related or personnel-related patterns
- Cleaning frequency and contamination recurrence
- Differences between room states and campaign types
- Seasonal or operational changes in microbial recovery
Visual Explanation: EM Trend Dashboard
A data dashboard can help QA teams trend recovery by room, zone, method, operator activity, and shift. This makes investigation more targeted and evidence-based.

Common Environmental Monitoring Failures
Many weak EM programs fail not because sampling is absent, but because strategy, interpretation, and follow-up are weak.
Visual Explanation: Correct vs Weak EM Practice
A clear comparison can show the difference between risk-based monitoring with defined investigation paths and generic sampling programs with poor response quality.

| Weak Practice | Why It Is Risky | Better Approach |
|---|---|---|
| Sampling without rationale | Locations may miss the highest-risk points | Use risk-based site selection tied to process and airflow |
| Only reacting to action limits | Misses early signals of deterioration | Review alert-level trends and recurring signals |
| Minimal investigation after excursion | Root causes remain unresolved | Link excursion review to personnel, airflow, cleaning, and process factors |
| No link to training or behavior | Human-factor patterns remain hidden | Cross-review EM data with gowning, training, and interventions |
What Auditors Usually Look For in an EM Program
During inspection, auditors rarely focus only on raw counts. They look at whether the full monitoring system is justified, controlled, and actionable. That includes site selection, frequency rationale, limit setting, investigation quality, and CAPA follow-through.
Auditors often request:
- EM program SOPs and sampling maps
- Alert and action limit rationale
- Trend reports by room or campaign
- Excursion investigations and CAPA records
- Evidence linking EM to contamination control strategy
- Correlation with training, cleaning, airflow, and intervention control
Best-Practice Model for a High-Performance EM Program
The strongest environmental monitoring systems share several features:
- Risk-based sampling design
- Clear distinction between viable and non-viable goals
- Defined alert and action logic
- Routine trend review, not just event review
- Cross-functional investigation involving QA, operations, and microbiology
- Direct linkage to personnel training, cleaning, airflow performance, and audit readiness
Environmental monitoring works best when it is part of a wider contamination control knowledge system rather than a standalone microbiology activity.
Support a Stronger Environmental Monitoring Program
Explore GMP-focused cleanroom solutions designed to support contamination control, audit readiness, and more consistent cleanroom operations in pharmaceutical and controlled manufacturing environments.
Frequently Asked Questions
What is environmental monitoring in a cleanroom?
Environmental monitoring is a GMP program used to detect, trend, and investigate viable and non-viable contamination in controlled environments.
What is the difference between viable and non-viable monitoring?
Non-viable monitoring measures airborne particles, while viable monitoring detects microorganisms through methods such as active air sampling, settle plates, and surface sampling.
Why are alert and action limits important?
Alert limits indicate early drift, while action limits signal a more serious excursion that requires investigation and documented response.
How should EM sampling locations be chosen?
Locations should be risk-based and linked to critical process points, airflow behavior, personnel activity, intervention zones, and historical excursion patterns.
What does a weak EM program usually look like?
Weak programs often sample without rationale, fail to trend data effectively, investigate poorly, and do not connect results to cleaning, behavior, or process controls.
Does environmental monitoring replace room classification?
No. Room classification and environmental monitoring are related but different. Classification defines expected cleanliness, while EM checks whether control remains effective during use.
How does EM connect to personnel training?
Human behavior often affects EM outcomes. Repeated excursions may point to gowning, movement, intervention, or aseptic discipline issues that require retraining.
What do auditors expect from an EM program?
Auditors typically expect clear sampling rationale, trend analysis, alert and action limit logic, investigation quality, CAPA follow-through, and linkage to contamination control strategy.



