Cleanroom Quality Engineering
The Future of Cleanroom Decontamination: From Reactive Cleaning to Predictive Quality Assurance
This guide reframes decontamination as a measurable quality system—combining digital monitoring, predictive analytics, and validation-ready documentation to reduce risk, improve efficiency, and strengthen audit readiness in batch processing facilities.

1) Introduction
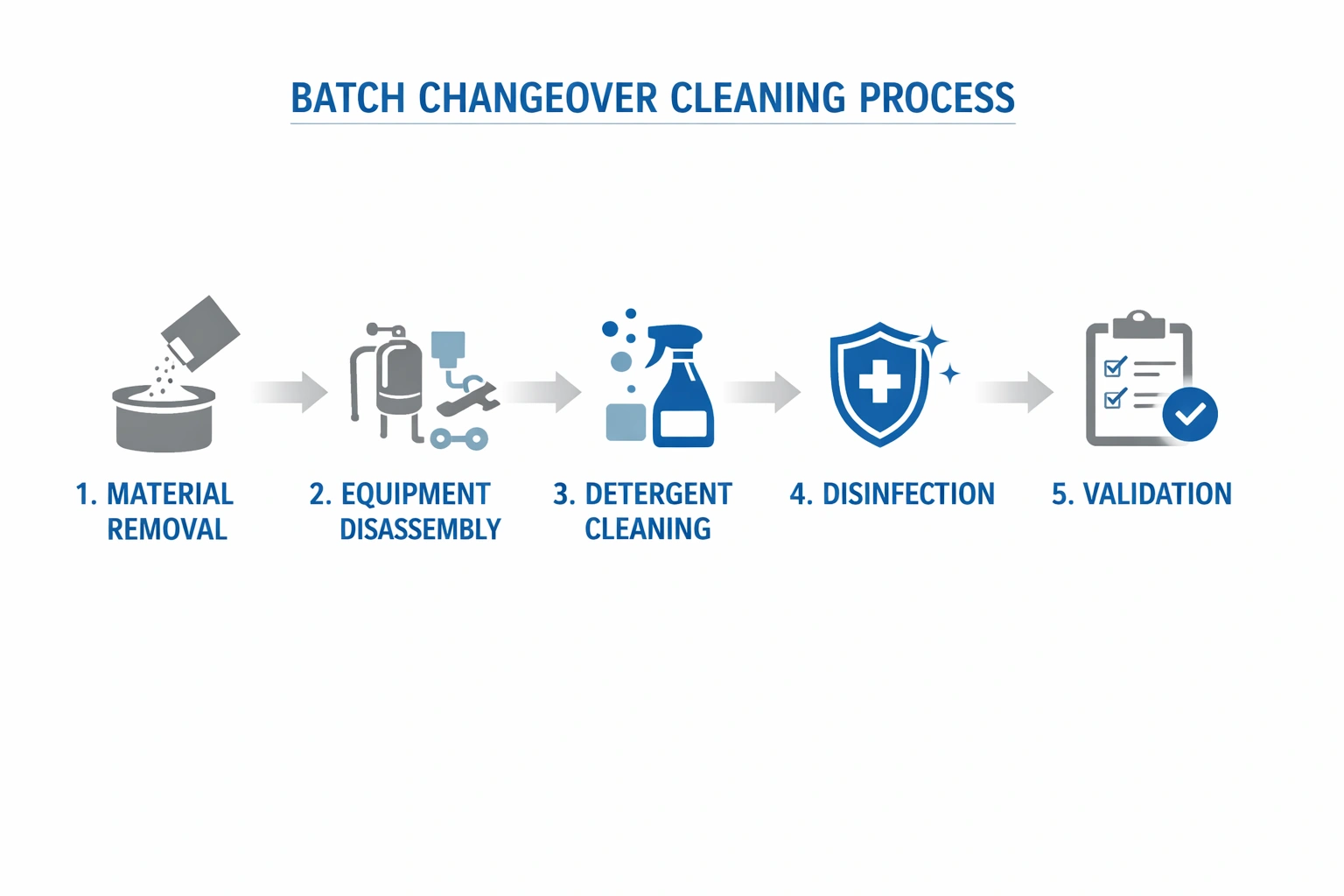
Cleanroom decontamination is at an inflection point. While pharmaceutical manufacturing moves toward Industry 4.0, many decontamination programs still rely on manual records and reactive verification. That gap creates avoidable risk: preventable contamination events, investigation burden, and audit pressure.
2) The Digital Transformation Imperative
Beyond paper: digital convergence
Traditional programs suffer from information silos, limited trend visibility, and reactive response. Digital convergence addresses this through continuous monitoring, integrated data, and analytics-driven control.

Data quality continuum
- Acquisition: validated sensors, automated sampling, barcode/RFID material tracking.
- Integration: unified data layer + ERP/MES/QMS connectivity and audit-ready trails.
- Analytics: SPC, anomaly detection, deviation root cause acceleration.
- Utilization: decision support, prescriptive work instructions, continuous improvement loops.
3) Predictive Decontamination: The New Paradigm
Predictive decontamination anticipates issues before they materialize by combining production context, environmental trends, equipment health, and execution behaviors into a dynamic risk model.

The four pillars
- Predictive risk assessment: trend + pattern recognition for early intervention.
- Prescriptive optimization: recommended cleaning paths, staffing, and chemical volumes.
- Autonomous QA: self-monitoring, self-correcting workflows and compliance evidence.
- Digital twin simulation: “what-if” planning, training, and validation acceleration.
4) Advanced Decontamination Technologies
Next-generation disinfection methods improve efficacy, reduce residues, and strengthen repeatability. Hybrid approaches also reduce manual variability and support stronger documentation.

Examples of innovation areas
- HPV / VHP evolution: smarter dosing, reduced residues, improved distribution controls.
- Electrostatic spraying: more uniform coverage, less chemical usage, faster turnarounds.
- Non-chemical methods: dry ice blasting for complex equipment where suitable.
- Sustainability: reduced water/chemical footprint with validated performance.
5) Human Factors Engineering in Decontamination
The strongest SOP still fails if it overloads operators. Human-centered design reduces cognitive load, improves ergonomics, and limits the probability of hidden deviations.

High-impact practices
- Digital work instructions with in-process checks and error-proofing.
- Role-based training with performance verification and consistent documentation behavior.
- Fatigue-aware scheduling and standardized tool selection for repeatability.
6) Regulatory Compliance in the Digital Age
Compliance documentation is shifting from “after-the-fact reporting” to “continuous evidence.” Systems that automatically time-stamp actions, lock records, and integrate with QMS reduce audit friction.
7) ROI: Why this becomes a strategic advantage
A predictive decontamination system reduces deviations, compresses changeover time, stabilizes environmental performance, and strengthens batch release confidence—creating measurable return beyond compliance.
- Lower contamination event probability and investigation workload.
- More stable throughput through optimized scheduling and resource planning.
- Better audit readiness with traceable, consistent execution evidence.
8) Conclusion
Cleanroom decontamination is no longer “just cleaning.” The next generation approach is a digital, predictive QA capability—validated, measurable, and continuously improving.
Need a validation-ready cleaning program?
Share your cleanroom class, disinfectant strategy, and changeover frequency. We’ll recommend a decontamination approach with documentation support (SOP-ready + audit-friendly).



