MIDPOSI disposable cleanroom mops are designed for pharmaceutical cleanrooms, ISO-controlled environments, laboratories, medical device production and contamination-sensitive manufacturing areas where facilities require low-lint cleaning tools, simplified mop replacement workflows and stronger contamination-control consistency.

A disposable cleanroom mop is a single-use or limited-use cleanroom mop cover designed for GMP and ISO-controlled environments where facilities need low-lint cleaning tools, simplified replacement workflows and reduced reuse-related contamination concerns.
Disposable cleanroom mops are commonly used in pharmaceutical manufacturing, medical device production, laboratory cleanrooms, biotechnology facilities and semiconductor support environments.
Disposable cleanroom mops are single-use or limited-use mop covers designed for controlled environments where repeated mop washing, storage, inspection and reuse may create additional contamination-control concerns.
Unlike general janitorial mop heads, disposable cleanroom mop covers should be selected for low-lint performance, material compatibility, surface contact, packaging format, frame compatibility and suitability for cleanroom cleaning SOPs.
They are commonly used with compatible cleanroom flat mop systems, cleanroom mop handles, and defined cleaning procedures for floors, walls, ceilings and controlled surfaces.
Disposable mop covers are often selected when the facility wants a cleaner, simpler and more controlled alternative to repeated mop washing, storage, inspection and reuse.
Single-use mop covers help reduce contamination concerns associated with previous use, incomplete washing, uncontrolled storage or repeated handling.
Disposable workflows can make it easier to define one-area-one-mop-cover procedures and reduce uncertainty during routine cleanroom cleaning.
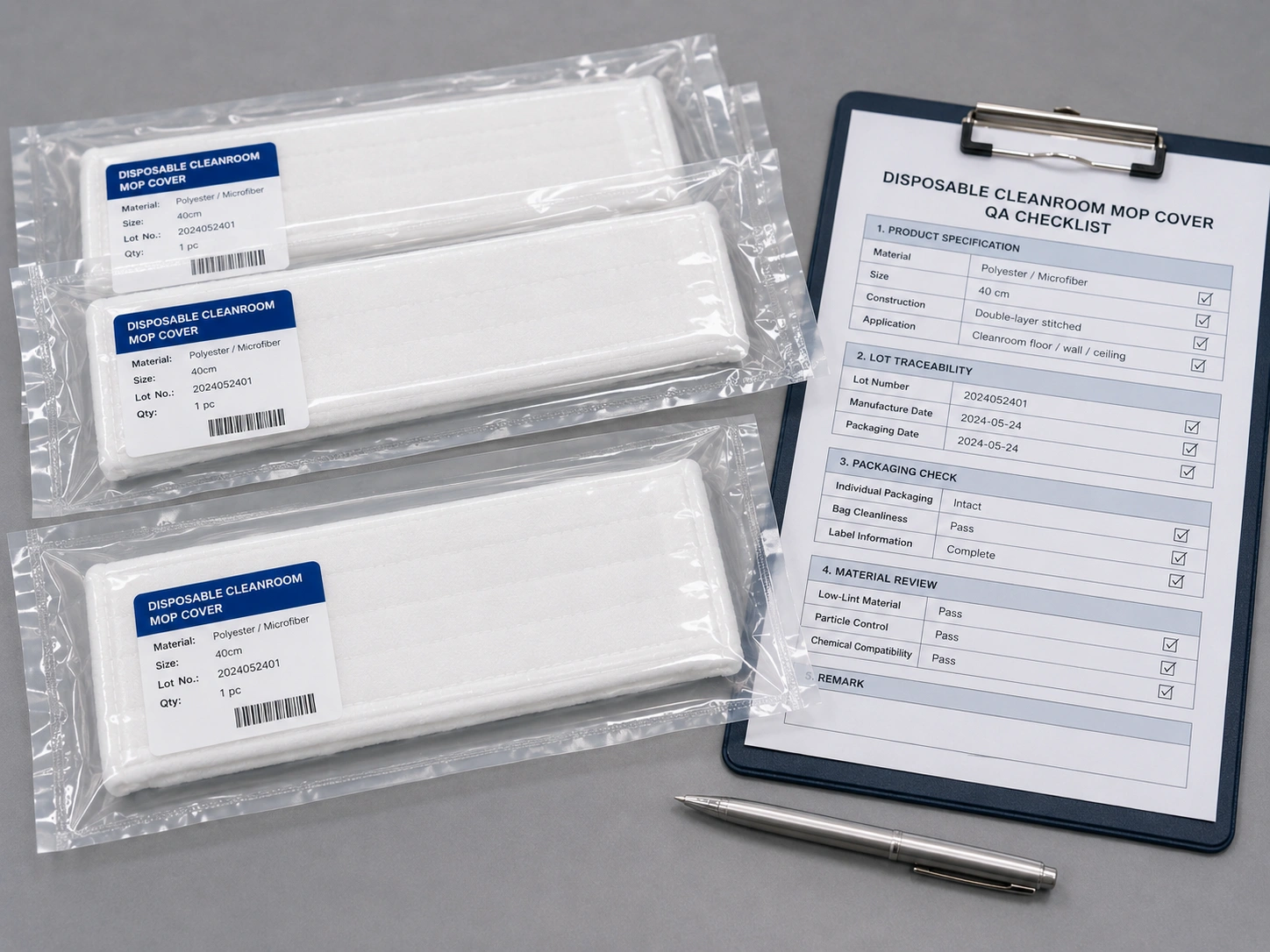
Clean packaging, lot traceability and consistent product specifications can help procurement and QA teams review cleanroom consumables more efficiently.
A disposable mop cover works best when it is matched with the correct flat mop frame, cleanroom handle, bucket or wringer workflow, disinfectant SOP and area zoning procedure.

Disposable low-lint mop cover for cleanroom floor, wall, ceiling and controlled surface cleaning.
Compatible frame that supports stable surface contact and efficient cleaning coverage.
Smooth handle suitable for controlled-environment cleaning and repeatable operator movement.
Defined cleaning sequence for zone control, disinfectant use and mop cover replacement.
Disposable cleanroom mops are suitable for controlled-environment applications where reuse control, zone separation, packaging traceability and cleaning documentation are important.
Supports controlled cleaning workflows in GMP facilities, formulation areas, filling support areas and QA-managed environments.
Supports surface cleaning where particles, residues and production-area cleanliness must be controlled.
Suitable for routine cleaning of laboratory cleanrooms, controlled corridors and process-adjacent areas.
Useful where low-lint cleaning tools are required for controlled floors, walls and work-adjacent surfaces.
Supports consistent wet mopping and removal of residues from cleanroom floors.
Flat mop systems with suitable handles can support vertical and overhead cleaning tasks.
The right choice depends on contamination-control goals, cleaning frequency, sterilization capability, waste policy, documentation needs and total operating cost.
| Selection Factor | Renrumsmoppe til engangsbrug | Reusable Cleanroom Mop |
|---|---|---|
| Reuse-related contamination risk | Lower, because the mop cover is replaced after use or a defined use cycle | Depends on washing, sterilization, inspection and storage control |
| SOP simplicity | Easier to define one-area or one-session use | Requires lifecycle tracking and reprocessing procedures |
| Cost structure | Higher consumable cost per use | Lower cost per use when reprocessing is well controlled |
| Validation workload | May simplify some reuse and lifecycle concerns | Requires validation of cleaning, sterilization, storage and reuse limits |
| Best fit | High-control areas, contamination-sensitive workflows and simplified tool control | Routine cleaning areas with strong reprocessing control |
Disposable mop covers can be integrated into controlled cleaning procedures by defining when a cover is attached, where it is used, how it is removed and when a fresh cover is required for the next area.
| Workflow Step | Formål | Cleanroom Control Value |
|---|---|---|
| Prepare fresh mop cover | Use a clean disposable cover before cleaning a defined area | Supports traceable, controlled tool use |
| Attach to compatible frame | Secure the cover to a flat mop frame with stable surface contact | Improves cleaning consistency and coverage |
| Clean assigned area | Use the mop cover according to the facility cleaning sequence | Supports area zoning and SOP control |
| Remove and dispose | Remove the used cover after the defined use cycle | Helps reduce reuse-related contamination concerns |
| Replace for next area | Use a fresh cover for the next zone or cleaning step | Supports cleaner separation between controlled areas |
Use the table below as a practical procurement guide when comparing disposable cleanroom mop options.
| Krav | Recommended Focus | Hvorfor det betyder noget |
|---|---|---|
| GMP facility cleaning | Low-lint disposable mop cover with documentation support | Supports QA review and contamination-control expectations |
| Sterile-area support | Sterile cleanroom mop option if required by internal SOP | Helps reduce reuse-related contamination concerns in higher-control areas |
| Cleanroom floor mopping | Flat mop cover with suitable absorbency and surface contact | Improves cleaning coverage and wetting consistency |
| Rengøring af væg og loft | Disposable mop cover matched with suitable flat frame and handle | Supports vertical and overhead surface cleaning |
| Desinfektionsmiddelkompatibilitet | Confirm compatibility with facility cleaning chemicals | Reduces material degradation and shedding concerns |
| Sporbarhed | Lot information, packaging details and supplier documentation | Supports purchasing, QA and internal approval workflows |
For B2B purchasing, disposable cleanroom mops should not be evaluated by price alone. Procurement and QA teams should also review material, packaging, compatibility, lot traceability and available supplier documentation.

Before placing a bulk order, purchasing teams should confirm the following details with the supplier.
Tell us your cleanroom class, application area, mop frame size, preferred packaging format and whether sterile options are required. MIDPOSI can help recommend a suitable disposable cleanroom mop solution for your workflow.
Continue exploring MIDPOSI cleanroom mop resources for system selection, GMP facility use and B2B procurement guidance.
Learn how mop covers, frames, handles, buckets and workflows work together as a complete cleanroom mop system.
View Cleanroom Mop System →Compare cleanroom mop heads and mop covers for low-lint cleaning, disposable workflows and frame compatibility.
Se guide til moppehoveder →Learn how bucket and wringer workflows support liquid separation and saturation control.
View Bucket Workflow →These answers are written for purchasing managers, QA teams, cleanroom supervisors and contamination-control buyers.
A disposable cleanroom mop is a single-use or limited-use mop cover designed for controlled environments where particles, residues and reuse-related contamination risks must be controlled.
Disposable cleanroom mops are designed for controlled environments where low-lint performance, contamination control and defined replacement workflows are important. Regular janitorial mops are not designed for GMP or ISO cleanroom cleaning requirements.
Disposable cleanroom mops are often better for reducing reuse-related contamination concerns and simplifying SOP control. Reusable mop systems may be more economical when washing, sterilization, storage and lifecycle validation are well managed.
Not always. Some facilities use reusable cleanroom mops with validated reprocessing procedures, while others prefer disposable mop systems to simplify contamination-control workflows and reduce reuse-related risk.
Yes. Disposable cleanroom mops can be used in GMP facilities when the product is suitable for the application and properly qualified according to the facility’s internal cleaning SOP and QA requirements.
No product should be described as eliminating all cross-contamination risk by itself. Disposable mop covers can help reduce reuse-related cross-contamination risk when used correctly in a defined cleanroom cleaning workflow.
No. Disposable means single-use or limited-use; it does not automatically mean sterile. If sterile supply is required, buyers should request sterile packaging and sterility-related documentation from the supplier.
Replacement frequency should be defined by the facility SOP. Common approaches include one cover per area, one cover per cleaning session, or one cover per defined zone to support contamination-control separation.
Buyers should check material, low-lint performance, frame compatibility, packaging format, disinfectant compatibility, sterility requirements, lot traceability, MOQ and available supplier documentation.
MIDPOSI supports pharmaceutical, laboratory, medical device and controlled-environment buyers with disposable cleanroom mop solutions for floor, wall, ceiling and contamination-control cleaning workflows.
MIDPOSI supports B2B buyers with disposable cleanroom mop options for GMP, ISO, pharmaceutical, laboratory, medical device, electronics and controlled-environment cleaning applications.


Vi kontakter dig inden for 1 arbejdsdag, vær venligst opmærksom på e-mailen med suffikset “*@midposi.com”.