
Figur 1: Farmaceutisk autoklave (Consolidated SR-24C) viser kammer i rustfrit stål, digitalt kontrolpanel med flere trykmålere og dobbelthængslet dørdesign. Autoklaver validerer renrumsmoppesterilisering gennem placering af biologiske indikatorer, parametrisk overvågning (temperatur, tryk, tid) og belastningskonfigurationskvalifikation for at opnå SAL 10⁻⁶.
Hvad betyder "autoklaverbar" i renrumsrengøringsværktøjer?
Definition af autoklavering: temperatur, damp, tryk
Autoklavering er fugt-varme sterilisering ved hjælp af mættet damp under tryk for at opnå valideret mikrobiel dødelighed. Standard farmaceutiske cyklusser fungerer ved 121-134°C (250-273°F) med 15-30 psi manometertryk (103-207 kPa over atmosfærisk). Ved 121°C trænger damp ind i porøse belastninger og kondenserer på køligere overflader og frigiver latent varme, der denaturerer proteiner og forstyrrer cellulære strukturer i vegetative bakterier, sporer, svampe og vira.
Cyklusdesign målretter et Sterility Assurance Level (SAL) på 10⁻⁶ - en sandsynlighed på ≤1 ud af 1.000.000 for, at en levedygtig mikroorganisme overlever processen. Validering bruger biologiske indikatorer (BI'er), der indeholder sporer af Geobacillus stearothermophilus (tidligere Bacillus stearothermophilus), a heat-resistant organism with documented D₁₂₁-value (decimal reduction time at 121°C) typically ≥1 minute. An overkill approach delivers ≥12-log reduction (12 times the D-value) to meet SAL requirements; product-specific approaches calculate F₀ (equivalent minutes at 121°C) based on measured bioburden and D-values.
For renrumsmopper har autoklavering to funktioner: terminal sterilisering (opnåelse af SAL 10⁻⁶ for grad A/B brug) og oparbejdning mellem brug (fjernelse af biobyrde, der er opstået under rengøring, samtidig med at materialets ydeevne bevares). "Autoklaverbar" betyder, at materialet modstår gentagen udsættelse for 121°C mættet damp uden funktionsfejl - men markedsføringskrav specificerer sjældent, hvor mange cyklusser eller hvilke ydeevnemålinger, der opretholdes.
Påkrævet sterilitetssikkerhedsniveau (SAL 10⁻⁶ for aseptisk)
EU GMP Annex 1, afsnit 4.29 påbyder, at "desinfektionsmidler og rengøringsmidler skal være sterile", når de bruges i klasse A og klasse B miljøer. "Steril" betyder i regulatoriske termer demonstreret SAL 10⁻⁶ - ikke visuel renhed, ikke biobyrde <10 CFU, men probabilistisk sikkerhed valideret gennem overkill-cyklusser eller biobyrde/D-værdiberegninger.
SAL-validering for autoklaverbare mopper kræver:
Biologisk indikatorplacering: BI'er placeret ved værst tænkelige kolde steder (identificeret under varmefordelingsundersøgelser) inden for moppebelastninger. Tætpakket last, lagdelte stoffer og forseglede poser skaber udfordringer med dampgennemtrængning. Validering bekræfter, at alle BI'er er inaktiveret på tværs af på hinanden følgende kørsler.
Cyklus parametrisk overvågning: Kontinuerlig registrering af kammertemperatur, tryk og tid. Minimum eksponering (typisk 15–30 minutter ved 121°C eller 3–10 minutter ved 134°C) skal opnås på alle steder i lasten, med F₀-beregning, der bekræfter kumulativ dødelighed ≥8–12 minutter ækvivalent.
Load configuration qualification: Worst-case scenarios (maximum mop count, densest packing, containers/pouches used in practice) must be validated. A cycle that sterilizes 10 loose mop heads may fail when 20 heads are tightly stacked or wrapped in autoclave pouches.
Routine BI monitoring: Periodic biological indicator runs (frequency defined in validation protocol, often weekly or per-batch) confirm ongoing sterilization efficacy. Environmental monitoring of autoclave water quality, steam purity, and chamber bioburden supplements parametric release.
Grade C and D areas may accept disinfected (not sterilized) mops, but pharmaceutical best practice increasingly extends SAL 10⁻⁶ validation to reusable cleaning tools across all classified zones to prevent bioburden migration from lower to higher grades.
Forskellen mellem "Ren", "Steril" og "Autoklaverbar"
These terms are often conflated in procurement specifications, creating qualification gaps that surface during audits:
Clean: Visibly free from soil, particulates, and gross contamination. Achieved through laundering with pharmaceutical-grade detergents, validated rinse cycles, and clean-room drying. “Clean” mops may still harbor 10³–10⁶ CFU bioburden and generate hundreds of particles per m². Acceptable for unclassified areas; insufficient for ISO-classified cleanrooms.
Steril: Validated to SAL 10⁻⁶ via terminal sterilization (autoclaving, gamma irradiation, ethylene oxide). Microbiological testing (USP <71> sterility test) and parametric monitoring confirm absence of viable organisms. Sterile mops are required for Grade A/B areas per Annex 1 Section 4.29. Sterility degrades over time (expiration dating) and is compromised by improper storage or packaging breaches.
Autoklaverbar: Material withstands moist-heat sterilization without unacceptable degradation. This is a material property, not a sterility state. Autoclavable materials can be made sterile through validated cycles, but “autoclavable” alone does not mean “currently sterile” or “suitable for pharmaceutical use.” A consumer-grade polyester towel may survive autoclaving but generate 10,000 particles/m² and shed fibers—technically autoclavable, functionally disqualified.
Procurement specifications must separate material capability (“autoclavable to 121°C for 50 cycles per validation data”) from process outcomes (“sterilized to SAL 10⁻⁶ per validated cycle”) and performance maintenance (“particle generation <100/m² maintained through qualified service life”).
Hvorfor genanvendelige værktøjer skal modstå gentagne cyklusser
Sterile engangsmopper (gamma-bestrålede, individuelt pakket) opnår SAL 10⁻⁶ gennem leverandørvalidering og undgår fuldstændigt problemet med cyklustælling. Genanvendelige autoklaverbare systemer skal dog opretholde ydeevnen på tværs af 50-200+ cyklusser for at retfærdiggøre kapitalinvestering og driftskompleksitet.
Økonomisk driver: Reusable mops cost \$60–\$150 per head but deliver \$1–\$3 per use when amortized over 100–200 uses (including laundering and autoclave costs). Single-use mops cost \$15–\$30 per use. For a 1,000 m² Grade C area mopped 5× per week (260 uses/year), reusable systems save \$3,000–\$7,000 annually per mop. Facilities mopping large areas (pharmaceutical manufacturing suites, biologics production zones, device assembly cleanrooms) realize five-figure annual savings with reusable systems—but only if mops survive the full qualified cycle count without premature failure.
Operational driver: Facilities with in-house autoclaves and laundry can turn around reusable mops overnight, supporting daily cleaning schedules without large inventory buffers. Gamma-sterilized single-use mops require 2–4 week vendor lead times and larger safety stocks, complicating inventory management for multi-shift operations.
Regulatory driver: Annex 1 Section 4.20 defines consumable service life as “the period of time or number of cycles for which a cleanroom consumable is suitable for use.” QA must document cycle limits, degradation monitoring, and retirement criteria. Mops rated for 50 cycles but used for 80 cycles without requalification create audit findings—even if they “look fine” visually.
Misforståelser: "Autoklaverbar" ≠ "Altid egnet til aseptisk"
Marketing materials tout “autoclavable” as a premium feature, implying suitability for aseptic manufacturing. Regulatory reality is more nuanced:
Misconception 1: All autoclavable materials are equivalent. Materials survive autoclaving through different mechanisms. Polyester relies on high glass transition temperature (Tg ~250°C) and hydrophobic structure resisting steam penetration. Polypropylene uses crystalline structure and heat-stabilizing additives. Microfiber polyester/polyamide blends depend on balanced copolymer ratios—slight formulation changes cause one material to last 100 cycles, another to fail after 20. “Autoclavable” without documented cycle validation is meaningless.
Misconception 2: If it doesn’t melt, it’s qualified. Dimensional stability is necessary but insufficient. Mops may retain shape while experiencing fiber damage (increasing particle generation from 60 to 300 particles/m²), chemical degradation (losing tensile strength, becoming brittle), or residue accumulation (binding disinfectants, harboring biofilm). Performance qualification tracks particle generation, mechanical strength, and microbial cleanliness—not just visual integrity.
Misconception 3: Sterile = contamination-free forever. Sterility applies at the moment of sterilization completion. Uncontrolled cooling, non-sterile storage, or packaging breaches introduce recontamination. Annex 1 requires sterile materials to be “used immediately” or packaged/stored under validated conditions with defined hold times. Autoclaved mops removed from the chamber and left uncovered become non-sterile within hours.
Misconception 4: Grade C/D don’t need validated autoclavable mops. Mens bilag 1 eksplicit kræver sterilitet for grad A/B, udvider risikovurderinger af kontamineringskontrolstrategi (CCS) i stigende grad valideret sterilisering til grad C/D for at forhindre migration af biobyrder. Mopper, der bruges i klasse D beklædningsrum, bærer forurening til personale, som derefter går ind i klasse B produktionsområder. QA-teams, der validerer hele faciliteters rengøringsprogrammer, behandler autoklaverbart værktøj som et systemdækkende krav, ikke kun en aseptisk zonespecifikation.
Hvorfor Pharma & Biologiske faciliteter kræver autoklaverbare mopper
Overholdelsesdrivere: EU GMP Annex 1, ISO 14644, PDA TR 70
Lovgivningsrammer konvergerer på et enkelt krav: rengøringsværktøjer, der anvendes i klassificerede områder, skal valideres for ikke at indføre forurening. Autoklaverbare mopper opfylder dette gennem tre kontrolveje:
EU GMP Annex 1 (gælder august 2023) establishes the sterility baseline. Section 4.29: “Disinfectants and detergents should be sterile when used in Grade A and B areas.” Section 4.20 extends to consumables: “Materials used should be selected to minimize the generation of particles and microorganisms… The number of laundry/sterilization cycles which cleanroom garments can undergo should be defined based upon their qualification.” Autoclavable mops operationalize this by enabling validated sterilization and defined service life.
ISO 14644-5 (drift) requires that “cleaning processes shall be validated” and “materials and equipment used for cleaning shall be suitable for the cleanroom classification.” ISO 14644-18 (Assessment of Suitability of Consumables) provides the assessment framework: consumables must demonstrate controlled emissions (particles, viable organisms, chemical residues) and documented service life. Autoclavable systems meet ISO requirements by combining material qualification (low particle generation) with sterilization validation (bioburden control).
PDA Technical Report 70 (Cleaning Validation) addresses reprocessing of reusable cleaning tools. TR 70 specifies that facilities must validate laundering/sterilization processes to remove soil, residues, and bioburden to defined limits, demonstrate that reprocessing does not degrade tool performance (particle generation, absorbency, structural integrity), and establish retirement criteria (maximum cycles, inspection checkpoints). Autoclavable mop validation protocols directly implement TR 70 guidance.
Forebyggelse af overførsel af biobyrder mellem klasse A/B/C rum
Pharma facilities are zoned by contamination risk: Grade A aseptic cores (filling needles, stopper bowls) operate under unidirectional airflow with the tightest particle and microbial limits; Grade B backgrounds (rooms surrounding Grade A) provide secondary protection; Grade C supports aseptic processing preparation; Grade D covers final packaging and non-sterile compounding.
Non-sterile or inadequately sterilized mops create bioburden migration pathways:
Scenario 1: Grade C → Grade B contamination. A mop used in a Grade C material prep area (bioburden limit: ≤100 CFU/m³ active air) picks up environmental organisms (spore-forming Bacillus species, environmental molds, skin flora shed by gowned personnel). If the mop is “disinfected” with 70% IPA but not sterilized, surviving spores remain viable. The same mop used in Grade B (bioburden limit: ≤10 CFU/m³) introduces spores to floors directly adjacent to Grade A zones. Spores aerosolize during mopping, contaminate gowns, and transfer to Grade A during personnel/material entry.
Scenario 2: Cross-contamination between product lines. A facility manufactures penicillin-based antibiotics in one suite and cephalosporin antibiotics in another. Beta-lactam cross-contamination is a regulatory red-flag; even trace penicillin residues in cephalosporin products trigger allergic reactions in sensitized patients. Mops used in penicillin areas and then “cleaned” with detergent retain API residues that migrate to cephalosporin areas. Autoclave sterilization at 121°C combined with validated laundering removes residues below HPLC detection limits, breaking the cross-contamination chain.
Scenario 3: Biofilm formation in non-sterilized tools. Mops stored damp or incompletely sterilized develop biofilms—matrix-encased microbial communities resistant to disinfectants. Pseudomonas, Burkholderia, and other water-associated organisms colonize mop fibers, bucket interiors, and wringer mechanisms. Biofilm bacteria shed during use, contaminating floors and triggering environmental monitoring failures. Autoclave cycles disrupt biofilms through thermal destruction and steam penetration, preventing chronic contamination sources.
Autoclavable systems prevent these scenarios by ensuring every mop entering a classified area is at SAL 10⁻⁶, eliminating viable contamination before use.
Sikring af desinfektionsmiddelintegritet — ingen udskillelse, ingen udvaskbare produkter
Disinfectant efficacy depends on concentration, contact time, and absence of interference. Mop materials affect all three:
Particle shedding neutralizes disinfectants. Mops generating 1,000+ particles/m² release fiber fragments, textile debris, and polymer particles into disinfectant solutions. Particles adsorb quaternary ammonium compounds (quats), reducing free active concentration below validated efficacy thresholds. Hydrogen peroxide decomposes on cellulose fibers and certain polymer surfaces. Autoclavable sealed-edge polyester generates <100 particles/m², minimizing disinfectant binding and maintaining solution activity throughout the cleaning cycle.
Chemical leachables compromise sterility assurance. Some polymers release plasticizers, stabilizers, or degradation products that inactivate disinfectants or promote microbial growth. Low-quality polypropylene leaches antioxidants that neutralize peroxide-based sporicides. Microfiber polyamide components can leach nylon oligomers. Autoclavable pharmaceutical-grade materials are validated for extractables/leachables (E&L) per ICH Q3D and USP <661>, ensuring no interference with cleaning agents.
Fiber retention prevents cross-contamination. Non-autoclavable microfiber mops degrade into sticky, residue-retaining surfaces that bind API powders, cleaning agents, and microbial contamination. Validated autoclavable polyester resists residue retention through smooth fiber surfaces and hydrophobic chemistry, enabling thorough laundering and preventing carryover between uses.
Risikoreduktion for miljøovervågningsfejl
Environmental monitoring (EM) programs track particle counts, viable air/surface samples, and personnel microbial contamination. Cleaning-tool-related EM failures follow three patterns:
Particle excursions during mopping. Non-validated mops generate particle bursts when wetted, wrung, or dragged across floors. ISO Class 5 areas (≤3,520 particles ≥0.5 µm/m³) have zero margin; a mop shedding 500 particles/m² temporarily pushes floor-level counts above limits. Even if the excursion resolves within 30 minutes (HEPA filtration removes particles), it triggers investigation cycles, CAPA documentation, and potential production holds. Autoclavable low-linting polyester mops generating <50 particles/m² eliminate this failure mode.
Post-cleaning bioburden increases. EM failures occur when surface swabs or contact plates show higher CFU counts after cleaning than before—evidence that mops redistributed contamination rather than removing it. Root causes: inadequate sterilization (mops harboring bioburden), biofilm growth in mop systems, or disinfectant inactivation by contaminated tools. Autoclave-validated mops at SAL 10⁻⁶ eliminate the “contaminated tool” variable, simplifying investigations and reducing false-positive findings.
Chronic low-level bioburden trends. Facilities using non-sterilized reusable mops often see persistent Grade C/D bioburden (30–50 CFU/m³ air samples, 10–20 CFU contact plates) that resists improvement despite validated disinfection protocols. The source: mops accumulating resistant organisms (spore-formers, biofilm species) through incomplete sterilization. Switching to autoclavable systems with validated 121°C cycles typically reduces baseline bioburden 50–70%, improving EM compliance margins.
Valideringskrav: Varmemodstand, partikelafgivelse
Pharma QA teams qualifying autoclavable mops must document four performance attributes:
1. Heat resistance across qualified cycle count. IQ/OQ/PQ protocols include:
- IQ: Verify material specifications (polyester fiber type, sealed-edge construction, frame/handle materials), confirm autoclave calibration and biological indicator lot qualification
- OQ: Execute 3–5 consecutive autoclave cycles at 121°C/30 min with mop loads, place BIs at cold spots (center of tightly packed mop bundles), confirm all BIs inactivated and parametric limits met (minimum F₀ ≥8 min)
- PQ: Test mop performance at 0 cycles (new), 25–30 cycles (mid-life), and 50–100 cycles (end of qualified life); measure particle generation per ISO 14644-14, visual inspection (tears, edge degradation, discoloration), mechanical strength (tensile testing, abrasion resistance)
2. Particle shedding stability. ISO 14644-14 (Assessment of Suitability for Use of Equipment by Airborne Particle Concentration) provides the test method: operate mop heads in a controlled environment with optical particle counters measuring downstream concentrations. Acceptance criteria: <100 particles ≥0.5 µm/m² for ISO Class 5–7 use, <200 particles/m² for Class 8. Qualification tests mops at start of life and after every 20–25 autoclave cycles to detect degradation. Mops exceeding particle limits are retired even if they retain physical integrity.
3. Dimensional stability and functional performance. Autoclaving causes polymer shrinkage in low-quality materials. Measure mop head dimensions (length, width, thickness) before and after 10, 30, 50, 80, and 100 cycles; define tolerance limits (typically ±5% dimensional change). Test functional parameters: fluid absorbency (mL retained per gram dry weight), wring-out efficiency (% fluid released under standardized pressure), and frame attachment security (pull force required to detach mop from frame). Degradation beyond defined limits triggers retirement.
4. Disinfectant compatibility post-autoclave. Pharmaceutical cleaning programs rotate disinfectants (IPA, quats, peroxide, bleach) to prevent microbial resistance. Validation tests simulate worst-case sequential exposure: autoclave → 70% IPA → autoclave → 5% H₂O₂ → autoclave → 1000 ppm bleach → autoclave, repeating through 50 cycles. Confirm particle generation, visual integrity, and mechanical strength remain within acceptance criteria. Materials that pass autoclave-only testing but fail combined autoclave + chemical exposure are disqualified.

Figure 2: Sealed-edge polyester mop head showing heat-sealed perimeter construction and aluminum frame attachment. Sealed edges prevent fiber shedding and particle generation, maintaining <100 particles/m² through 50–100 autoclave cycles at 121°C. This construction is critical for pharmaceutical GMP compliance per ISO 14644-14 particle generation requirements.
Materialer egnet til autoklavering (puder + rammer + håndtag)
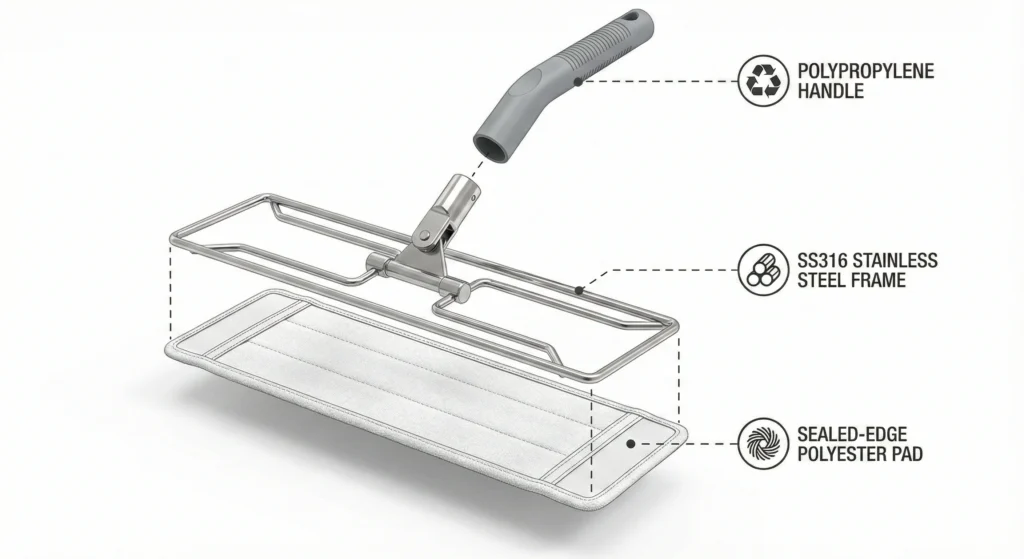
Moppepuder i polyester med forseglet kant
Polyester dominates pharmaceutical autoclavable mop heads because it uniquely combines heat resistance, chemical compatibility, and low particle generation.
Material chemistry: Polyethylene terephthalate (PET) polyester has a glass transition temperature (Tg) of ~80°C and melting point of ~260°C. At autoclave temperatures (121–134°C), polyester remains in its solid crystalline state, well below melting. The polymer’s aromatic structure and hydrophobic ester linkages resist hydrolysis from steam exposure. High-molecular-weight continuous-filament polyester (used in pharmaceutical-grade mops) maintains tensile strength >70% after 100 autoclave cycles.
Sealed-edge construction: Critical for particle control. Cut edges expose fiber ends that fray and shed particles during mopping. Pharmaceutical-grade mop pads use heat-sealed perimeters (laser-cut and thermally bonded) or ultrasonic welding to encapsulate all edges. Some designs use continuous-loop knitting (tubular construction with no cut edges). Sealed edges prevent unraveling and particle generation, maintaining <80 particles/m² through 50–100 cycles.
Performance specifications:
- Autoklave holdbarhed: 50–100 cycles at 121°C/30 min (standard polyester), 100–150 cycles (premium continuous-filament polyester)
- Partikelgenerering: <50 particles ≥0.5 µm/m² (new), <100 particles/m² (after 50 cycles)
- Kemisk kompatibilitet: Resistant to 70% IPA, 3–10% H₂O₂, quats up to 2000 ppm, sodium hypochlorite up to 1% (higher bleach concentrations cause gradual yellowing and strength loss but maintain <100 particles/m² through qualified life)
- Absorberingsevne: 4–6× dry weight (lower than microfiber but sufficient for pharmaceutical disinfectant application)
- Koste: \$60–\$150 per mop head (40–60 cm width)
Procurement specifications: “100% polyester or polyester-dominant blend (≥90%), continuous-filament construction, sealed edges (heat-sealed, ultrasonically welded, or continuous-loop knit), qualified for 50–100 autoclave cycles at 121°C per vendor validation data, particle generation <100/m² per ISO 14644-14 maintained through qualified service life.”
Mikrofiberblandinger (Forsigtig & Kvalifikationsbehov)
Microfiber mops (polyester/polyamide blends, typically 80/20 or 70/30) offer superior absorbency (6–8× dry weight) and soil capture compared to 100% polyester. However, microfiber presents autoclave validation challenges:
Polyamide (nylon) degradation: Polyamide 6 and 6,6 (common microfiber components) have lower heat resistance than polyester. Nylon Tg is 50–60°C with melting points of 220–260°C—closer to autoclave temperatures. Repeated 121°C exposure causes chain scission (molecular weight reduction), embrittlement, and fiber fusion. SEM imaging of microfiber after 20 autoclave cycles shows filament clumping and surface damage.
Accelerated particle generation: Studies document microfiber particle generation increases from ~100 particles/m² (new) to 300–800 particles/m² after 20–40 autoclave cycles—disqualifying performance for ISO Class 5–7 environments. The split-fiber structure (microfibers are mechanically split to create fissures for soil capture) delaminates under thermal stress, releasing sub-micron fiber fragments.
Residue retention: Damaged microfiber becomes tacky, retaining disinfectant residues, API powders, and organic soil. This increases bioburden risk (organic residues support microbial growth) and cross-contamination potential (residues transfer between cleaning cycles).
Limited service life: Pharmaceutical-grade microfiber qualified for autoclaving typically achieves 30–50 cycles—lower than polyester’s 50–100 cycles. Cost per use remains competitive due to lower unit cost (\$25–\$80 vs \$60–\$150 for polyester), but facilities must track cycle counts rigorously and retire mops earlier.
Recommended applications: Microfiber autoclavable mops are acceptable for ISO Class 7–8 (Grade C/D) areas where higher absorbency benefits outweigh reduced service life, and particle generation <200/m² meets classification requirements. Grade A/B applications should use polyester; the risk of mid-service-life particle excursions with microfiber is unacceptable in aseptic zones.
Qualification requirements: Demand vendor validation data showing particle generation curves from 0 to qualified end-of-life cycles (e.g., 0, 10, 20, 30, 40, 50 cycles), visual/SEM evidence of fiber integrity, and dimensional stability testing. Establish in-house cycle tracking and inspection protocols to detect early degradation.
Rammer i rustfrit stål
Mop frames connect heads to handles and must survive 200+ autoclave cycles without corrosion, warping, or mechanical failure.
Material specifications: SS316 (18% chromium, 14% nickel, 2.5% molybdenum) or SS304 (18% chromium, 8% nickel) stainless steel. SS316 offers superior corrosion resistance in high-chloride environments (important for facilities using bleach-based sporicides); SS304 is acceptable for IPA/quat/peroxide-only programs.
Konstruktion: Continuous-bend or welded one-piece frames eliminate threaded connections and crevices that trap bioburden. Mop head attachment should use sealed pockets (polyester sleeves) or smooth clip mechanisms—no exposed Velcro or hook-and-loop fasteners that harbor contamination and shed particles.
Autoklave holdbarhed: SS316/SS304 withstand 200+ autoclave cycles at 121°C without functional degradation. Passivation (chemical treatment forming a protective chromium oxide layer) may be required after 100–150 cycles if surface discoloration or micro-pitting appears, but mechanical integrity and particle generation performance remain acceptable.
Koste: \$70–\$200 per frame (40–60 cm width). Higher upfront cost than polypropylene (\$25–\$80), but amortized over 200+ cycles vs 50–100 cycles for PP, stainless steel offers lower total cost of ownership.
Procurement specifications: “SS316 or SS304 stainless steel, one-piece welded or continuous-bend construction, sealed mop head attachment (no exposed Velcro), electropolished or passivated surface finish, qualified for 200+ autoclave cycles at 121°C.”
Anodiseret aluminium rammer (begrænsninger)
Anodized aluminum frames offer lighter weight (150–250g vs 300–500g for stainless steel) and lower cost (\$40–\$120), but present qualification challenges:
Autoclave-induced corrosion: Anodization (electrochemical oxide coating) protects aluminum from oxidation, but 121°C steam gradually degrades the anodic layer. Repeated cycles cause pitting, white oxide formation, and surface roughness—increasing particle generation and creating bioburden harbors.
Limited cycle life: Pharmaceutical-grade anodized aluminum frames typically achieve 50–100 autoclave cycles before surface degradation disqualifies them. This matches polyester mop head service life, allowing synchronized retirement, but offers no advantage over polypropylene frames at similar cost.
Kemisk uforenelighed: Aluminum corrodes rapidly in alkaline environments. Facilities using alkaline detergents for laundering or sodium hypochlorite (bleach, which generates alkalinity through hydrolysis) should avoid aluminum frames. Even anodized surfaces fail under sustained bleach exposure.
Recommended applications: ISO Class 7–8 applications with IPA/quat-only disinfection programs and weight-sensitive workflows (e.g., ceiling mopping, wall cleaning requiring extended-reach handles). Grade A/B applications should use stainless steel to avoid mid-service-life corrosion risks.
Autoklaverbare polypropylenhåndtag
Handles connect frames to operators and must be autoclavable, ergonomic, and sealed against bioburden infiltration.
Material specifications: High-temperature polypropylene (PP) formulated with heat stabilizers, typically glass-transition temperature ~0°C and melting point ~165°C. Standard PP melts or warps at 121°C; pharmaceutical-grade autoclavable PP uses copolymers and stabilizers to maintain rigidity at sterilization temperatures.
Konstruktion: One-piece extrusion or injection-molded design with no hollow cavities or threaded caps that trap moisture and bioburden. Frame attachment should use sealed threaded connections with autoclavable gaskets or one-piece molded frame-handle assemblies.
Autoklave holdbarhed: High-temperature PP withstands 50–100 cycles at 121°C. Degradation manifests as brittleness, surface cracking, and thread stripping on attachment points. Visual inspection after every 20–25 cycles detects early failure signs.
Stainless steel vs polypropylene handles: SS316 handles (120–150 cm, \$80–\$150) withstand 200+ cycles and offer superior chemical resistance, but are heavier (400–600g) and costlier. Autoclavable PP handles (\$30–\$70, 150–250g) provide ergonomic benefits and adequate service life for Grade C/D applications. Facilities should match handle cycle life to frame cycle life—pairing 50-cycle PP handles with 200-cycle stainless frames creates waste; pairing 100-cycle PP with 100-cycle polyester mops optimizes synchronized retirement.
Procurement specifications: “Autoclavable polypropylene (high-temperature formulation) or SS316 stainless steel, one-piece sealed construction, qualified for ≥50 autoclave cycles at 121°C, frame attachment threads sealed with autoclavable gaskets.”
Kompatibilitet med desinfektionsmidler (IPA, H₂O₂, QAC)
Autoclavable materials must withstand pharmaceutical disinfectant rotation without cumulative degradation:
70% Isopropyl alcohol (IPA): Daily use in Grade A/B/C areas. Polyester, stainless steel, and autoclavable PP show excellent compatibility—no swelling, discoloration, or strength loss after 100+ exposure cycles. Microfiber polyamide may swell slightly but remains functional.
Hydrogen peroxide (H₂O₂, 3–10%): Weekly sporicidal cleaning. Polyester and SS316 resist peroxide oxidation. Autoclavable PP shows gradual surface chalking (whitening) after 50+ peroxide exposures but maintains mechanical integrity. Microfiber polyamide degrades faster—yellowish discoloration and strength loss after 30–50 cycles. Combined autoclave + peroxide stress accelerates microfiber degradation; limit to 30–40 total cycles in peroxide-rotation programs.
Quaternary ammonium compounds (quats, 200–2000 ppm): 2–3× weekly general disinfection. Well-tolerated by all autoclavable materials. Residue management critical—quats form films on surfaces that reduce subsequent disinfectant activity. Validated laundering protocols must remove quat residues between uses.
Natriumhypochlorit (blegemiddel, 500-5000 ppm): Bredspektret sporicid brug 1-2 gange ugentligt. Polyester tåler blegemiddel, men viser gradvis gulning og 20–30 % trækstyrketab efter 50+ eksponeringer kl. >1000 ppm. SS316 modstår kloridkorrosion; SS304 kan vise pitting efter 100+ eksponeringer over 2000 ppm. Autoklaverbar PP og mikrofiber nedbrydes begge hurtigt i blegemiddel - overfladerevner, skørhed og accelereret partikeldannelse. Faciliteter med blegemiddeltungt CCS bør specificere SS316-rammer, 100 % polyestermoppehoveder og begrænse moppens levetid til 50–70 cyklusser ved blegning >1000 ppm bruges rutinemæssigt.
Validering tilgang: Create a compatibility matrix documenting pass/fail for each material + disinfectant combination, with “pass” defined as: no visible degradation (cracking, yellowing, surface texture change beyond acceptable limits), particle generation <100/m² maintained, mechanical strength retention >70%, after 50 cycles of worst-case sequential exposure (autoclave → disinfectant → autoclave).
Autoklav temperatur & Cyklusbegrænsninger (validering forklaret)
121°C / 20–30 min standardcyklusser
The 121°C/15–30 minute cycle is pharmaceutical autoclaving’s workhorse, balancing validated lethality with material preservation.
Cycle parameters: 121°C (250°F) saturated steam at 15 psi gauge pressure (103 kPa above atmospheric), exposure time 15–30 minutes depending on load density and steam penetration requirements. F₀ (cumulative lethality equivalent to 121°C exposure) typically reaches 8–15 minutes for porous loads like mop heads.
Why 121°C: At this temperature, G. stearothermophilus spores (the biological indicator standard for steam sterilization) exhibit D-value ~1.5 minutes (time to achieve 1-log or 90% reduction). A 15-minute exposure delivers 10-log reduction; 30 minutes provides 20-log reduction—massive overkill ensuring SAL 10⁻⁶ even with high bioburden or poor steam penetration.
Material tolerance: Polyester, SS316 og autoklaverbar PP tåler alle 121°C uden akut skade. Dette er kvalifikationsgrundlaget - materialer, der fejler ved 121°C, diskvalificeres uanset omkostninger eller ydeevnefordele.
Cykelkvalifikation: IQ/OQ validerer autoklaveudstyret (temperaturfordeling, dampkvalitet, dørtætningsintegritet). PQ validerer specifikke belastninger: pak moppehoveder i worst-case-konfiguration (maksimalt antal, tætteste arrangement, i pose, hvis det bruges i praksis), placer BI'er i geometrisk centrum og periferi, kør 3 på hinanden følgende cyklusser, bekræft, at alle BI'er er inaktiverede og minimum F₀ opnået på alle sensorplaceringer. Dokumenter cyklusregistreringer (tids-/temperatur-/trykdiagrammer) og BI-resultater til regulatorisk inspektion.
134°C korte cyklusser
High-temperature short-cycle sterilization (134°C/3–10 minutes) is used in some pharmaceutical facilities to increase autoclave throughput.
Cycle parameters: 134°C (273°F) at 30 psi gauge (207 kPa above atmospheric), exposure time 3–10 minutes. F₀ equivalence: 3 minutes at 134°C delivers approximately the same lethality as 30 minutes at 121°C (due to exponential relationship between temperature and D-value per z-value ~10°C for spores).
Material risk: Higher temperature accelerates polymer degradation. Polyester maintains integrity but shows faster color fading and tensile strength loss—50-cycle life at 134°C vs 100-cycle life at 121°C. Autoclavable PP approaches its melting point (165°C); service life drops to 30–50 cycles, with warping risk if chamber temperature overshoots or cooling is too rapid. Microfiber polyamide degrades severely—fiber fusion and particle generation increases appear after 10–20 cycles at 134°C.
Recommended applications: 134°C cycles are acceptable for stainless steel frames/handles (no degradation) and premium continuous-filament polyester mops where 50-cycle life is sufficient. Avoid 134°C for microfiber, standard-grade polyester, and polypropylene components. If facility autoclaves operate at 134°C for other loads (surgical instruments, glassware), establish separate 121°C cycles for cleanroom mops or accept reduced service life and track cycle counts accordingly.
Validering: Same IQ/OQ/PQ framework as 121°C cycles. BI placement critical—134°C reduces safety margin for cold spots. Service life studies must be conducted at 134°C to determine actual cycle limits; do not extrapolate from 121°C data.
Gentagen autoklaveksponering & Aldringsstudier
Materials degrade cumulatively across autoclave cycles. Service life determination requires accelerated aging validation:
Aging study design: Test mops at intervals throughout projected service life—0 cycles (baseline), 20 cycles, 40 cycles, 60 cycles, 80 cycles, 100 cycles—measuring:
- Particle generation per ISO 14644-14 (<100/m² acceptance criterion)
- Visual integrity (tears, edge degradation, discoloration)
- Dimensional stability (length, width, thickness within ±5% of original)
- Mechanical strength (tensile test, abrasion resistance)
- Functional performance (absorbency, wring-out efficiency)
Plot performance metrics vs cycle count to identify degradation curves. Mop service life is defined as the cycle count where any parameter exceeds acceptance criteria—typically when particle generation reaches 100/m² (for ISO 5–7 use) or mechanical strength drops below 70% of original.
Typical degradation patterns:
- Polyester: Gradual yellowing after 40–60 cycles (cosmetic only, no functional impact); particle generation stable <80/m² through 80 cycles, rising to 90–100/m² at 100 cycles; tensile strength 85–90% retention at 100 cycles. Service life: 80–100 cycles (retire before particle limit breach).
- Mikrofiber: Particle generation 100–150/m² at 30 cycles, 200–400/m² at 50 cycles; fiber fusion visible under magnification after 20 cycles; absorbency drops 20–30% by 40 cycles. Service life: 30–40 cycles.
- Autoclavable PP frames: Surface cracking appears after 60–80 cycles; threads strip or crack at 80–100 cycles. Service life: 70–80 cycles (visual inspection after every 20 cycles to catch early failure).
Requalification triggers: If in-use mops show unexpected degradation (particle excursions, broken frames, premature discoloration), stop use and investigate. Possible causes: autoclave temperature overshoots, steam quality issues (superheated steam or wet steam), contamination with incompatible chemicals (e.g., bleach exposure not part of original validation), or vendor material formulation changes. Requalify using fresh samples and revise cycle limits if needed.
Krympningsrisiko i puder af lav kvalitet
Polymer shrinkage from steam exposure is a common failure mode for non-pharmaceutical-grade mops:
Mechanism: Polymers contain residual stresses from manufacturing (spinning, weaving, heat-setting). Autoclave heat provides energy for polymer chains to relax toward lower-energy conformations—manifesting as dimensional contraction. Low-quality polyester may shrink 5–15% in length/width after 5–10 cycles; pharmaceutical-grade materials use controlled heat-setting and stress-relief annealing during manufacturing to minimize residual stress, limiting shrinkage to <3% over 100 cycles.
Impact: Shrunken mop heads no longer fit frames properly, creating loose attachment points that generate particles through friction. Reduced surface area decreases floor coverage and fluid capacity. Tight fabric increases stiffness, reducing operator maneuverability and particle capture efficiency.
Qualification testing: Measure mop head dimensions (length, width, thickness at three locations) at 0, 10, 20, 30, 50 cycles. Acceptance criterion: <5% dimensional change through qualified service life. Reject vendors who cannot provide shrinkage validation data or who show >5% shrinkage in first 10 cycles (indicating inadequate manufacturing heat-treatment).
Metaldeformation i rammer uden laboratoriekvalitet
Metal frames fail through corrosion (discussed earlier under aluminum) or mechanical deformation:
Welded joint failure: Frames using spot welds or tack welds (rather than continuous welds) develop stress cracks at weld points after 30–50 autoclave cycles. Steam penetrates cracks, accelerating corrosion and joint failure. Pharmaceutical-grade frames use continuous-bead welding or one-piece stamped/bent construction.
Thread stripping: Threaded handle attachment points experience cyclic thermal expansion/contraction. Low-quality stainless steel (e.g., SS201, a lower-nickel grade marketed as “stainless”) or soft aluminum alloys strip threads after 20–40 cycles. SS316 and high-strength aluminum alloys maintain thread integrity through 200+ cycles.
Warping: Thin-gauge aluminum (<2 mm) or stamped steel frames may warp under autoclave pressure differentials, especially during rapid cooling. Warped frames create uneven floor contact (reducing cleaning efficacy) and stress mop head attachment points (causing tears).
Qualification testing: Load frames into autoclave at maximum operational density, run 20 cycles, inspect for welds cracks (dye penetrant testing), thread damage (go/no-go gauges), and warping (flatness measurement on reference surface). Acceptance: no visible damage, thread engagement >80% of original, flatness deviation <2 mm across frame length.
Sådan specificeres autoklavevalidering korrekt i indkøb
Procurement specifications must separate vendor marketing from validation-ready documentation:
Required vendor data:
- Material composition: Exact polymer type (e.g., “PET polyester, continuous-filament, 150 denier” not “polyester blend”), fiber construction (sealed-edge method, knit pattern), metal grade (SS316, autoclavable PP formulation)
- Autoclave cycle qualification: Temperatur, tid, tryk og belastningskonfiguration brugt i leverandørvalidering; antal testede cyklusser (minimum 50 for genanvendelig kvalifikation); acceptkriterier for partikeldannelse, dimensionsstabilitet, mekanisk styrke
- Partikelgenereringsdata: ISO 14644-14 testrapporter, der viser partikelantal ved 0, 25, 50, 75, 100 cyklusser (eller leverandørens kvalificerede grænse); testbetingelser (mættet moppe, 500 g downforce, standardiseret slagmønster); specifikationer for optisk partikeltæller
- Bestemmelse af levetid: Metode, der anvendes til at fastlægge cyklusgrænser (ældningsundersøgelser, accelereret test, historiske feltdata); pensionskriterier (partikelgrænse, visuel defektliste, tærskel for mekanisk styrke)
- Overensstemmelsescertifikater: Lot-specific CoC confirming material matches specification; autoclave cycle records (if vendor pre-sterilizes); biological indicator results (if applicable)
Sample specification language:
“Autoclavable cleanroom mop head: 100% polyester continuous-filament, sealed-edge construction (heat-sealed or ultrasonically welded perimeter), qualified for minimum 50 autoclave cycles at 121°C/30 min per vendor validation study. Vendor shall provide: (1) ISO 14644-14 particle generation test report documenting <100 particles ≥0.5 µm/m² maintained through qualified cycle count, (2) dimensional stability data showing <5% length/width change through qualified cycles, (3) material certificates of analysis, (4) service life determination methodology and retirement criteria. Mop heads shall be supplied with lot-specific certificates of conformance and cycle tracking documentation enabling facility service life monitoring.”
Red flags in vendor claims:
- “Autoclavable” without specified cycle limits or validation data
- “Tested to 121°C” without duration, load configuration, or performance metrics
- Particle generation data only for new/unused mops (no aging study)
- “Suitable for cleanrooms” without ISO class or GMP grade specification
- Refusal to provide material composition or manufacturing process details
Faciliteter bør pilotteste små mængder (10-20 moppehoveder) med in-house autoklavevalidering før faciliteten overtages. QA-ledet test identificerer leverandørdatahuller og verificerer ydeevnen under faktiske brugsforhold (facilitetsspecifikke desinfektionsmidler, autoklaveudstyr, operatørteknikker).
Autoklaverbare vs engangsmopper til renrum — Sådan vælger du
Sammenligningstabel
| Kriterium | Autoklaverbar Genanvendelig | Gamma-steriliseret engangs |
| Pris pr. brug | $2–$7 (systemomkostninger amortiseret over 50-200 anvendelser + hvidvask/autoklave) | $15–$30 (engangsbrug, leverandørsteriliseret) |
| Sterilitetssikring | SAL 10⁻⁶ via in-house autoklavevalidering; kræver BI-overvågning og parametrisk frigivelse | SAL 10⁻⁶ via leverandør-gamma-bestråling (25–50 kGy); sterilitetsattester og dosisoptegnelser leveres |
| Partikelgenerering | <100 particles/m² when qualified; degrades 30–100 cycles depending on material; requires periodic re-testing | <50 particles/m² (no degradation concern; single-use eliminates aging variable) |
| Cross-contamination risk | Low to moderate; validated laundering removes residues to <HPLC detection limits; requires segregation by room/product | Zero; single-use eliminates batch-to-batch carryover |
| SOP complexity | High; requires autoclave IQ/OQ/PQ, cycle tracking, laundering validation, service life monitoring, retirement protocols | Low; receiving inspection and disposal only |
| Beholdning | Moderate; 3–5× daily usage quantity to support laundering/sterilization rotation (e.g., 30 mops for facility using 10/day) | Høj; 1-2 ugers forsyning plus sikkerhedslager til leverandørens leveringstider (f.eks. 100-200 mopper til anlæg, der bruger 10/dag) |
| Infrastruktur | Kræver valideret autoklave, tøjvask i farmaceutisk kvalitet (eller outsourcet service) og kontrolleret opbevaring | Ingen steriliseringsinfrastruktur; kræver kun kontrolleret opbevaring, der opfylder kravene til pakkens integritet/udløb |
| Bedste applikationer | ISO 6-8 produktion af store områder (>500 m² daglig mopping), faciliteter med eksisterende autoklavekapacitet, omkostningsfølsomme operationer | ISO 5 Grade A/B aseptiske kerner, multi-produkt/høj-potens API-faciliteter, der kræver nul krydskontamineringsrisiko, faciliteter uden autoklavekapacitet |
| Miljøpåvirkning | Lavere affaldsmængde; genanvendelige systemer genererer kun affald ved endt levetid | Higher waste volume; each use generates disposal of mop head + packaging (some vendors offer recycling programs) |
| Audit documentation | Extensive; autoclave validation records, BI monitoring logs, cycle tracking, service life studies, retirement records | Moderate; vendor sterility certificates, receiving inspection records, storage compliance, expiration tracking |
Total Cost of Ownership (TCO) Analyse
Scenario: 1,000 m² Grade C manufacturing area mopped 5× per week (260 uses/year)
Autoclavable reusable system (5-year TCO):
- System cost: \$300 × 3 sets (rotation inventory) = \$900
- Mop head replacement: \$100 × 8 replacements (80-cycle life, 260 uses/year = 3.25 mop head lifecycles/year) × 5 years = \$4,000
- Laundering: \$1.50/cycle × 260/year × 5 years = \$1,950
- Autoclave: \$0.75/cycle (utilities, BI, operator time) × 260/year × 5 years = \$975
- Validation: \$8,000 (IQ/OQ/PQ, laundering validation, service life studies, one-time)
- Total 5-year cost: \$15,825 (\$3,165/year, \$12.17 per mopping event)
Gamma-sterilized disposable system (5-year TCO):
- Mop cost: \$22/unit × 260 uses/year × 5 years = \$28,600
- Disposal: \$0.50/unit × 260/year × 5 years = \$650
- Validation: \$1,000 (receiving inspection protocols, storage procedures, one-time)
- Total 5-year cost: \$30,250 (\$6,050/year, \$23.27 per mopping event)
Reusable system saves \$14,425 over 5 years (48% TCO reduction) for this scenario.
Scenario: 200 m² Grade A/B aseptic filling suite mopped 3× per week (156 uses/year)
Autoclavable reusable:
- System cost: \$350 × 3 = \$1,050
- Mop head replacement: \$120 × 5 replacements × 5 years = \$3,000
- Laundering: \$2/cycle (higher grade for aseptic tool laundering) × 156/year × 5 years = \$1,560
- Autoclave: \$1/cycle (dedicated aseptic tool autoclave) × 156/year × 5 years = \$780
- Validation: \$12,000 (higher validation burden for Grade A/B qualification)
- Investigation risk: 2 EM failures/5 years (reduced from 3 with non-validated tools) × \$8,000 avg cost = \$16,000
- Total 5-year cost: \$34,390 (\$6,878/year, \$44.10 per mopping event)
Gamma-sterilized disposable:
- Mop cost: \$28/unit × 156 uses/year × 5 years = \$21,840
- Disposal: \$0.50/unit × 156/year × 5 years = \$390
- Validation: \$2,000 (receiving/storage protocols)
- Investigation risk: 0.5 EM failures/5 years (lowest risk option) × \$8,000 = \$4,000
- Total 5-year cost: \$28,230 (\$5,646/year, \$36.17 per mopping event)
Disposable system saves \$6,160 (18% TCO reduction) for this high-risk, lower-volume application.
TCO decision framework: Reusable systems achieve ROI advantage at higher usage volumes (>200–300 uses/year) and lower risk classifications (Grade C/D). Disposable systems optimize for high-risk low-volume applications (Grade A/B <200 uses/year) where investigation cost avoidance outweighs higher per-use cost.
Risiko for kontaminering på tværs af rum
Reusable systems require validated cleaning between uses to prevent room-to-room contamination:
Laundering validation: Pharmaceutical textile reprocessing follows PDA TR 70 guidance. Validation demonstrates that laundering removes soil, API residues, and bioburden to below detection limits. Critical parameters: water quality (WFI or purified water for final rinse), detergent type/concentration (pharmaceutical-grade, residue-tested), wash temperature and cycle time, rinse cycles (minimum 3–5 with final rinse <10 CFU/100 mL, <0.25 EU/mL endotoxin), and drying method (HEPA-filtered air, validated temperature/time preventing microbial growth).
Segregation protocols: Color-coding (blue for Grade A/B, green for Grade C, yellow for Grade D, red for waste areas) prevents accidental cross-use. Physical segregation (separate storage for each grade/product line) reinforces procedural controls. Labeling (room assignment, service date, cycle count) enables traceability.
Worst-case residue testing: Intentionally contaminate mops with worst-case challenge (high-potency API powder, concentrated cleaning agent, high bioburden organism cocktail), launder per validated protocol, test for residues via HPLC (API) or TOC (cleaning agents) and bioburden via surface swab. Acceptance: API <1% of lowest therapeutic dose (product-specific calculation), cleaning agents <LOD, bioburden <10 CFU/mop.
Engangssystemer eliminerer kompleksitet i validering af hvidvask og giver absolut sikkerhed for nul overførsel - den regulatoriske præference for multi-produktfaciliteter, højpotente API'er og beta-lactam fremstilling.
SOP Integration og Workflow Design
Autoklaverbar genanvendelig arbejdsgang:
- Brug moppe i det udpegede område pr. valideret moppe-SOP
- Efter brug opsamles i dertil beregnet beholder til forurenet værktøj
- Overførsel til vaskerum (dagligt eller pr. batch-protokol)
- Vask pr. valideret cyklus (dokumentbatch, dato, operatør)
- Undersøg for skader (revner, nedbrydning af kanten, misfarvning); gå på pension, hvis der er mangler
- Indlæs i autoklaven i kvalificeret konfiguration, placer BI'er pr. PQ-protokol (hvis rutinemæssig BI-kørsel er planlagt)
- Udfør valideret autoklavecyklus; optag parametriske data (tid, temp, tryk, F₀)
- Allow controlled cool-down; transfer to sterile storage area maintaining packaging/hold time per validation
- Issue to production with cycle count documentation (track toward service life limit)
- Repeat steps 1–9 until mop reaches qualified cycle limit, then retire per validated destruction procedure
Disposable workflow:
- Receive gamma-sterilized mops in sealed vendor packaging
- Inspect packaging integrity (no tears, seals intact) and verify documentation (sterility certificate, expiration date, lot traceability)
- Store in controlled conditions (temperature, humidity, segregated by lot) per validation
- Issue to production maintaining packaging integrity until point of use
- Open in designated gowning/airlock area per aseptic technique
- Use once per validated mopping SOP
- Dispose per pharmaceutical waste protocols (segregated by grade/product if needed for investigation traceability)
Disposable workflows eliminate steps 3–8 from reusable protocols, reducing operator training burden and procedural deviation risk. However, reusable workflows offer greater operational flexibility (no vendor dependency for emergency restocking, overnight turnaround vs 2–4 week lead times).
Ydeevne for miljøovervågning
EM programs track cleaning tool performance through two metrics:
Particle count trends: Optical particle counters positioned during mopping operations (or post-mopping recovery testing) detect degrading mops. Baseline: <50 particles/m² for new autoclavable mops, <30 partikler/m² til engangsartikler. Trending: den månedlige gennemsnitlige partikeldannelse under mopping bør forblive flad. Opadgående tendenser (f.eks. 50 → 70 → 95 partikler/m² over 3 måneder) indikerer moppenedbrydning, der kræver tidligere pensionering eller undersøgelse af autoklave-/vaskeprocesdrift.
EM-fejlundersøgelser bør skelne værktøjsfejl fra operatørteknik eller problemer med desinfektionsmiddel. Hvis flere rum viser en stigning i partikel-/biobelastningen samtidigt, skal du have mistanke om værktøjsnedbrydning (mopper nærmer sig udtjent levetid). Hvis det er isoleret til ét rum eller operatør, skal du have mistanke om teknik eller desinfektionsmiddel.
Verifikation af reduktion af biobelastning: Pre-mopping and post-mopping surface swabs or contact plates measure cleaning efficacy. Acceptance: ≥2-log reduction (99% removal) for routine cleaning, ≥3-log reduction (99.9% removal) for sporicidal cleaning. Autoclavable mops at SAL 10⁻⁶ and disposable sterile mops both achieve this when used with validated disinfectants and techniques. Non-sterile or inadequately sterilized mops often show <1-log reduction or even bioburden increases (contaminated tool redistributing organisms).

Figure 3: Pharmaceutical Grade B/C cleanroom showing gowned personnel in proper protective equipment and manufacturing environment. EU GMP Annex 1 Section 4.29 requires sterile cleaning materials (including mops) in Grade A/B areas, achieved through validated autoclave cycles delivering SAL 10⁻⁶.
MIDPOSI autoklaverbar moppesystemanbefaling (ISO 5–8)
MIDPOSI autoclavable cleanroom mop systems are engineered for pharmaceutical contamination control, combining validated material performance with turnkey documentation packages that reduce facility qualification timelines from months to weeks.
Autoklave-validerede polyester moppepuder
MIDPOSI sealed-edge polyester mop pads use continuous-filament PET polyester in a tubular-knit construction with heat-sealed perimeters. No exposed fiber ends; no cut edges; no particle shedding pathways.
Performance specifications:
- Particle generation: <80 particles ≥0.5 µm/m² (new), <100 particles/m² maintained through 80 autoclave cycles at 121°C/30 min (per ISO 14644-14 testing)
- Autoclave durability: Qualified for 80–100 cycles with visual, dimensional, and mechanical integrity acceptance criteria documented
- Chemical compatibility: Validated for pharmaceutical disinfectant rotation (70% IPA daily, 5% H₂O₂ weekly, 1000 ppm quat 2× weekly, 500 ppm sodium hypochlorite weekly) with no particle generation increase or mechanical strength loss >15% after 80 combined cycles
- Absorbency: 5× dry weight (460 mL per 40 cm mop head, 780 mL per 60 cm head)
- Available sizes: 30 cm, 40 cm, 60 cm width options for flat mops; 350 mm and 450 mm for tubular mop heads
Validation package: Particle generation test report (ISO 14644-14 protocol, tested at 0, 20, 40, 60, 80 cycles), autoclave aging study (dimensional stability, tensile strength retention, visual integrity across 100 cycles), chemical compatibility matrix (pass/fail for IPA, peroxide, quats, bleach), material certificates of analysis (fiber type, dye compliance, heavy metals), lot traceability (batch records linking raw material source to finished product).
Rammer i rustfrit stål
MIDPOSI mop frames use SS316 stainless steel in continuous-bend or TIG-welded one-piece construction. Mop head attachment via sealed polyester pockets (no Velcro, no exposed fasteners).
Performance specifications:
- Material: SS316 (18% Cr, 14% Ni, 2.5% Mo) with electropolished finish (<0.4 µm Ra surface roughness)
- Autoclave durability: >200 cycles at 121°C without functional degradation; passivation service available if discoloration appears after 150+ cycles
- Construction: Continuous-bend design (30 cm, 40 cm, 60 cm widths) or welded tube frame (for adjustable/articulating heads)
- Handle attachment: Threaded stainless steel connection with sealed gasket (autoclavable silicone, rated 200+ cycles)
Validation package: Material certificates (mill certs confirming SS316 composition), weld inspection reports (dye penetrant testing for weld integrity), autoclave qualification (200-cycle test with visual and mechanical inspection at 50, 100, 150, 200 cycle intervals), particle generation testing (frame + mop head assembly <100 particles/m² system performance).
Håndtag af polypropylen/aluminium
MIDPOSI offers both high-temperature polypropylene (cost-optimized, 80-cycle life) and SS316 stainless steel (premium durability, 200+ cycle life) handles.
Autoclavable PP handles:
- Material: High-temperature polypropylene copolymer with heat stabilizers, melting point 165°C
- Autoclave durability: 80 cycles at 121°C (tested to 100 cycles with retirement at 80 to maintain safety margin)
- Construction: One-piece injection-molded tube, sealed threaded frame connection with autoclavable gasket
- Lengths: 120 cm, 140 cm, 160 cm fixed-length; 90–180 cm telescoping (sealed joint design)
- Cost: \$38–\$68 depending on length
SS316 handles:
- Material: SS316 stainless steel tube, electropolished
- Autoclave durability: >200 cycles
- Construction: One-piece welded tube or seamless extrusion, sealed frame connection
- Lengths: 120 cm, 150 cm fixed-length; 100–200 cm telescoping
- Cost: \$95–\$145 depending on length
Selection guidance: Match handle cycle life to mop head and frame life. If using 80-cycle polyester mops with 200-cycle SS316 frames, choose PP handles (80 cycles) for synchronized retirement—or choose SS316 handles (200 cycles) that outlast 2–3 mop head lifecycles, reducing long-term cost.
Triple-Bucket System til desinfektionsmiddelintegritet
MIDPOSI triple-bucket cart systems integrate with autoclavable mop heads/frames/handles to deliver complete workflow validation.
Configuration: Three 12-liter stainless steel buckets on wheeled cart frame, graduated volume markings (500 mL increments), integrated press-type wringer positioned over waste bucket. Color-coded lids (blue = disinfectant, green = rinse, red = waste) prevent operator confusion.
Materiale: SS316 stainless steel buckets and frame, autoclavable casters (high-temperature nylon or stainless), welded construction (no particle-generating joints).
Autoclave compatibility: Entire cart (buckets, frame, wringer) autoclavable at 121°C as assembled unit. Qualification supports Grade B/C use with sterilized mop systems.
Validation package: Fluid segregation validation (worst-case testing: mop 500 m² with single bucket fill, measure disinfectant concentration every 100 m², verify remains within 90–110% of target through completion), disinfectant contact time verification (measure residual wetness duration on floor surface), bucket/wringer bioburden testing (swab test post-autoclave confirming <1 CFU per 25 cm² surface area), operator training SOP with photographic workflow documentation.

Kvalifikationsrapporter tilgængelige
MIDPOSI provides turnkey validation documentation reducing facility IQ/OQ/PQ burden:
Particle generation report: ISO 14644-14 protokoltestning; moppe + stel + håndtagssystem testet som brugt; optiske partikeltællerspecifikationer og kalibreringsregistreringer; testmiljø (ISO klasse 5 kammer, kontrolleret temperatur/fugtighed); resultattabel, der viser partikelantal ved 0, 20, 40, 60, 80 cyklusintervaller med acceptkriterier og bestået/ikke-bestået bestemmelse.
Autoklave kvalifikationsrapport: Aging study design (cycle count intervals, test parameters); visual inspection results (photographs showing mop condition at each interval); dimensional stability measurements (length, width, thickness at three locations per mop); mechanical strength testing (tensile test results, abrasion resistance, thread pull force for frames); functional performance (absorbency, wring-out efficiency); retirement criteria and service life determination rationale.
Chemical compatibility report: Test matrix (all facility disinfectants vs all system components); exposure protocol (concentration, contact time, number of cycles); evaluation criteria (visual degradation, particle generation re-test, mechanical strength retention); pass/fail results per material-chemical combination; recommendations for facility-specific disinfectant rotation.
Disinfectant compatibility testing: Sequential exposure simulation (autoclave → IPA → autoclave → peroxide → autoclave → quat → autoclave through 50 cycles); particle generation trending (verify <100/m² maintained); visual/mechanical assessment; acceptance statement.
Material certificates: Lot-specific certificates of analysis (fiber type, dye batch, metal composition); certificates of conformance (product meets specification); traceability documentation (batch records linking CoA to finished product lot numbers).
IQ/OQ/PQ skabeloner: Pre-written protocols customizable to facility-specific details (autoclave model, cycle parameters, room classifications); includes acceptance criteria, data recording forms, deviation/investigation procedures; reduces validation authoring time from 40–60 hours to 8–12 hours of customization.
Hurtig leveringstid, teknisk support
MIDPOSI supply chain and technical support:
Lead times: Stock items (standard polyester mop heads 40 cm and 60 cm, SS316 frames, PP handles 120 cm and 150 cm) ship within 5 business days. Custom configurations (special sizes, logo printing, color-coded options) ship within 15 business days. Validation documentation packages deliver within 3 business days of order (electronic PDF delivery).
Engineering support: Pre-sale technical consultation (30–60 min call with QA/validation teams to review facility requirements, recommend configurations, discuss validation approach). Sample evaluation programs (facilities can request 5–10 sample mops for in-house pilot testing before capital commitment). Validation protocol review (MIDPOSI technical team reviews facility IQ/OQ/PQ drafts and provides feedback on acceptance criteria, test methods, data recording). Post-sale troubleshooting (investigation support if mops show unexpected degradation or EM impacts).
Training: On-site or virtual operator training covering proper mopping technique, autoclave loading configuration, cycle count tracking, visual inspection for retirement criteria, and corrective actions for damaged tools.
Conversion CTA:
Request Validation Report — Download complete ISO 14644-14 particle generation test report, autoclave aging study, and chemical compatibility matrix for MIDPOSI polyester mop systems. Includes IQ/OQ/PQ protocol templates. [Request button]
Request OEM Quotation (12h Response) — Submit facility requirements (ISO class, floor area, autoclave specifications, disinfectant program) for custom system quotation. Pricing includes volume discounts for multi-site procurement. Engineering consultation included. [Request button]
Download Technical Data Sheet (PDF) — Printable specification sheet covering material composition, performance specifications, autoclave durability, chemical compatibility, available sizes, and ordering information. [Download button]
FAQ — Autoklaverbare renrumsmopper
Hvad er forskellen mellem "autoklaverbar" og "steril"?
“Autoclavable” describes a material property—the ability to withstand moist-heat sterilization at 121–134°C without unacceptable degradation. “Sterile” describes a microbiological state—validated absence of viable organisms to Sterility Assurance Level (SAL) 10⁻⁶. An autoclavable mop can be made sterile through validated autoclave cycles, but merely being autoclavable does not mean it is currently sterile. Sterility is achieved through a validated process (autoclaving, gamma irradiation) and maintained through controlled storage/packaging until point of use. Procurement specifications must address both: materials must be autoclavable (withstand repeated cycles without performance loss) og steriliseringsprocessen skal valideres (vis SAL 10⁻⁶ gennem biologiske indikatorer og parametrisk overvågning).
Hvor mange autoklavecyklusser kan en polyestermoppe modstå?
Forseglede polyestermopper af farmaceutisk kvalitet tåler 50-100 autoklavecyklusser ved 121°C/30 minutter, før de når ydeevnegrænserne. Premium kontinuerlig filament polyester opnår 80-100 cyklusser; standard polyester af farmaceutisk kvalitet opnår 50-80 cyklusser. Den begrænsende faktor er ikke akut materialefejl (smeltning, rivning), men gradvis stigning i partikelgenerering - mopper genererer <80 partikler/m² når nye typisk når 90-100 partikler/m² med 80 cyklusser, nærmer sig ISO Klasse 5-7 acceptgrænsen på <100 particles/m². Facilities should retire mops at 70–80% of vendor-validated cycle count (e.g., if vendor qualifies to 100 cycles, retire at 70–80 cycles) to maintain safety margin and prevent mid-service-life classification failures. Microfiber blends achieve only 30–50 cycles due to polyamide thermal degradation. Higher-temperature cycles (134°C) reduce service life 30–50% for all materials.
Kan mikrofibermopper autoklaveres?
Ja, men med betydelige begrænsninger. Mikrofiber (polyester/polyamidblandinger, typisk 80/20 eller 70/30) kan overleve autoklavering, men polyamid nedbrydes hurtigere end 100 % polyester. Autoklaverbar mikrofiber af farmaceutisk kvalitet opnår 30-50 cyklusser ved 121°C, før partikeldannelsen overskrider ISO-grænserne (stiger fra ~100 partikler/m² nye til 200-400 partikler/m² med 40-50 cyklusser). Mikrofiber viser også fiberfusion (filamenter, der smelter sammen under SEM), dimensionskrympning (5-10% gange 30 cyklusser) og restretention (beskadigede fibre bliver klæbrige). Til Grade A/B-applikationer er 100 % polyester med forseglet kant overlegen (længere levetid, mere stabil partikeldannelse, bedre dimensionsstabilitet). Mikrofiber forbliver acceptabelt for ISO-klasse 7–8 (kvalitet C/D), hvor forbedret absorberingsevne (6–8× tørvægt vs. 4–6× for polyester) fordele opvejer reduceret holdbarhed og partikeldannelse <200/m² meets classification. Facilities using microfiber should: (1) qualify to 30–40 cycles maximum, (2) trend particle generation every 10 cycles, (3) avoid 134°C cycles (severe polyamide degradation), (4) restrict use to Grade C/D areas.
Har alle autoklaverbare moppekomponenter brug for samme levetid?
No, but mismatched cycle life creates waste or operational complexity. Optimal strategy: match component service life to enable synchronized retirement.
Matched service life example: 80-cycle polyester mop heads + 80-cycle polypropylene frames + 80-cycle PP handles. All components retire together after 80 cycles—no waste from discarding functional long-life components, no risk from using short-life components beyond qualification.
Mismatched service life example: 80-cycle polyester mop heads + 200-cycle SS316 frames + 200-cycle SS316 handles. Frames and handles outlast 2–3 mop head lifecycles. Economically optimal (lower long-term cost), but requires inventory tracking (pairing new mop heads with used frames/handles of known cycle history) and periodic frame/handle inspection (verify no degradation after 80, 160 cycles before re-pairing).
Worst mismatch scenario: 80-cycle mop heads + 50-cycle frames + 80-cycle handles. Frames fail mid-service-life, forcing retirement of functional mop heads and handles—wasting capital and requiring emergency frame procurement.
Procurement guidance: For operational simplicity, match all component service life (accept higher upfront cost for long-life components or accept shorter system life with lower-cost components). For cost optimization, pair long-life frames/handles (SS316, 200+ cycles) with shorter-life mop heads (polyester, 80 cycles), accepting inventory complexity for multi-decade frame/handle utilization.
Er gamma-steriliserede mopper bedre end autoklaverbare til klasse A?
Not necessarily “better,” but often preferred for specific risk/operational profiles:
Gamma-sterilized disposable advantages for Grade A:
- Zero cross-contamination risk (single-use eliminates batch-to-batch carryover)
- No service life degradation (every mop is “new” performance)
- Simpler validation (vendor supplies sterility certificates; facility only validates receiving/storage)
- Lower investigation burden (if EM failure occurs, single-use mops are eliminated as chronic contamination source)
Autoclavable reusable advantages for Grade A:
- Lower cost per use (\$3–\$7 vs \$20–\$30 for disposables)
- Operational independence (overnight turnaround vs 2–4 week vendor lead times)
- Environmental benefit (lower waste volume)
Decision framework: If Grade A area is small (<200 m²) and mopping frequency is low (2–3× per week), disposables justify premium cost through investigation risk reduction—a single EM failure investigation (\$8,000–\$15,000 cost) negates years of per-use savings. If Grade A area is large (>500 m²) or usage is high (daily mopping), reusable systems deliver strong ROI—but require robust autoclave validation, service life tracking, and laundering protocols. Multi-product or high-potency API facilities should default to disposables due to cross-contamination risk intolerance regardless of area size. Facilities with validated autoclave infrastructure and GMP laundry capabilities can safely use autoclavable systems in Grade A, accepting higher validation burden for long-term cost savings.
Hvordan validerer jeg, at autoklavering rent faktisk steriliserer mine mopper?
Sterilization validation follows a three-tier IQ/OQ/PQ framework:
IQ (installationskvalifikation): Verify autoclave meets specifications (chamber size, temperature range, pressure capability), confirm calibration of temperature probes and pressure gauges (within ±1°C and ±0.5 psi), document biological indicator lot qualification (species confirmation, population count, D-value and z-value certificates), verify steam quality (condensate conductivity <5 µS/cm, non-condensable gas content <3.5%).
OQ (operationel kvalifikation): Udfør undersøgelser af varmefordeling i tomme kammer (termoelementer på 9-12 steder i hele kammeret, kortlæg temperaturensartethed, identificer kolde pletter - typisk geometrisk centrum og nær dør). Kør varmegennemtrængningsundersøgelser i belastet kammer med moppebelastninger i worst-case konfiguration (maksimalt moppeantal, tætteste pakning, autoklaveposer, hvis brugt). Placer termoelementer på formodede kolde steder (midten af tætstablede mopper, midten af poserne). Bekræft minimumstemperatur ≥121°C og minimum eksponeringstid ≥15 minutter opnået på alle steder. Beregn F₀ (kumulativ dødelighed) ud fra tid-temperaturdata; accept ≥8 min for dampcyklusser.
PQ (Performance Qualification): Kør 3 på hinanden følgende vellykkede cyklusser med værst tænkelige belastninger. Placer biologiske indikatorer (Geobacillus stearothermophilus sporestrimler eller selvstændige BI'er, population ≥10⁶ CFU, D₁₂₁ ~1,5 min) på kolde steder identificeret i OQ plus center og periferi af moppebelastninger. Inkluder positiv kontrol BI (usteriliseret, inkuberet for at bekræfte levedygtighed). Efter cyklus overføres BI'er aseptisk til vækstmedium (tryptisk sojabouillon), inkuber ved 55-60°C i 7 dage. Accept: alle proces-BI'er viser ingen vækst (indikerer ≥6-log reduktion); positiv kontrol viser vækst (bekræfter, at BI'er var levedygtige). Dokumenter parametriske data (tids-/temperatur-/trykdiagrammer), der viser validerede parametre med cyklus opfyldt.
Rutinemæssig overvågning: Ongoing sterilization assurance via: parametric release (every cycle’s time/temp/pressure data confirms validated parameters met), periodic BI runs (frequency per facility protocol—weekly, monthly, or per-batch), autoclave maintenance (annual preventive maintenance, steam quality testing quarterly, chamber leak testing semi-annually).
Hvilken dokumentation har jeg brug for til FDA/EMA-audits?
Regulatory inspectors examining autoclavable cleanroom mop validation expect:
1. Equipment qualification records: Autoclave IQ/OQ/PQ protocols with executed data, biological indicator lot certificates (species, D-value, z-value, expiration), autoclave maintenance records (PM schedules, calibration certificates for temperature/pressure instrumentation), steam quality test results.
2. Material qualification records: Leverandørvalideringsrapporter (partikelgenerering i henhold til ISO 14644-14, autoklaveholdbarhedsundersøgelser, kemiske kompatibilitetsmatricer), materialecertifikater for analyse og overensstemmelse, undersøgelser af bestemmelse af levetiden (dimensionel stabilitet, fastholdelse af mekanisk styrke, visuel integritet på tværs af kvalificeret cyklustælling).
3. Procesvalideringsposter: Validering af hvidvask (demonstrerer restfjernelse til under detektionsgrænser), autoklavecykluskvalificering (varmefordelings- og penetrationsundersøgelser for moppebelastninger), værst tænkelige lastdefinition (maksimalt moppeantal, emballagekonfiguration), acceptkriterier for parametriske og BI-data.
4. Rutinemæssig overvågningsregistrering: Batch-registreringer for hver steriliseringscyklus (dato, operatør, cyklusparametre, belastningsbeskrivelse, BI-resultater, hvis det er relevant), cyklustællings-sporingslogfiler (linker moppe-serienumre eller partikoder til kumulative cyklusser, tilbagetrækning ved kvalificeret grænse), visuel inspektionsregistrering (periodisk inspektion for revner, kantnedbrydning, trend-EM-farvning og misfarvning af biopartikler), moppeoperationer).
5. Standarddriftsprocedurer: Autoklavedrift SOP (påfyldning, cyklusvalg, aflæsning, holdetider), SOP for hvidvask (vaskeparametre, vandkvalitetskrav, vaskemiddelspecifikationer), moppebrug og opbevarings-SOP (rumtildelinger, sterilhåndteringsteknik, opbevaringsforhold/holdetider), SOP for tilbagetrækning og bortskaffelse (grænser for cyklustælling, kriterier for visuelle defekter, bortskaffelsesprocedurer).
6. Forureningskontrolstrategi (CCS): Risikovurdering, der begrunder valg af autoklaverbar vs engangsbrug pr. facilitetszone, erklæringer om udstyrs egnethed (hvorfor valgte moppematerialer/steriliseringsmetode opfylder kravene i bilag 1), træningsrekord (operatørkvalifikation om moppeteknik, autoklavedrift, cykelsporing, inspektionsprocedurer).
7. Efterforskningsprotokoller (hvis relevant): CAPA-dokumentation for eventuelle EM-fejl sporet til rengøringsværktøjer, OOS-undersøgelser, når partikeldannelsen overskred grænserne, steriliseringsfejl (BI positive resultater, parametriske afvigelser).
Revisorer kan spørge: "Hvordan ved du, at dine mopper forbliver egnede gennem deres levetid?" (forvent trenddata, der viser stabil partikelgenerering på tværs af cyklusser). "Hvad sker der, hvis denne moppe ved et uheld bliver brugt ud over dens kvalificerede cyklusantal?" (forvent SOP, der beskriver afvigelsesrapportering, risikovurdering, potentiel produktpåvirkningsevaluering). "Hvordan forhindrer du krydskontaminering mellem produkter/rum?" (forvent valideringsdata for hvidvask, adskillelsesprotokoller, worst-case testresultater af rester).









