A QA-focused technical guide for GMP material entry into Grade A/B zones—covering peel-and-pass logic, packaging integrity risks, and audit-defensible controls aligned with EU GMP Annex 1 expectations.

Sterility is achieved during manufacturing, but most contamination events occur during aseptic transfer—when materials move from uncontrolled areas to Grade C, then B, and finally A. Without redundant barriers, the mop packaging surface can act as a carrier for particulates and bioburden.
In practice, sterile mop packaging functions as a controlled boundary: the outer bag is stripped at the interface, while the inner bag remains sterile until the point of use. This is the operational basis of peel-and-pass.
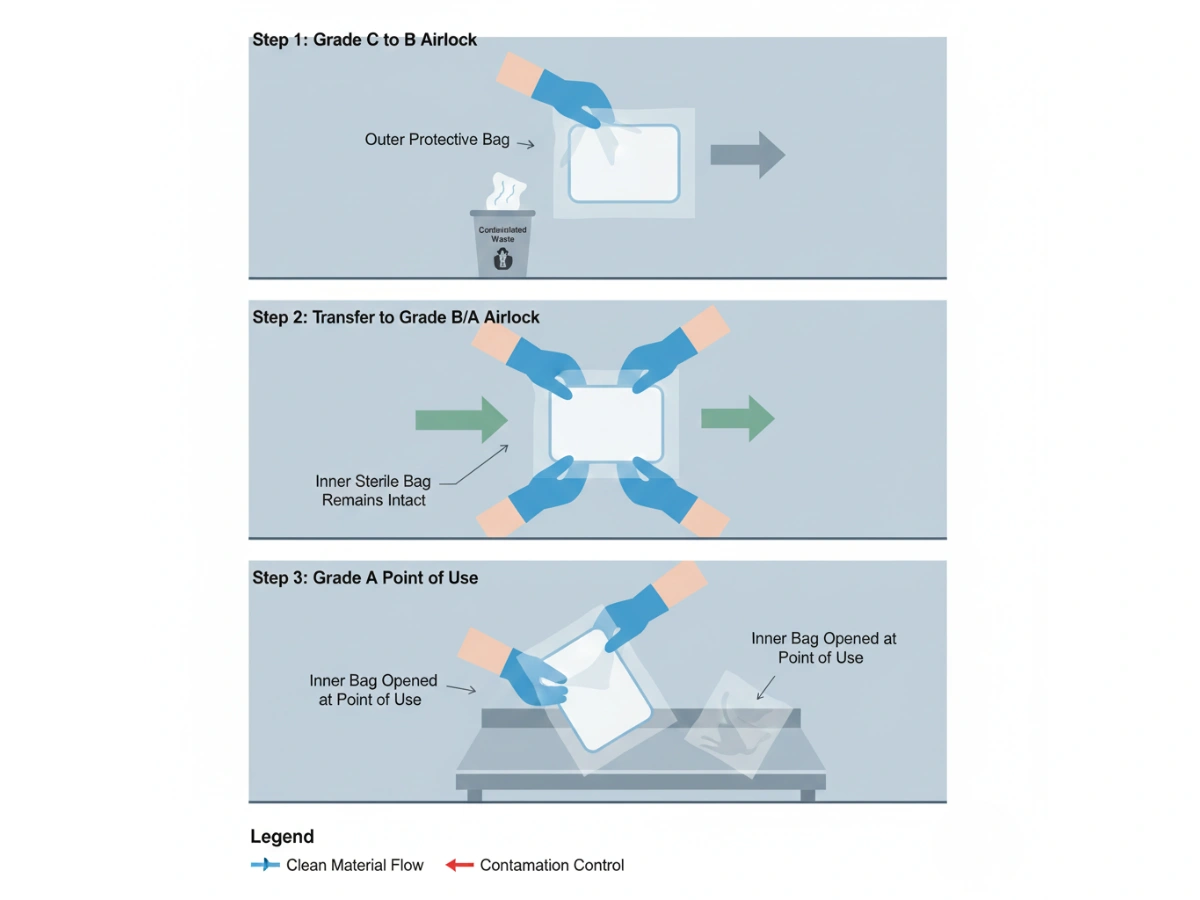

For Grade A/B operations, the core question is not “sterile at release,” but “sterile at point-of-use.” Double-bagging reduces transfer variability by removing the external surface at the boundary rather than relying on wipe-down success every time.

Use this simplified logic to align packaging selection with grade, transfer method, and validation burden. For Grade A/B, an engineered barrier is typically preferred over manual disinfection dependence.
| Decision Factor | Single-Bag Packaging | Double-Bag Packaging |
|---|---|---|
| Transfer control | Requires validated wipe-down and discipline | Peel-and-pass removes contamination at boundary |
| Failure mode | Single point of failure | Redundant sterile barrier layers |
| Audit defensibility | Heavily dependent on SOP execution evidence | Stronger engineered control narrative for CCS |
| Operational workload | Higher (disinfection, monitoring, deviations) | Lower (standardized strip-at-boundary process) |



سوف نتصل بك في غضون يوم عمل واحد ، يرجى الانتباه إلى البريد الإلكتروني الذي يحتوي على اللاحقة "*@midposi.com".