A technical decision framework for QA and Procurement teams navigating GMP, ISO 14644, and contamination control strategies.
Under EU GMP Annex 1 and ISO cleanroom standards, cleaning tools are no longer treated as generic consumables. They are considered part of the Contamination Control Strategy (CCS), directly influencing sterility assurance and audit outcomes.
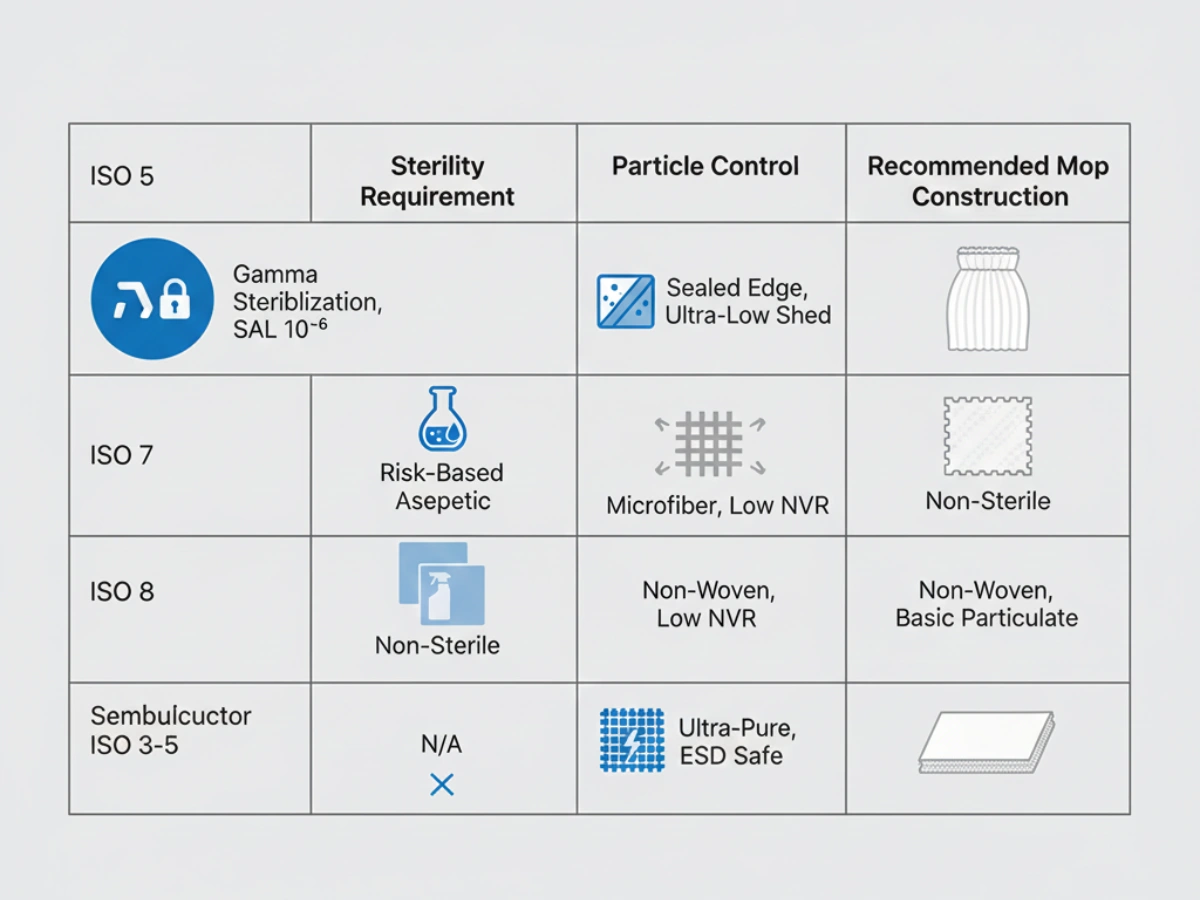
A mop suitable for a Semiconductor ISO 5 environment may be unacceptable in a Pharmaceutical Grade B zone. Selection must therefore be driven by industry risk profile و cleanroom classification.
Use the framework below to access grade-specific and industry-specific mop selection guidance.
Annex 1, Grade A–D, sterile vs controlled non-sterile mops
Low endotoxin, NVR control, ISO Class 5 environments
Particle shedding, ESD risk, ISO Class 3–7
ISO 13485 compliance and cost-efficient contamination control
Audit readiness, change control, batch genealogy
SAL 10⁻⁶, gamma irradiation, aseptic transfer
ISO 14644-1 limits and mop construction logic
Federal Standard 209E mapping to modern ISO grades


In pharmaceutical and aseptic processing, mopping is a sterility assurance activity— not a housekeeping task.
Next Step for QA Managers:
Request a supplier qualification checklist covering endotoxin,
NVR, sterility assurance, and batch traceability.


سوف نتصل بك في غضون يوم عمل واحد ، يرجى الانتباه إلى البريد الإلكتروني الذي يحتوي على اللاحقة "*@midposi.com".