Cleanroom Mop Bucket System Guide: GMP Setup, Materials, Wringer Options and Workflow
The cleanroom mop bucket is not a commodity janitorial supply. In a controlled environment, it becomes part of the contamination control process. The way a bucket separates disinfectant, rinse water, and waste liquid can affect disinfectant concentration, floor contact time, operator technique, and the repeatability of the entire cleaning workflow.
This guide explains how to compare single-, dual-, and triple-bucket systems, when to choose stainless steel or cleanroom-grade polypropylene, how wringer design affects cleaning performance, and how to fit the bucket system into a practical GMP cleaning SOP.

Cleanroom Mop Bucket System: What Buyers Should Check First
A cleanroom mop bucket system should be evaluated as part of the cleaning process, not as a standalone cart. The key question is whether the system helps operators maintain clean-to-dirty separation while following a consistent procedure.
For most pharmaceutical, biotech, medical device, and controlled manufacturing facilities, the first review should focus on the bucket configuration, bucket material, wringer design, drainability, labeling, and compatibility with written SOPs.
Single-, dual-, and triple-bucket systems provide different levels of fluid separation and contamination control.
SS316 stainless steel is preferred for long service life; cleanroom-grade PP is lighter and easier to handle.
Press-type wringers have fewer moving parts, while roller wringers may provide stronger liquid extraction.
The system should support repeatable disinfectant use, mop change-out, waste handling, and operator training.
What Makes a Cleanroom Mop Bucket Different?
Why Standard Commercial Buckets Fail in Cleanrooms
Standard janitorial mop buckets are designed for general cleaning speed and convenience. Cleanroom mop bucket systems are designed for controlled cleaning, clean-to-dirty separation, and repeatable use inside classified or controlled areas.
A common commercial bucket may include rough plastic surfaces, exposed seams, open joints, low-grade casters, or wringer parts that are difficult to clean. These design details can trap residue, shed particles, or make it hard to verify that the equipment has been properly cleaned before the next use.
In cleanrooms, the bucket must support the cleaning process instead of becoming another source of contamination. Important design points include smooth surfaces, cleanable joints, chemical resistance, proper drainage, and compatibility with the facility’s disinfectant rotation.
GMP and ISO 14644 Contamination-Control Expectations
Cleanroom cleaning tools should not compromise the controlled environment they are meant to protect. In practice, this means bucket systems should minimize particle generation, support written cleaning procedures, and help operators keep disinfectant, rinse water, and dirty waste liquid separated.
ISO-classified rooms and GMP-related facilities often require documented cleaning methods, defined disinfectant contact time, controlled mop change-out, and regular review of environmental monitoring trends. A suitable mop bucket system helps make these routines easier to repeat and easier to explain during internal or customer audits.
Design Differences: Welds, Joints, Drainability and Surface Finish
A cleanroom bucket system should use smooth, easy-to-clean surfaces and avoid unnecessary crevices. For stainless steel systems, continuous welds, rounded internal corners, and polished surfaces are preferred. For polypropylene systems, smooth molded surfaces and cleanable edges are important.
Drainability is also important. If liquid remains trapped in the bottom of a bucket, in a corner, or around the wringer area, the system becomes harder to clean and harder to control. A good system should allow operators to empty, rinse, dry, and inspect the bucket without complicated handling.
If you are comparing bucket materials together with mop frames, handles, and mop heads, see MIDPOSI’s complete
cleanroom mop system guide
for a broader view of how each component works together in a controlled cleaning workflow.
Cleanroom Mop Bucket Configurations: Single-, Dual- and Triple-Bucket Systems
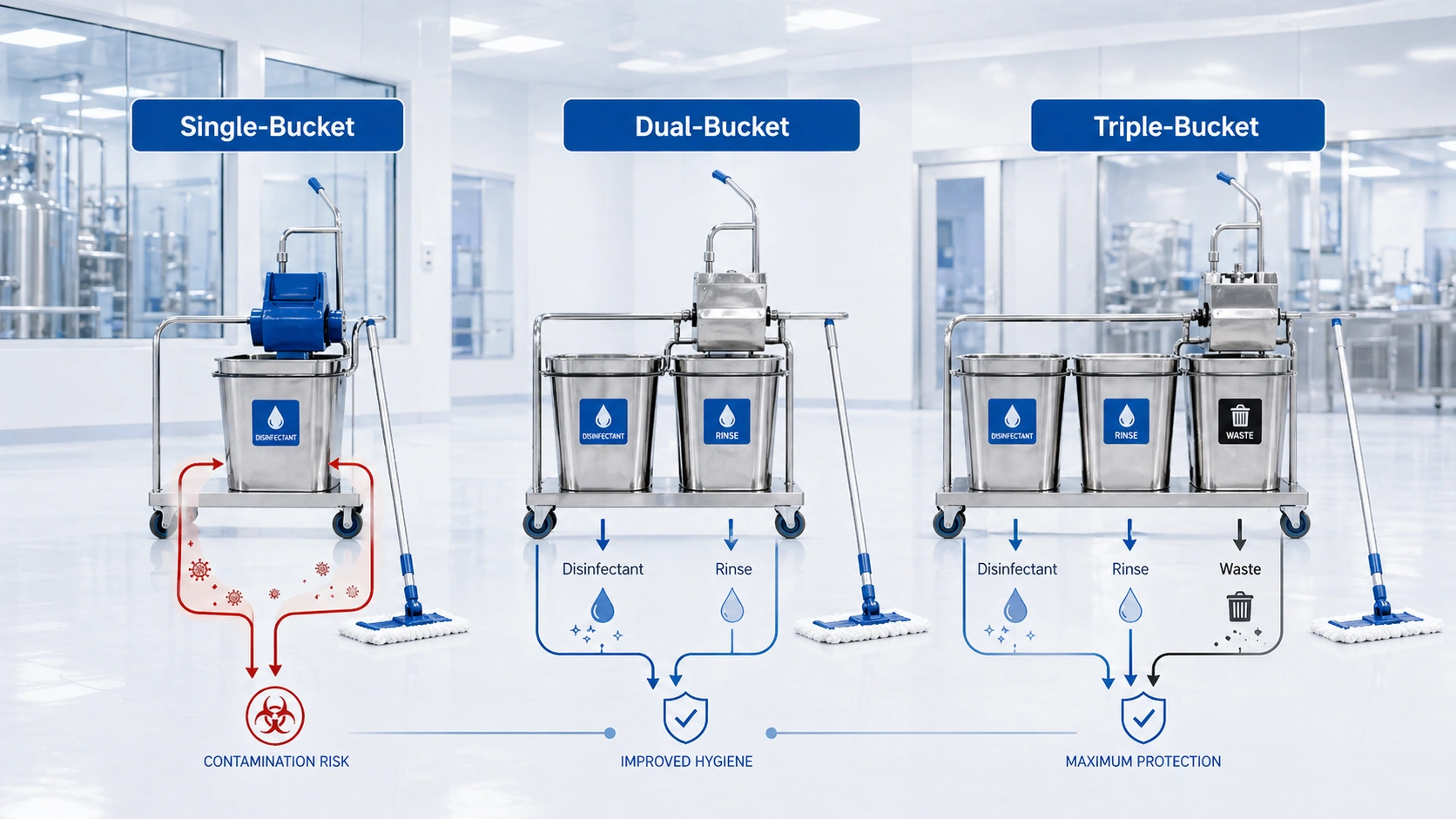
Single-Bucket System: Limited Use in Controlled Areas
A single-bucket system uses one bucket for the cleaning solution. The mop is dipped, applied to the surface, and then returned to the same bucket. This is simple, but it also allows soil and removed contamination to return to the cleaning liquid.
For non-critical support spaces, this may be acceptable when the risk level is low and the procedure is clearly defined. For GMP-related cleanroom cleaning, however, a single-bucket process is usually difficult to justify because it provides weak separation between clean solution and dirty return liquid.
Dual-Bucket System: Common for Controlled Cleaning
A dual-bucket system separates the active cleaning or disinfectant solution from rinse water. Operators apply solution from the clean bucket, rinse the mop in the second bucket, wring the mop, and then return to the clean solution.
This configuration gives better control than a single bucket because dirty liquid is not returned directly into the disinfectant bucket. It is often used in controlled areas where the cleaning risk is moderate and the facility has clear change-out rules for rinse water and mop heads.
Triple-Bucket System: Strongest Fluid Segregation
A triple-bucket system separates disinfectant, rinse water, and waste liquid. The waste bucket receives liquid wrung from the mop, which helps prevent contaminated liquid from mixing back into the disinfectant or rinse bucket.
For higher-risk pharmaceutical, biotech, aseptic support, and GMP cleaning workflows, this layout provides the strongest process control. It is easier to explain because each bucket has a clear function: one for active solution, one for rinse, and one for waste collection.
Single-Bucket
Lowest complexity, but the weakest separation. Best reserved for low-risk or non-critical support areas where the procedure does not require strong fluid segregation.
Dual-Bucket
Separates clean solution and rinse water. Suitable for many controlled cleaning routines when rinse water change-out and mop handling are properly defined.
Triple-Bucket
Separates disinfectant, rinse water, and waste liquid. Preferred when contamination control, disinfectant stability, and process repeatability are priorities.
How Bucket Configuration Affects Bioburden Control and Disinfectant Use
Bucket configuration affects how quickly the cleaning liquid becomes loaded with soil, residue, and removed bioburden. The more separation the system provides, the easier it is to keep the active solution under control during the cleaning cycle.
A triple-bucket system can reduce unnecessary dilution of the active solution because waste liquid is sent to a dedicated bucket. This can also help facilities define more practical rules for solution change-out, waste handling, and cleaning coverage per prepared batch.
Wringer vs Press Systems

Roller Wringer
Roller wringers use rollers to squeeze liquid from the mop head. They can provide efficient liquid extraction and are useful when operators need faster handling during larger-area cleaning.
The trade-off is that roller systems may include more moving parts. Bearings, shafts, rollers, and connection points should be reviewed carefully for cleanability, corrosion resistance, and particle generation risk.
Press-Type Wringer
Press-type wringers use a basket, plate, or compression mechanism to remove liquid from the mop head. They are often simpler in construction and may be easier to clean and inspect.
For higher-grade or more sensitive cleanroom workflows, a press-type wringer can be attractive because it uses fewer moving components. The final choice should depend on the mop head type, cleaning area, operator ergonomics, and sterilization requirements.
How Mop Buckets Reduce Contamination
Separation of Clean and Dirty Fluids
The main contamination-control function of a mop bucket system is fluid separation. Clean disinfectant should remain as clean and consistent as possible during the cleaning cycle, while contaminated liquid should be directed away from the active solution.
In a well-designed triple-bucket system, operators can follow a simple logic: apply clean solution, remove dirty liquid into waste, rinse the mop, remove rinse liquid into waste, and return to clean solution. This makes the workflow easier to train and easier to audit.
Preventing Disinfectant Dilution and Residue Re-Deposition
Disinfectant dilution is a practical risk in any cleaning process that sends used liquid back into the same solution. When contaminated liquid returns to the active disinfectant, the concentration and cleanliness of the solution become harder to control.
Clear separation between disinfectant, rinse water, and waste helps reduce this problem. It also helps prevent the mop head from carrying residue back onto surfaces that have already been cleaned.
Minimizing Operator-Induced Particle Generation
Bucket design also affects operator movement. If a cart is unstable, difficult to push, or awkward to wring, operators may use excessive force or repeat unnecessary movements. In cleanrooms, those movements can contribute to particle generation.
Smooth-rolling casters, stable frames, appropriate wringer height, and clear bucket positioning help operators clean with less unnecessary motion. This is especially important when working in full cleanroom gowning.
Validating Bucket Systems During Environmental Monitoring
Environmental monitoring data can help confirm whether the cleaning process is working as intended. If particle or microbial results trend upward after cleaning, the facility should review not only the disinfectant but also the mop head, bucket system, wringer, operator technique, and change-out frequency.
A good bucket system does not replace validation. It supports validation by making the cleaning process more consistent and easier to repeat.
Recommended Bucket Materials: Stainless Steel vs Polypropylene

SS304 and SS316 Stainless Steel
Stainless steel bucket systems are commonly selected for facilities that need long service life, repeated cleaning, and strong mechanical durability. SS316 is often preferred when the system may contact stronger disinfectants or when corrosion resistance is a key requirement.
Stainless steel systems should be reviewed for weld quality, surface finish, cleanability, caster design, and whether the wringer and frame are also suitable for the intended cleaning routine.
Cleanroom-Grade Polypropylene
Polypropylene bucket systems are lightweight, easy to move, and cost-effective. They can be useful for facilities that need practical handling, multiple dedicated sets, or lower initial investment.
The main point is service life. PP buckets should be inspected regularly for cracking, whitening, warping, surface damage, or loss of fit around lids and wringer components.
Chemical Compatibility Matrix
| Disinfectant or Process | SS316 Stainless Steel | Cleanroom-Grade PP |
|---|---|---|
| 70% IPA | Generally compatible when surfaces are properly maintained. | Generally compatible at normal room temperature. |
| Hydrogen Peroxide | Good compatibility, depending on concentration and exposure time. | Good compatibility for many controlled cleaning applications. |
| Sodium Hypochlorite | Requires careful control because chloride exposure can increase corrosion risk. | No pitting corrosion risk, but repeated oxidizer exposure may age the polymer. |
| Autoclave Exposure | Strong long-term durability when system components are designed for steam exposure. | Can be suitable depending on resin grade and cycle frequency, but requires regular inspection. |
| Weight and Handling | Heavier, more durable, and more stable. | Lighter and easier for operators to move. |
| Typical Selection Logic | Best for long service life and high-use GMP cleaning programs. | Best for lightweight handling, dedicated sets, or lower initial investment. |
How Material Choice Affects Long-Term Cost
Stainless steel usually costs more at the beginning, but it may provide better long-term value when the facility uses the system frequently and needs a durable setup. PP can be more economical when the use case is lower volume, the system is dedicated by area, or the facility prefers lighter handling.
The best choice is not only about purchase price. It should also include expected replacement frequency, cleaning method, disinfectant exposure, operator ergonomics, and the cost of requalification when equipment changes.
Ideal Mop Bucket Setup for Pharma and Biotech
Grade A/B/C/D Cleaning Tool Expectations
Higher-risk areas require stronger control over cleaning tools and cleaning workflow. In practical terms, this means dedicated tools, clear labeling, defined transfer procedures, and bucket systems that reduce the chance of mixing clean and dirty liquids.
Grade A/B support workflows often benefit from the strongest segregation approach, while Grade C/D areas may use dual- or triple-bucket systems depending on the facility’s contamination-control strategy.
Complete Bucket System Configuration
A complete mop bucket system normally includes the buckets, cart frame, wringer, casters, mop handle storage, labels, and sometimes drip shields or tool holders. Each part should be reviewed as part of the same workflow.
For a triple-bucket arrangement, the most common layout is disinfectant, rinse water, and waste. The wringer should be positioned so liquid is directed into the waste bucket without splash-back.
For pharmaceutical, biotech, and medical device users, the bucket workflow should be matched with room grade, disinfectant rotation, and internal SOP requirements. For application-specific selection guidance, visit
cleanroom mop solutions for GMP facilities.
SOP Integration: Disinfectant Rotation, Mop Change-Out and Waste Handling
The SOP should state which solution is used, how it is prepared, how long it remains in use, where waste liquid goes, and how often mop heads and rinse water are changed. Without these details, even a good bucket system can be used inconsistently.
Operators should be trained on the exact sequence: where to load the mop, where to wring, when to rinse, when to change the mop head, and when to empty the waste bucket.
Recommended Bucket Sizes, Fill Levels and Label Coding
Bucket size should balance cleaning coverage and operator handling. Overfilled buckets are harder to move and more likely to spill. Underfilled buckets may require frequent interruption and refill.
Labels should identify bucket function, area or grade, solution type, preparation time, expiry time, and operator initials when required by the facility procedure.
Visual Management: Color Coding for Room Classes and Workflows

Color coding is useful because it reduces confusion during routine cleaning. A facility may code by room grade, product area, disinfectant type, or cleaning sequence. The important point is consistency: operators should be able to recognize the correct tool quickly.
Labels and color bands should be durable enough for the cleaning process. If labels fade, peel, or become unreadable, the visual management system loses its value.
Autoclave Cycle Guidance for Buckets and Components
When buckets or components are sterilized by autoclave, they should be loaded in a way that allows steam contact and drainage. Nested buckets, trapped water, and overloaded carts can interfere with a reliable cycle.
After sterilization, components should be cooled, stored, and transferred according to the facility procedure. Inspection after repeated cycles is important, especially for polymer parts and wringer components.
Validation-Friendly Workflow Checklist
- Pre-use preparation: Confirm that buckets, mop heads, handles, and wringer components are clean, dry, labeled, and suitable for the target area.
- Solution control: Record disinfectant type, concentration, preparation time, expiry time, and operator identification when required by the SOP.
- Fluid separation: Keep disinfectant, rinse water, and waste liquid clearly separated during cleaning.
- Mop change-out: Define when mop heads are replaced, such as by area cleaned, visible soil, room transition, or batch requirement.
- Waste handling: Empty and dispose of waste liquid according to the site procedure before the bucket becomes difficult to move or prone to splashing.
- Post-use cleaning: Clean, rinse, dry, inspect, and store the bucket system according to the defined workflow.
Validation-Friendly Cleaning Workflow

Step 1: Prepare the Bucket System
Verify the bucket labels, confirm the target area, and prepare the required cleaning solution. If the system uses three buckets, load disinfectant into the first bucket, rinse water into the second bucket, and leave the third bucket ready for waste collection.
Step 2: Mop with a Controlled Pattern
Apply the cleaning solution using a controlled pattern, such as overlapping S-pattern strokes. The goal is to avoid missed areas while reducing unnecessary operator movement.
Step 3: Wring into the Waste Bucket
After the mop contacts the floor, wring contaminated liquid into the waste bucket rather than returning it to the disinfectant bucket. This is the main advantage of the triple-bucket workflow.
Step 4: Rinse the Mop
Rinse the mop in the rinse bucket to release soil and residue from the mop head. After rinsing, wring again into the waste bucket.
Step 5: Reload with Fresh Disinfectant
Return the mop to the disinfectant bucket only after the rinse and waste steps are complete. This keeps the active solution cleaner for longer and makes the workflow more consistent.
Disinfectant Contact Time
The floor or surface should remain wet for the contact time defined in the facility procedure. If the surface dries too quickly, the operator may need to adjust solution volume, mopping speed, or section size.
How to Avoid Residue Pooling
Residue pooling can happen when too much liquid is applied or when the floor has low areas. Operators should avoid over-wetting and should follow the site’s procedure for managing excess liquid.
Operator Technique Factors
Good equipment cannot compensate for poor technique. Operators should avoid skipping rinse steps, walking over wet cleaned surfaces, mixing tools between areas, or continuing to use a mop head after it is visibly soiled.
When a facility needs simplified change-out and reduced laundering dependency, a
disposable cleanroom mop
can be considered for high-risk, high-turnover, or campaign-based cleaning programs.
For cleaning tasks focused on particle capture and surface contact, a
microfiber cleanroom mop
may provide better wiping performance than general-purpose mop materials.
Cleanroom Mop Bucket Selection Checklist
| Selection Point | What to Check | Why It Matters |
|---|---|---|
| Room Classification | ISO class, GMP grade, support area, production area, or non-classified area. | Higher-risk areas usually require stronger segregation and more controlled workflows. |
| Bucket Configuration | Single-, dual-, or triple-bucket arrangement. | Configuration determines how well clean and dirty fluids are separated. |
| Material | SS316, SS304, or cleanroom-grade PP. | Material affects durability, weight, cleaning compatibility, and long-term cost. |
| Wringer Type | Roller wringer or press-type wringer. | Wringer design affects liquid extraction, cleanability, and operator handling. |
| Drainability | Bucket shape, internal corners, waste handling, and post-use drying. | Poor drainability can leave residue and make inspection harder. |
| Visual Management | Labels, color coding, function identification, and area dedication. | Clear identification reduces operator error and tool mix-up. |
| SOP Compatibility | Fit with disinfectant rotation, mop change-out, and transfer procedures. | The equipment should support the written workflow rather than complicate it. |
Cleanroom Mop Bucket System FAQ
What is a cleanroom mop bucket system?
A cleanroom mop bucket system is a controlled cleaning setup used to manage disinfectant, rinse water, and waste liquid during cleanroom floor cleaning. It is designed to support cleaner workflows, reduce cross-contamination risk, and help operators follow repeatable procedures.
Is a triple-bucket system better than a dual-bucket system?
A triple-bucket system provides stronger fluid separation because it keeps waste liquid separate from both disinfectant and rinse water. A dual-bucket system may still be suitable for lower-risk or moderate-risk controlled cleaning tasks when change-out rules are clearly defined.
Should I choose stainless steel or polypropylene buckets?
Stainless steel is usually selected for durability, long service life, and frequent use. Polypropylene is often selected for lightweight handling, lower initial cost, and dedicated-area use. The right choice depends on disinfectant chemistry, cleaning frequency, sterilization method, and operator ergonomics.
Which wringer type is better for cleanroom cleaning?
Press-type wringers are often simpler and easier to inspect because they have fewer moving parts. Roller wringers may provide stronger liquid extraction but should be checked carefully for cleanability, maintenance, and particle-control requirements.
How does a cleanroom mop bucket system support GMP cleaning?
It supports GMP cleaning by making the process more controlled and repeatable. A suitable system helps operators separate clean and dirty liquids, follow defined change-out rules, manage waste, and document the cleaning workflow more clearly.
Need a Cleanroom Mop Bucket System for Your Facility?
MIDPOSI supports cleanroom cleaning programs with mop systems, bucket carts, disposable mop options, microfiber mop heads, and application-specific guidance for pharmaceutical, biotech, medical device, and controlled manufacturing environments.



