In pharmaceutical manufacturing, contamination control failures traced to cleaning tools represent one of the most preventable sources of environmental monitoring excursions and regulatory observations. A 2025 FDA warning letter specifically cited “non-sterile wipes within the ISO 5 aseptic processing area” and “failure to establish an adequate system for cleaning and disinfecting” as CGMP violations that jeopardized batch sterility. The root cause: reusable cleaning tools carrying bioburden and particle contamination from prior use, laundered inadequately, and introduced into aseptic zones without validated reprocessing.
Engangsmopper til renrum eliminerer denne forureningsvej fuldstændigt. Hver moppecyklus begynder med en frisk, valideret moppepude - ingen krydskontamineringsrisiko fra tidligere rum, ingen vasketøjsrester, ingen genbehandlingsvalideringsbyrde. For faciliteter, der opererer under ISO 14644-1 og det reviderede EU GMP Annex 1 (gælder i august 2023), leverer engangsmopper nul krydskontaminering, forudsigelig partikelydelse og dramatisk forenklet rengøringsvalideringsdokumentation. Det regulatoriske landskab og den samlede omkostningsøkonomi favoriserer i stigende grad engangsrengøringsværktøjer i aseptisk fremstilling, fyld-finish-linjer og højrisiko farmaceutiske produktionsområder.
This is not “disposables are always better.” Reusable mop systems remain cost-effective in ISO 8 support areas, large-scale manufacturing with validated in-house laundering, and facilities with robust reprocessing infrastructure. But when contamination control is tight (ISO 5–7 aseptic cores), environmental monitoring failures are frequent, or validation resources are limited, disposable cleanroom mops deliver superior contamination control and lower total cost of ownership despite higher unit price.
Denne vejledning undersøger de regulatoriske drivkræfter bag vedtagelse af engangsmoppe, kvantificerer de vigtigste fordele (nul krydskontaminering, forudsigelig ydeevne, reduceret valideringsbyrde), sammenligner de samlede ejeromkostninger med genanvendelige systemer, kortlægger anvendelsestilfælde efter ISO-klassificering og anbefaler MIDPOSI engangsmoppeprodukter, der matcher faciliteternes krav. Til sidst vil du have en klar beslutningsramme for, hvornår engangsrenrumsmopper retfærdiggør deres omkostninger gennem reduceret kontamineringsrisiko, forenklet overholdelse og undgåede EM-undersøgelsescyklusser.

Why Disposable Mops Are Growing in Pharma
The shift toward disposable cleanroom mops is not a trend driven by vendor marketing—it reflects fundamental changes in regulatory expectations, measurable contamination control failures traced to reusable tools, and the economics of environmental monitoring in modern pharmaceutical manufacturing.
Key Advantages of Disposable Cleanroom Mops (Zero Cross-Contamination)
Disposable cleanroom mops deliver four measurable advantages over reusable systems: guaranteed zero cross-contamination, predictable particle and microbial performance, lower validation and documentation burden, and improved labor efficiency. These benefits compound in high-risk pharmaceutical applications where contamination control margins are tight and regulatory scrutiny is intense.
Guaranteed Zero Cross-Contamination
Cross-contamination—the transfer of contaminants from one location, product, or operation to another—is the fundamental risk that reusable cleaning tools introduce. Every reusable mop pad accumulates bioburden, particles, and chemical residues during use. Laundering reduces these contaminants but rarely achieves sterility or complete residue removal. When that mop re-enters a classified area, it carries contamination from prior environments.
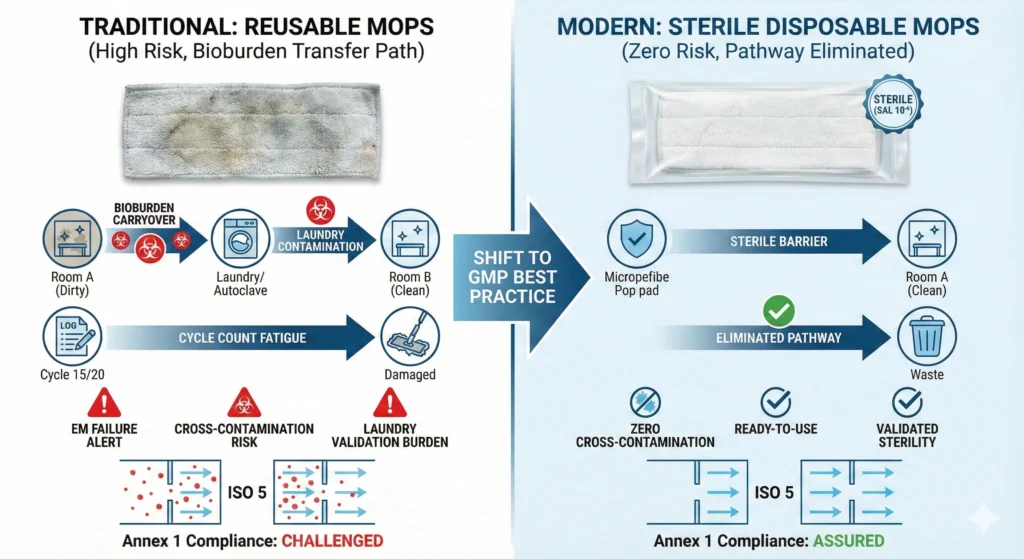
Disposable cleanroom mops eliminate this pathway:
- Fresh mop every cycle: Each mopping operation begins with a new mop pad removed directly from validated sterile packaging (for gamma-irradiated disposables) or from sealed low-lint packaging (for non-sterile disposables). No bioburden carryover from previous rooms, no particle accumulation from prior mopping passes, no laundry detergent residues.
- No room-to-room contamination transfer: Facilities using reusable mops typically maintain separate inventory for different ISO grades (ISO 5 mops never used in ISO 8 areas). Even with perfect segregation protocols, labeling errors and inventory shortages create cross-assignment risk. Disposable systems eliminate this failure mode—mops cannot be reused, so cross-area contamination is impossible.
- No laundry-related contamination: Reusable mops pass through laundry facilities where cross-contamination opportunities abound: mixed-load washing, inadequate rinse cycles, shared equipment handling both cleanroom gowns and general facility linens. Disposable mops bypass the entire laundry system, removing a multi-step contamination pathway.
Validation Simplification Through Elimination of Reprocessing Variables
Cleaning validation protocols for reusable tools must account for worst-case reprocessing scenarios: maximum cycle count (end of qualified life), maximum bioburden challenge, minimum laundering efficacy. Each variable introduces uncertainty into contamination control performance.
Disposable mops present zero reprocessing variables. Material qualification testing (particle generation, chemical compatibility, bioburden baseline) validates the mop in its as-received, never-used state—the same state presented during actual use. This eliminates validation complexity:
- No cycle-count degradation studies: Reusable mop validation requires testing at multiple lifecycle points (new, 25 cycles, 50 cycles, 100 cycles) to demonstrate particle generation remains within acceptance criteria. Disposable validation tests only the single “new” condition.
- No laundering efficacy validation: Facilities using reusable mops must validate laundry cycles, detergent concentrations, bioburden reduction, and residue removal. Disposable systems have no laundering step to validate.
- No reprocessing failure modes: Cross-contamination during laundry, autoclave sterilization failures, tracking errors assigning mops to wrong areas—none of these failure modes exist when tools are single-use.
Predictable Particle & Microbial Performance
Environmental monitoring programs depend on stable, predictable contamination sources to detect true contamination events. When cleaning tools shed variable particle counts and bioburden across their lifecycle, they introduce noise into EM data that obscures real contamination signals.
Stable Particle Generation (<100 Particles/m²)
Sealed-edge disposable polyester mops generate consistent particle counts across all units:
- Typical performance: <50–100 particles ≥0.5 µm per square meter mopped for validated pharmaceutical-grade disposables
- No degradation over time: Every disposable mop performs identically—no edge separation, no fiber breakage, no autoclave-induced dimensional changes. Particle generation on mop #1 matches mop #1,000.
- Simplified EM trending: When particle counts remain stable during cleaning operations, environmental monitoring teams can confidently attribute particle spikes to contamination events (personnel gowning failures, material transfer contamination, equipment malfunctions) rather than cleaning tool degradation.
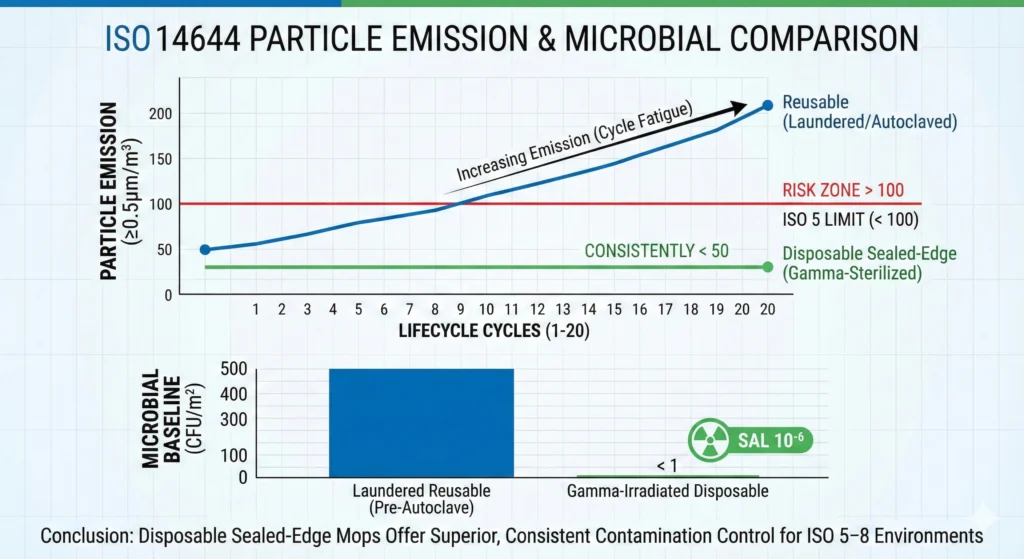
Reusable mops, by contrast, increase particle generation across their qualified life. A polyester sealed-edge reusable mop might start at 50 particles/m² when new, increase to 100 particles/m² by cycle 50, and reach 200–300 particles/m² near end of qualified life (cycle 100–150). This 4× to 6× particle generation increase creates time-dependent contamination contribution that complicates EM data interpretation.
Predictable Bioburden Baseline (Gamma-Sterile Disposables)
Gamma-irradiated disposable mops deliver Sterility Assurance Level (SAL) 10⁻⁶—a one-in-a-million probability of non-sterile units. This terminal sterilization method achieves complete bioburden elimination:
- Zero viable microorganisms: Gamma irradiation (typically 25–50 kGy dose) inactivates all bacterial vegetative cells, spores, fungi, and viruses through DNA damage. Unlike autoclaving (which may leave heat-resistant spores viable if cycle parameters drift) or chemical disinfection (which leaves disinfectant residues and may not achieve sporicidal efficacy), gamma irradiation delivers absolute sterility without residue.
- Validated sterility certificates: Gamma-sterilized disposables ship with Certificates of Sterilization documenting dose delivered, biological indicator (BI) results, and SAL achievement. Facilities can accept these certificates subject to vendor qualification (per Annex 1 Section 4.34), avoiding in-house sterility testing.
- Predictable EM performance: Grade A/B areas using gamma-sterile disposable mops establish stable bioburden baselines. Any microbial detection during post-cleaning environmental monitoring represents contamination from other sources (personnel, air handling, material transfer)—not cleaning tool bioburden carryover.
Non-sterile disposable mops (for ISO Class 7–8 applications) still deliver predictable bioburden performance through validated manufacturing controls. Pharmaceutical-grade disposables maintain bioburden <10 CFU/mop pad through cleanroom manufacturing, sterilized packaging materials, and validated storage. This baseline remains constant across all units, unlike reusable mops where laundry efficacy variability introduces bioburden fluctuations.
Lower Validation & Dokumentationsbyrde
Validation workload is a hidden cost in reusable cleaning tool programs. Facilities must develop protocols, execute studies, analyze data, and maintain documentation for multiple validation elements—each consuming QA specialist time, microbiology lab resources, and equipment qualification hours.

What Reusable Mops Require:
- Laundry equipment qualification (IQ/OQ/PQ): Installation qualification verifying equipment specifications, operational qualification validating cycle parameters (temperature distribution, detergent concentration, rinse efficacy), performance qualification demonstrating bioburden reduction under worst-case loading
- Cleaning tool material qualification: Particle generation testing at new condition and end-of-life (maximum cycle count); chemical compatibility validation across all facility disinfectants; dimensional stability testing through autoclave cycles
- Cycle-count tracking system validation: Validating barcoding or RFID tracking systems, testing retirement trigger alerts, demonstrating traceability throughout mop lifecycle
- Sterility or bioburden verification (if autoclaved): Biological indicator placement studies, sterilization cycle validation, sterility assurance level (SAL) demonstration
- Periodic revalidation: Annual or biennial revalidation after equipment changes, cycle parameter modifications, or detergent formula updates
What Disposable Mops Require:
- Material qualification only: Particle generation testing (typically conducted by vendor with validation report provided), chemical compatibility verification with facility-specific disinfectants
- Receiving inspection: Visual inspection for packaging integrity, verification of sterility certificates (for gamma-sterile units), lot number documentation
- Vendor qualification: Audit of manufacturer’s cleanroom controls, sterilization process validation, quality management system—typically satisfied through review of vendor-provided validation package
Validation Hour Reduction
A representative pharmaceutical facility transitioning from reusable to disposable mops in a 500 m² ISO Class 7 aseptic support area might reduce validation workload by:
- Initial validation: 120–160 hours (reusable laundry validation + tracking system setup) vs 20–30 hours (disposable material qualification + receiving procedure)
- Annual revalidation: 40–60 hours (laundry revalidation + cycle-count system verification) vs 0 hours (no revalidation required; receiving inspection is part of routine operations)
- Investigation support: 8–12 hours per EM excursion (forensic review of mop assignment history, laundry logs, cycle counts) vs 2–3 hours (simple verification of disposable lot number and certificate review)
For QA teams operating with limited headcount—common in biotech startups, contract manufacturing organizations, and facilities running lean operations—this validation burden reduction translates directly to bandwidth for higher-value activities: cleaning validation for new products, environmental monitoring program improvements, deviation investigations.
Labor & Time Efficiency
Operational efficiency compounds across hundreds or thousands of mopping cycles per year. Small procedural improvements—eliminating cycle-count logging, removing mop inspection steps, simplifying disposal—accumulate into measurable labor savings.
No Cycle-Count Tracking
Reusable mop programs require operators to log every mop use:
- Pre-use: Scan mop barcode or RFID tag, verify cycle count within qualified range, document mop assignment to specific room/area
- Post-use: Return mop to soiled laundry staging, scan out of inventory, trigger laundry/autoclave cycle if at predetermined count threshold
- Periodic audits: QA conducts monthly or quarterly audits of cycle-count logs to verify compliance, identify tracking gaps, and enforce retirement of over-aged mops
Disposable systems eliminate this entire workflow. Operators remove mop from packaging, use once, discard into classified waste—no scanning, no logging, no cycle-count verification. For facilities mopping 200–500 m² daily across multiple shifts, this procedural simplification saves 10–15 minutes per shift (aggregate across all mopping operations)—roughly 60–75 hours per year of operator time recovered.
No Laundering, Drying, Re-Distribution
Reusable mop logistics create multi-step workflows:
- Soiled laundry collection: Operators transfer used mops from cleanroom areas to laundry staging, maintaining segregation by ISO grade
- Hvidvaskning: Laundry staff load mops into washers (segregated by grade), execute validated wash cycles, transfer to dryers
- Sterilization (if required): Load dried mops into autoclave, execute validated cycle, verify biological indicators
- Re-distribution: Transport clean/sterile mops back to cleanroom areas, replenish inventory at designated storage locations
Each step consumes labor hours and introduces delay. A typical reusable mop turnaround time is 24–48 hours (longer if autoclaving is required or if laundry is outsourced). Facilities must maintain 2× to 3× inventory to ensure adequate mop availability during laundry cycles.
Disposable mops collapse this logistics chain into two steps:
- Receiving and storage: Accept disposable mop shipments, conduct receiving inspection, store in designated cleanroom inventory areas
- Disposal: Fjern brugte mopper fra renrum i klassificerede affaldsbeholdere efter facilitetens affaldshåndteringsprotokoller
Ingen hvidvaskning, ingen sterilisering, ingen omfordeling. Beholdningskrav falder til 1× (mopper er nødvendige til nuværende drift) plus sikkerhedslager (typisk én ekstra kuffert pr. SKU). For små faciliteter med begrænset vaskeriinfrastruktur eller bioteknologiske startups uden egen vaskerikapacitet, eliminerer engangssystemer behovet for at bygge eller outsource vaskeridrift helt.
Særligt værdifuldt for små-team GMP-faciliteter
Bioteknologiske startups, specialproducenter og kontraktfremstillingsorganisationer, der kører slanke operationelle teams, drager uforholdsmæssig stor fordel af engangsmoppesystemer:
- Intet dedikeret vaskepersonale: Faciliteter med <20 cleanroom personnel often cannot justify full-time laundry operators. Disposable systems eliminate this headcount requirement.
- Simplified training: Operator training for disposable systems requires one SOP (“Remove from package, use once, discard”) versus multiple SOPs for reusable systems (mop inspection, cycle-count logging, laundry segregation, autoclave loading).
- Reduced CAPA workload: Investigations into cycle-count tracking errors, laundry segregation failures, and autoclave sterilization deviations disappear when tools are single-use.
Changes in EU GMP Annex 1 (2023–2025)
The revised EU GMP Annex 1 (effective August 25, 2023) fundamentally repositions contamination control as a site-wide strategic framework. Prior versions treated cleaning and disinfection as procedural requirements; the 2023 revision elevates Contamination Control Strategy (CCS) to a mandatory, documented system that defines critical control points and assesses the effectiveness of design, procedural, technical, and monitoring measures across the facility.
Cleaning Tools as CCS Elements
Annex 1 Section 2.5 explicitly lists “cleaning and disinfection” among the core CCS elements requiring validation, and Section 4.33–4.35 mandates that disinfectants and detergents used in Grade A and Grade B areas “should be sterile prior to use.” The regulation further requires validated disinfection processes, with cleaning preceding disinfection to ensure effective residue removal. These requirements apply not just to chemicals but to the tools that apply them—mop heads, wipes, and cleaning implements must maintain sterility and validated performance within aseptic zones.
The phrase “sterile tools for manipulations” (Section 8.16) extends this logic: if operators manipulate materials in aseptic processing areas using sterile implements, the cleaning tools contacting those same classified surfaces must meet equivalent sterility and contamination control standards.
Hidden Contamination Vectors and Transfer Disinfection
Annex 1 Section 4.11–4.12 identifies materials and equipment transfer as “one of the greatest potential sources of contamination,” requiring validated transfer disinfection and monitored programs. Reusable cleaning tools cross-transfer between rooms and areas constantly—a mop used in a Grade C support corridor one day may be laundered and deployed in a Grade B background the next shift. Without validated reprocessing (laundering, disinfection, sterility verification), the mop becomes a “hidden contamination vector” carrying bioburden, particle residue, and chemical residues between classified environments.
The regulation’s emphasis on smooth, impervious surfaces that minimize shedding and tolerate repeated disinfection (Section 4.5–4.7) applies equally to cleaning tool selection. Disposable mops eliminate the degradation pathway: no cycle-count tracking, no autoclave-induced edge separation, no laundry-induced fiber breakage. Every mop starts fresh, meeting particle generation and sterility specifications without cumulative wear.
Laundry and Reprocessing Risk
Annex 1 Section 7.17 explicitly flags garment laundering as a contamination and cross-contamination risk, requiring qualified laundry processes, segregation from production areas, and defined cycle limits. The same contamination pathways affect reusable mop systems. Laundry facilities handling both gowning and cleaning tools introduce cross-contamination opportunities: inadequate detergent rinse cycles leave surfactant residues, incomplete bioburden reduction transfers microbial loads, and mixed-load processing contaminates Grade A/B tools with particles from lower-grade items.
Facilities without validated in-house laundry operations must outsource—adding supply chain complexity, turnaround delays, and third-party contamination risk. Disposable mops bypass the entire laundry system, collapsing reprocessing logistics into a single step: remove mop pad from sterile packaging, use once, discard into classified waste.
Root Cause: Reusable Tools Driving EM Failures
Environmental monitoring programs at pharmaceutical facilities routinely detect contamination spikes during and immediately after floor cleaning. When investigation teams trace these excursions back to root cause, reusable cleaning tools emerge as repeat offenders.
Residual Bioburden and Cross-Carryover
Reusable mop heads contact floor surfaces laden with microbial contamination, disinfectant residues, and particulate matter. Even after laundering, bioburden and chemical residues persist:
- Incomplete bioburden reduction: Standard laundry cycles (hot water wash, detergent, tumble dry) reduce microbial load but do not achieve sterility. Bacterial vegetative cells survive in fabric interstices, and spore-formers (Bacillus, Geobacillus) tolerate laundry conditions entirely. A reusable mop entering a Grade A/B area without terminal sterilization (autoclaving or gamma irradiation) introduces viable microorganisms directly onto surfaces subsequently contacted by sterile product or product-contact equipment.
- Cross-carryover between areas: Mops used in ISO Class 8 support areas accumulate higher bioburden and particle counts than mops dedicated to ISO Class 5 aseptic cores. If laundry segregation is inadequate—mixed-load washing, shared laundry equipment, improper tracking—Grade A/B mops become contaminated with Grade C/D residues. A single cross-assignment event (using an ISO 8 mop in an ISO 5 room due to labeling error or inventory shortage) can seed bioburden into aseptic environments and trigger multi-week investigation cycles.
- Laundry detergent residues: Surfactants and fabric softeners remaining on mop fibers create sticky surfaces that attract and retain particles. These residues also interfere with disinfectant efficacy—cationic surfactants in fabric softeners neutralize anionic disinfectants (quaternary ammonium compounds), reducing microbial kill rates during mopping and leaving viable organisms on treated surfaces.
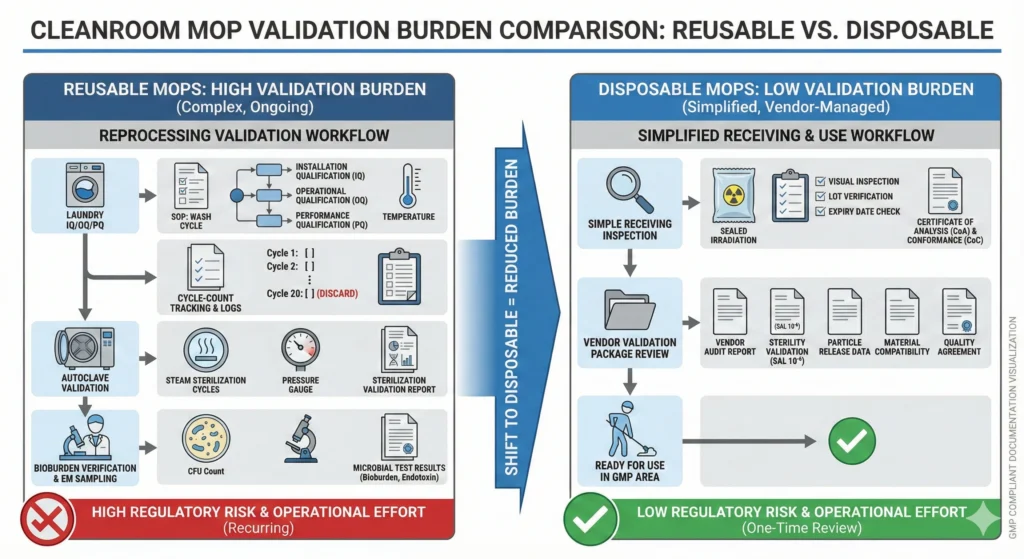
Reprocessing Documentation and Validation Burdens
Reusable cleaning tool programs impose extensive validation and documentation requirements:
- Laundering validation (IQ/OQ/PQ): Faciliteter skal kvalificere vaskeudstyr (vaskemaskiner, tørretumblere, autoklaver), validere cyklusparametre (tid, temperatur, vaskemiddelkoncentration) og demonstrere biobelastningsreduktion til acceptable grænser. Dette kræver mikrobiologiske udfordringsundersøgelser (podning af moppehoveder med målorganismer, bearbejdning gennem fuld vaskecyklus, genopretning og kvantificering af overlevende) og periodisk revalidering, når cyklusparametrene ændres, eller udstyret serviceres.
- Cycle-count sporing og pensionskriterier: Each reusable mop head has a qualified service life (e.g., 50 autoclave cycles for polyester sealed-edge mops). Facilities must implement tracking systems—barcoding, manual logs, or RFID tags—to record usage history and enforce retirement at end of qualified life. Tracking failures introduce risk: over-aged mops shed particles and bioburden, but premature retirement wastes inventory and increases cost.
- Sterility or bioburden verification: If reusable mops are autoclaved for use in Grade A/B areas, the sterilization cycle must be validated per ISO 11137 or equivalent, with sterility indicators placed in representative load configurations. Facilities without autoclave capacity must either send mops to external sterilization vendors (adding turnaround time and cost) or accept elevated bioburden risk.
- Investigation documentation: When EM excursions occur, investigation teams must review cleaning tool history—which mop was used, when was it last laundered, what was its cycle count, what areas was it previously used in. This forensic analysis requires complete traceability records. Gaps in documentation (missing laundry logs, unrecorded mop assignments) extend investigations and complicate CAPA closure.
Disposable mops eliminate most of this validation infrastructure. Single-use tools require only receiving inspection (verifying sterility certificates, visual inspection for packaging integrity) and material qualification (particle generation testing, chemical compatibility validation)—no laundry validation, no cycle-count tracking, no reprocessing documentation.
Increasing Adoption in Fill-Finish & Biotek
Market data shows accelerating adoption of validated single-use cleaning consumables in pharmaceutical and biotech manufacturing, driven by regulatory compliance pressures and operational economics.
Fill-Finish Lines and Aseptic Processing
Fill-finish operations—sterile filling, lyophilization loading, vial capping—operate under ISO Class 5 (Grade A) unidirectional airflow with ISO Class 7 (Grade B) backgrounds. Particle limits are extreme: 3,520 particles ≥0.5 µm per cubic meter for ISO 5, versus 352,000 particles/m³ for ISO 7. Any cleaning tool generating measurable particle contribution risks classification excursions.
Disposable cleanroom mops validated for particle generation (<50 particles/m² for polyester sealed-edge; <10 particles/m² for premium continuous-filament designs) deliver predictable, consistent performance across all uses. Reusable mops degrade over time—edge sealing separates, fibers break, particle generation increases—creating variable contamination contribution that complicates environmental monitoring trend analysis.
Biotech and Cell Therapy Manufacturing
Biologic drug substance and cell therapy manufacturing operate in smaller-batch, higher-potency paradigms than traditional small-molecule pharma. Contamination events that might be tolerable in a 10,000-dose tablet batch become catastrophic in a 50-dose personalized CAR-T product. Biotech facilities adopt disposable cleaning tools as part of broader single-use system strategies:
- Single-use bioreactors and fluid paths: Disposable bag-based systems eliminate cleaning validation for vessels and reduce cross-contamination risk between batches. Extending this logic to floor cleaning maintains system-wide single-use contamination control philosophy.
- Contract manufacturing and multi-product facilities: CDMOs handling multiple client products in the same cleanroom suites face stringent cleaning validation and cross-product contamination prevention requirements. Disposable mops simplify changeover—every new campaign begins with fresh cleaning tools, eliminating carryover risk from previous products without extensive cleaning validation studies.
- Lean operational models: Biotech startups and small manufacturers often lack in-house laundry infrastructure. Outsourcing reusable mop laundering adds cost, turnaround time, and third-party contamination risk. Disposable mops collapse the supply chain into direct vendor delivery of pre-sterilized, ready-to-use consumables.
Industry analysis confirms that EU GMP Annex 1 “compels pharmaceutical producers to switch to sterile-grade, single-use apparel and wipes,” driving adoption of validated disposable consumables across cleaning categories. Pre-saturated sterile wipes already capture substantial market share in aseptic suites; disposable mop systems follow the same adoption curve as facilities extend single-use contamination control strategies to floor cleaning.

Cost Comparison: Disposable vs Reusable
The higher unit cost of disposable cleanroom mops—typically $8–25 per mop pad versus $15–40 for a reusable mop with 50–100 cycle lifespan—creates initial sticker shock for procurement teams. But total cost of ownership (TCO) analysis reveals that disposable systems often deliver lower lifetime costs in high-risk pharmaceutical applications once reprocessing, validation, and contamination failure costs are included.
Direct Cost Elements
Engangsmoppesystemer
- Mop pad unit cost: $8–15 per pad for non-sterile polyester sealed-edge disposables (ISO 7–8 applications); $15–25 per pad for gamma-irradiated sterile disposables (ISO 5–6 Grade A/B areas)
- Handle systems: $50–150 per reusable aluminum or stainless steel handle (compatible with disposable pad attachment systems); handles have multi-year service life
- Storage and inventory: Minimal—disposables ship in compact packaging, require no segregation by cycle count or sterilization status
- Waste disposal: Classified pharmaceutical waste handling (typically $0.50–2 per pound); disposable mop pads weigh 50–150 grams, adding $0.05–0.30 per mop in waste disposal cost
Genanvendelige moppesystemer
- Initial mop head cost: $15–40 per reusable sealed-edge polyester or microfiber mop head
- Laundering cost per cycle: $2–5 per mop (commercial laundry processing, detergent, utilities); in-house laundering reduces this to $1–3 per cycle but requires capital equipment investment
- Autoclave sterilization (if required): $3–8 per cycle including utilities, biological indicators, documentation, and equipment amortization
- Cycle-count tracking system: $5,000–20,000 for barcode or RFID infrastructure (scanners, software, label printing) plus $0.50–1 per mop per cycle in tracking labor
- Inventory carrying cost: 2× to 3× mop inventory required to maintain adequate availability during laundry turnaround cycles
Hidden Indirect Costs (Often Overlooked)
The decisive TCO factors often lie in hidden operational costs that procurement teams underestimate when evaluating unit price alone.
Validation and Documentation Burden
- Initial laundry validation: 120–160 hours of QA specialist time ($60–100/hour) for IQ/OQ/PQ = $7,200–16,000
- Mop material qualification through lifecycle: 40–60 hours for particle testing at new and end-of-life conditions, chemical compatibility validation = $2,400–6,000
- Autoclave sterilization validation (if applicable): 60–80 hours for cycle development, biological indicator studies, SAL demonstration = $3,600–8,000
- Annual revalidation: 40–60 hours per year = $2,400–6,000 annually
- Disposable validation by comparison: 20–30 hours for material qualification (often provided by vendor) + receiving procedure development = $1,200–3,000 one-time cost, with no annual revalidation
Environmental Monitoring Investigation Costs
EM excursions traced to cleaning tool contamination trigger multi-week investigation cycles:
- Investigation labor: 20–40 hours per excursion (microbiologist time, QA documentation, deviation processing) = $1,200–4,000 per event
- Root cause analysis: Forensic review of mop assignment history, laundry logs, cycle counts, reprocessing records
- CAPA implementation: Procedure revisions, retraining, enhanced monitoring—often 40–80 additional hours = $2,400–8,000
- Batch impact risk: In worst-case scenarios, EM failures during aseptic filling trigger batch holds, sterility test expansion, or batch rejection—costs ranging from $50,000 (small batch hold and investigation) to $500,000+ (batch rejection and regulatory notification)
Faciliteter, der oplever selv en EM-fejl om året med moderat undersøgelsesbyrde ($5.000-10.000 i arbejdskraft og dokumentation), opvejer betydelige omkostninger til engangsmoppe.
Arbejdseffektivitetsgevinster
- Eliminering af sporing af cyklustælling: 60–75 timer om året sparet på tværs af alle moppeoperationer = $1.800–3.000 årlig værdi (ved $30/time blandet operatørpris)
- Eliminering af vaskerilogistik: 100–200 timer sparet om året (snavset opsamling, fyldning af vaskecyklus, sterilisering, omfordeling) = $3.000–6.000 årlig værdi
- Træningsforenkling: Reduceret SOP-kompleksitet sparer 10-20 timer om året i levering af træning og kompetencebekræftelse = $600-2.000 årlig værdi
Total Cost of Ownership Comparison (ISO Class 7 Aseptic Support Area Example)
Overvej et 500 m² ISO klasse 7 farmaceutisk produktionsstøtteområde, der vaskes dagligt (365 dage/år), hvilket kræver 2 moppepuder pr. rengøringscyklus:
Scenario A: Genanvendelige mopper
- Direkte omkostninger: 730 moppebrug/år ÷ 75-cyklus moppelevetid = 10 mopper købt årligt til $30 hver = $300
- Hvidvaskning: 730 cyklusser × $3 pr. cyklus (intern behandling) = $2.190
- Autoklave sterilisering: 730 cyklusser × $5 pr. cyklus = $3.650
- Cykelsporingsarbejde: 730 cyklusser × $0,75 pr. cyklus = $548
- Initial validation (amortiseret over 5 år): $12.000 ÷ 5 = $2.400 pr. år
- Annual revalidation: $4.000
- EM undersøgelsesbyrde (1 begivenhed hvert andet år, konservativt): $7.500 ÷ 2 = $3.750 årligt gennemsnit
- Samlet årlig TCO: $16.838
Scenario B: Engangsmopper (gamma-sterile)
- Direkte omkostninger: 730 moppepuder × 18 USD pr. pude = 13.140 USD
- Håndtere amortisering: $100 ÷ 5-årig levetid = $20 pr. år
- Waste disposal: 730 pads × $0,15 pr. pad = $110
- Indledende materialekvalifikation (amortiseret over 5 år): $2.000 ÷ 5 = $400 pr. år
- Ingen omkostninger til revalidering, tøjvask, sterilisering eller sporing
- Reduceret EM undersøgelsesbyrde: Næsten nul rengøringsværktøj-relaterede udflugter = $0
- Gevinster i arbejdseffektiviteten: $4.000 om året (sporing, vaskerilogistik, træning)
- Samlet årlig TCO: $13.670 – $4.000 (effektivitetsgevinster) = 9.670 USD nettoomkostninger
Resultat: Engangsmopper leverer 42 % lavere TCO ($9.670 vs. $16.838) på trods af højere enhedsomkostninger, drevet af eliminering af oparbejdningsinfrastruktur, reduktion af valideringsbyrde og undgåelse af EM-undersøgelser.
Decision Thresholds: When Disposables Justify Cost
Engangsmopper til renrum leverer en gunstig TCO under disse forhold:
- Højrisiko ISO-klassifikationer (ISO 5–7): Partikelgrænser og forureningskontrolmargener er stramme; EM-fejlrisiko og undersøgelsesbyrde er høj
- Faciliteter med EM-fejlhistorik: Selv en forureningsudflugt om året sporet til rengøringsværktøj ($5.000-10.000 undersøgelsesomkostninger) opvejer betydelige udgifter til engangsmoppe
- Små til mellemstore operationer (<1.000 m² renrumsareal): Genanvendelige systemer kræver faste validering og infrastrukturomkostninger, der ikke nedskaleres; engangsomkostninger skaleres lineært med brug
- Faciliteter uden in-house vaskeri: Outsourcet vasketøj tilføjer 5-10 USD pr. moppe pr. cyklus plus leveringsforsinkelser; engangsartikler eliminerer tredjepartsafhængighed
- Lean operationelle teams: Bioteknologiske startups og CMO'er med begrænset QA/vaskeri medarbejderstaben drager fordel af reduktion af valideringsbyrden og arbejdseffektivitetsgevinster
- Multiproduktfaciliteter (CDMO'er): Rengøringsvalidering for produktskift er forenklet, når rengøringsværktøjer er engangsbrug; ingen risiko for overførsel mellem kampagner
Genanvendelige mopper forbliver omkostningseffektive, når:
- Storskala produktion (>2.000 m² daglig mopping): Højt moppeforbrugsvolumen (1.000+ mopper/år) fører til engangsomkostninger over genanvendelige TCO, selv med oparbejdningsbyrde inkluderet
- ISO 8 støtteområder med lav EM risiko: Lavere krav til forureningskontrol reducerer undersøgelsesbyrden; genanvendelig økonomi forbedres i miljøer af lavere kvalitet
- Valideret in-house vaskeri infrastruktur findes allerede: Facilities with mature, qualified laundry operations and robust cycle-count tracking reduce reprocessing cost differential
- Sustainability prioritization: Some organizations prioritize waste reduction over TCO optimization; validated reusable systems with documented contamination control deliver acceptable risk profiles
Best Use Cases for Disposable Mops in GMP Environments
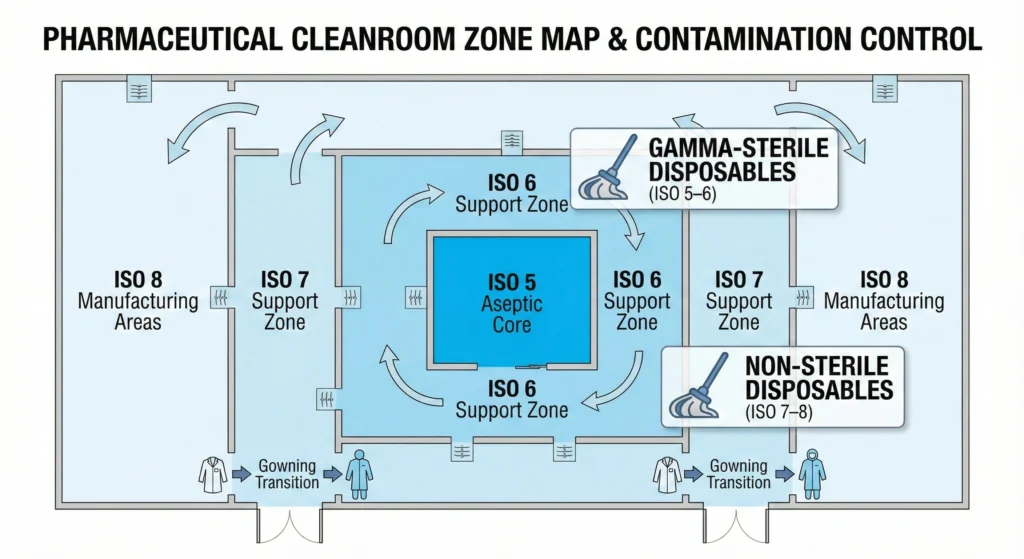
Disposable cleanroom mops deliver maximum value when contamination control requirements, operational constraints, and risk profiles align with their strengths. This section maps specific facility types, ISO classifications, and operational scenarios where disposable systems justify their cost through superior contamination control and reduced total cost of ownership.
ISO Class 5 (Grade A) Aseptic Cores
Krav: Unidirectional airflow, particle limit 3,520 particles ≥0.5 µm/m³, terminal sterilization or aseptic processing, zero bioburden tolerance
Why Disposables Excel:
- Gamma-sterile disposables deliver SAL 10⁻⁶: Reusable mops require autoclaving for sterility—introducing sterilization cycle validation burden, biological indicator requirements, and potential sterilization failures. Gamma-irradiated disposables arrive pre-sterilized with certificates, eliminating in-house sterilization requirements.
- Zero cross-contamination risk: Grade A areas demand absolute contamination control. A single mop cross-assignment (using an ISO 7 mop in Grade A due to inventory shortage) introduces bioburden into aseptic zones. Disposables eliminate this failure mode.
- Predictable particle performance: Partikelgrænserne er ekstreme (3.520/m³). Genanvendelige mopper udskiller stigende partikler efterhånden som de ældes; engangs polyestermopper med forseglet kant vedligeholdes <50 partikler/m² på tværs af alle anvendelser.
- Forenklet validering: Faciliteter undgår autoklaveringsvalidering, validering af sporing af cyklustællinger og undersøgelser af reduktion af vaskeri-biobyrde – kritisk for små bioteknologiske faciliteter med begrænsede QA-ressourcer.
Typiske applikationer:
- Aseptiske påfyldningslinjer (påfyldning af hætteglas, påfyldning af sprøjter, fyldning af frysetørring)
- Sterile blandingssuiter (hospitalapoteker, blandingsfaciliteter)
- Fremstilling af celleterapi (CAR-T-behandling, personlig medicinproduktion)
- Biologiske sikkerhedsskabe og isolator rengøring
MIDPOSI anbefaling: Gamma-bestrålede engangsmoppepuder af polyester med forseglet kant med valideret <50 partikler/m² ydeevne og SAL 10⁻⁶ sterilitetscertifikater
ISO Class 6–7 (Grade B/C) Aseptic Support Areas
Krav: Partikelgrænser 35.200/m³ (ISO 6) til 352.000/m³ (ISO 7), kontrolleret biobelastning, materialeoverførsel forureningskontrol
Why Disposables Excel:
- Forebyggelse af materialeoverførsel: Grad B/C områder fungerer som luftsluser og materialeoverførselszoner mellem områder af lavere kvalitet og klasse A-kerner. Genanvendelige mopper, der bruges i flere zoner, bliver forureningsvektorer. Engangsartikler sikrer, at hver rengøringscyklus begynder med et valideret, kontamineringsfrit værktøj.
- Reduceret EM undersøgelsesbyrde: Områder af klasse B/C står over for høj EM-kontrol – enhver partikel- eller biobelastningsudflugt udløser undersøgelse. Engangsmopper med forudsigelig ydeevne forenkler EM-trend og reducerer rengøringsværktøj-relaterede udflugter.
- Arbejdseffektivitet i multirumsfaciliteter: Aseptiske suiter inkluderer ofte 5-10 indbyrdes forbundne Grad B/C støtterum. Engangssystemer eliminerer moppesporing på tværs af rum, verifikation af cyklustælling ved hver brug og vaskeri adskillelseslogistik.
Beslutningsfaktorer:
- Faciliteter med hyppige EM-udflugter gavner de fleste - selv en undersøgelse om året ($5.000-10.000 omkostninger) opvejer betydelige udgifter til engangsmoppe
- Små til mellemstore operationer (<500 m² samlet klasse B/C-areal) se gunstige engangs-TCO
- Faciliteter uden valideret in-house vaskeri eliminerer tredjeparts vasketøjsafhængighed
Typiske applikationer:
- Fyld-finish linje baggrunde og støtte korridorer
- Materialeoverførselsluftsluser mellem Grad C og Grad A områder
- Beklædningsrum, der serverer aseptiske kerner
- Udstyr rengøring og iscenesættelse områder
MIDPOSI anbefaling: Gamma-bestrålede engangsartikler til kritiske Grad B-områder, der støder op til Grad A-zoner; ikke-sterile engangsartikler af polyester med forseglet kant, der er acceptable til Grad C-støtteområder med valideret <100 partikler/m² ydeevne
ISO Class 8 (Grade D) Manufacturing & Support Areas — Selective Use
Krav: Partikelgrænse 3.520.000/m³, rutinemæssig biobelastningsovervågning, lavere risiko for forureningskontrol
Når engangsartikler giver mening:
- Multiproduktfaciliteter (CMO'er/CDMO'er): Rengøring af produktskift kræver valideret eliminering af krydsproduktkontamination. Engangsmopper forenkler udskiftningen – ingen risiko for tidligere produktrester fra genanvendelige mopper.
- Fremstilling af højpotens lægemidler: Meget potente forbindelser (cytotoksiske stoffer, hormoner) kræver streng rengøringsvalidering for at forhindre operatøreksponering og krydskontaminering. Engangsrengøringsværktøj eliminerer risiko for overførsel uden omfattende rengøringsvalideringsundersøgelser.
- Faciliteter med begrænset vaskeri infrastruktur: Bioteknologiske startups og små producenter uden in-house vaskeri undgår at bygge eller outsource vaskeri ved at bruge engangsartikler på tværs af alle ISO-kvaliteter.
- Forureningshændelsesreaktion: Efter EM-udflugter eller kontamineringsbegivenheder kan faciliteter midlertidigt skifte til engangsartikler i berørte områder under undersøgelse af årsagen til årsagen og CAPA-implementering – for at sikre, at rengøringsværktøjer ikke bidrager til løbende kontaminering.
Når genanvendelige materialer forbliver optimale:
- Storskala ISO 8 støtteområder (>1.000 m² lager, korridorsystemer, generel produktion), hvor forureningsrisikoen er lav, og moppeforbruget er højt
- Faciliteter med valideret in-house vaskeri og robuste systemer til sporing af cyklustælling er allerede i drift
- Organisationer, der prioriterer affaldsreduktion og bæredygtighed, hvor forureningsrisikoprofilen er acceptabel
Typiske anvendelser for engangsartikler:
- Multi-produkt API-fremstillingspakker (CMO-scenarier)
- Områder til fremstilling af høj-potente lægemidler
- Udstyr, der renser mellemrum, der betjener zoner af højere kvalitet
- Forureningsundersøgelses isolationszoner
MIDPOSI anbefaling: Ikke-sterile engangsartikler af polyester med forseglet kant med valideret <100 partikler/m² ydeevne; omkostningseffektiv til selektiv udrulning, samtidig med at genanvendelige systemer til lavrisiko-støtteområder opretholdes
Biotech Startups & Small-Scale GMP Facilities
Driftsprofil: <20 renrumspersonale, begrænsede QA/valideringsressourcer, ingen intern vaskeriinfrastruktur, slank driftsmodel, agil fremstilling (små serier, hyppige produktskift)
Hvorfor engangsartikler er strategiske:
- Ingen investering i vaskeriinfrastruktur: Eliminerer $50.000-200.000 kapitalinvesteringer i industrielle vaskemaskiner, tørretumblere, autoklaver og vaskerumskonstruktion
- Reduktion af valideringsbyrden: QA-hold, der strækker sig tyndt over flere valideringsaktiviteter (udstyrskvalificering, rengøringsvalidering, procesvalidering) drager fordel af eliminering af vasketøjsvalidering, validering af cyklustælling og steriliseringsvalidering
- Forenklet operatøruddannelse: Enkelt SOP ("Fjern fra pakke, brug én gang, kasser") reducerer træningstid og kompetenceverifikationsbyrde
- Skalerbare omkostninger: Engangsomkostninger skaleres lineært med produktionsvolumen - ideel til startups i tidlige produktionsfaser med variabel batchfrekvens
- Hurtig opstart af anlæg: Nye GMP-suiter bliver hurtigere operationelle uden kvalificering af vaskeudstyr, udvikling af oparbejdningsprocedurer og validering af sporingssystem
Typiske profiler:
- Biotekstartups i fase I/II klinisk fremstilling (1-5 batches pr. måned)
- Celle- og genterapiproducenter (personlig medicin, små batchstørrelser)
- Akademiske medicinske centre med GMP-kompounderingssuiter
- Speciale API-producenter med <500 m² samlet renrumsareal
MIDPOSI anbefaling: Komplet engangsmoppeprogram på tværs af alle ISO-kvaliteter - gamma-sterile engangsartikler til aseptiske kerner af klasse A/B, ikke-sterile engangsartikler til grad C/D-støtteområder - parret med valideringspakke (partikeldata, sterilitetscertifikater, materialekompatibilitetsrapporter) for at accelerere regulatorisk beredskab
Contract Manufacturing Organizations (CMOs/CDMOs)
Operationel udfordring: Flere klientprodukter fremstillet i delte renrumssuiter; strenge krav til rengøringsvalidering og forebyggelse af kontaminering på tværs af produkter; hyppige produktskift
Hvorfor engangsartikler forenkler driften:
- Ingen risiko for produktoverførsel: Rengøringsvalideringsundersøgelser skal demonstrere <10 ppm rest af tidligere produkt. Engangsrengøringsværktøj eliminerer en overførselsvej fuldstændigt - ingen risiko for tidligere produktrester indlejret i genanvendelige moppefibre.
- Forenklet overgangsdokumentation: Tjeklister for produktskift inkluderer verifikation af rengøringsværktøj. Engangsartikler eliminerer retsmedicinsk sporing ("Blev denne moppe brugt i tidligere kampagne? Hvad var dens cyklusantal? Hvornår blev den sidst vasket?") - operatører bruger simpelthen friske engangsartikler.
- Kundens tillid: Farmaceutiske klienter, der udfører audits og due diligence, evaluerer kontrol med krydskontaminering. Engangsrengøringssystemer demonstrerer proaktiv kontamineringsforebyggelse og reduceret valideringskompleksitet.
- Fleksibel kapacitet: CMO'er oplever varierende produktionsplaner. Engangsomkostninger skaleres med den faktiske produktionsaktivitet, hvilket undgår faste driftsomkostninger for vaskeri i perioder med lav udnyttelse.
Typiske applikationer:
- Sterile fill-finish CMO'er, der håndterer flere kundeprodukter
- API-fremstilling CMO'er med multi-produkt suiter
- CDMO'er med små molekyler og biologiske lægemidler
- Specialiseret fremstilling (API'er med høj styrke, kontrollerede stoffer)
MIDPOSI anbefaling: Engangsmoppeprogrammer til aseptiske kerner (ISO 5-7 områder), hvor produktskift finder sted; selektiv genanvendelig implementering i dedikerede ISO 8-supportområder, der betjener enkeltproduktkampagner

MIDPOSI Disposable Cleanroom Mop Line (Product Selector)
MIDPOSI fremstiller engangsrenrumsmopper af farmaceutisk kvalitet, der er udviklet specifikt til GMP-kontamineringskontrolapplikationer. Alle produkter leveres med komplette valideringsdokumentationspakker – testdata for partikelgenerering, sterilitetscertifikater (for gammabestrålede produkter), materialekompatibilitetsrapporter og analysecertifikater – hvilket muliggør hurtig regulatorisk kvalifikation og facilitetstart.
MDCR-GS100 Gamma-Sterile Disposable Mop (ISO 5–6 Grade A/B)
Nøglespecifikationer:
- Materiale: 100 % forseglet kant polyester kontinuerlig filament strik, farmaceutisk renrum-fremstillet
- Sterilisering: Gammabestråling (25-50 kGy dosis) opnår Sterility Assurance Level (SAL) 10⁻⁶
- Partikelgenerering: <50 partikler ≥0,5 µm pr. kvadratmeter moppet (valideret i henhold til ISO 14644-1 testmetoder)
- Bioburden-baseline: Ingen levedygtige mikroorganismer (terminal sterilisering)
- Dimensioner: 40 cm × 15 cm moppehoved (kompatibel med MIDPOSI håndtagssystemer i aluminium/rustfrit stål)
- Emballage: Steril emballage i dobbeltposer med afrivningsforseglinger; inderpose velegnet til overførsel til klasse A/B områder
- Dokumentation: Steriliseringscertifikat (leveret dosis, biologiske indikatorresultater, SAL-validering), partikeltestdata, materialekompatibilitetsrapport (IPA, hydrogenperoxid, kvaternære ammoniumdesinfektionsmidler)
Validerede ansøgninger:
- Aseptiske påfyldningslinjer (påfyldning af hætteglas, samling af sprøjter, lyofilisering)
- Sterile blandingssuiter (hospitalapoteker, 503B-faciliteter)
- Celle- og genterapifremstilling af renrum
- Biologiske sikkerhedsskabe, isolatorer og systemer med begrænset adgangsbarriere (RABS)
Koste: $18–22 per moppepude (volumenpriser er tilgængelige for faciliteter, der bruger >500 bind om året)
Valideringsstøtte: Komplet valideringspakke inkluderet i den indledende ordre—partikelgenereringstestrapport i henhold til ISO 14644-1, Steriliseringscertifikat med dosiskortlægning og biologiske indikatorresultater, undersøgelse af ekstraherbare materialer, der viser <10 ppm udvaskelige efter kontakt med desinfektionsmiddel
MDCR-NS200 Non-Sterile Disposable Mop (ISO 7–8 Grade C/D)
Nøglespecifikationer:
- Materiale: Forseglet-kant polyester mikrofiberblanding, renrumsfremstillet med valideret lav-fnug ydeevne
- Bioburden-baseline: <10 CFU pr. moppepude (validerede produktionskontroller, rutinemæssig biobelastningstest)
- Partikelgenerering: <100 partikler ≥0,5 µm pr. kvadratmeter moppet
- Kemisk kompatibilitet: Valideret til isopropanol, hydrogenperoxid (3-6%), kvaternære ammoniumforbindelser, chlordioxid og phenoliske desinfektionsmidler
- Dimensioner: 40 cm × 15 cm moppehoved (universelt fastgørelsessystem, der er kompatibelt med de fleste renrumsmoppehåndtag)
- Emballage: Forseglet emballage med lavt fnug med genlukkelig lukning (50 moppepuder pr. pose)
- Dokumentation: Partikeltestdata, biobyrdegrundlinjerapport, materialekompatibilitetsvalideringsrapport
Validerede ansøgninger:
- ISO klasse 7 aseptiske støtteområder (fyld-finish-baggrunde, beklædningsrum)
- ISO klasse 8 farmaceutiske fremstillingsområder (API-produktion, tabletfremstilling, pakkepakker)
- Multiproduktfaciliteter, der kræver omskiftningsrengøring (CMO/CDMO-drift)
- Forureningsundersøgelses isolationszoner
Koste: 8-12 USD pr. moppepude (mængderabat ved >1.000 bind om året)
Valideringsstøtte: Leverandørvalideringspakke leveret - partikelgenereringstestrapport, bioburden-baselinedokumentation (gennemsnitlige CFU- og 95. percentilværdier), kemisk kompatibilitetsmatrix, der viser kompatibilitet med vigtige farmaceutiske desinfektionsmidler
MDCR-PSAT Pre-Saturated Disposable Mop (Ready-to-Use with IPA or Disinfectant)
Nøglespecifikationer:
- Materiale: Polyester med forseglet kant med valideret sugeevne og desinfektionsmiddeltilbageholdelse
- Mætningsmuligheder: 70% isopropanol/30% vand (USP-kvalitet); 3% hydrogenperoxid; eller facilitetsspecificeret desinfektionsmiddel (brugerdefineret mætning tilgængelig)
- Sterilisering: Gammabestråling efter mætning, der opnår SAL 10⁻⁶ (sterilt desinfektionsmiddel og steril moppe)
- Partikelgenerering: <50 partikler/m² (sammenlignelig med MDCR-GS100)
- Udløb: 24 måneders holdbarhed (forseglet folieemballage forhindrer fordampning)
- Emballage: Individuelle folieposer (klar til at overføres til klasse A/B-områder) eller 25-mopper etui til brug i klasse C/D
Validerede ansøgninger:
- Pletrensning og spildrespons i aseptiske områder
- Småareal mopping (<50 m² rum), hvor forberedelse af desinfektionsmiddel er ubelejligt
- Nødrengøring under produktion (ingen afbrydelse for blanding af desinfektionsmiddel)
- Faciliteter, der søger at eliminere præparat af desinfektionsmiddel og eksponering for operatørkontakt
Koste: 22-28 USD pr. moppepude (højere omkostninger på grund af mætning af desinfektionsmiddel og sterilisering efter mætning)
Valideringsstøtte: Komplet valideringspakke inklusive test af desinfektionsmiddeleffektivitet (log reduktion mod målorganismer), partikelgenereringsdata efter mætning, sterilitetscertifikat og kompatibilitetstest, der viser ingen overfladerester eller materialenedbrydning
MIDPOSI Handle Systems (Reusable, Compatible with All Disposable Mop Pads)
MDH-AL100 aluminiumshåndtag (ISO 7-8 applikationer):
- Letvægts anodiseret aluminium konstruktion (150 gram)
- Autoklaverbar til 134°C (valideret til 100 steriliseringscyklusser)
- Universal fastgørelsesklips kompatibel med MDCR-GS100, MDCR-NS200 og MDCR-PSAT moppepuder
- Pris: $85 pr håndtag
MDH-SS200 rustfrit stål håndtag (ISO 5–6 Grade A/B-applikationer):
- 316L rustfri stålkonstruktion (korrosionsbestandig, ingen partikelgenerering)
- Autoklaverbar til 134°C (valideret til 200+ steriliseringscyklusser)
- Glat overfladefinish uden sprækker (let desinfektion, ingen biobelastning)
- Universelt fastgørelsessystem
- Pris: $140 pr håndtag
How to Order and Request Validation Packages
MIDPOSI yder komplet teknisk og regulatorisk support til faciliteter, der evaluerer eller implementerer engangs-renrumsmoppeprogrammer:
Prøvesæt til pilottestning:
- Anmod om 10-20 moppeprøver (MDCR-GS100 eller MDCR-NS200) til intern evaluering
- Inkluderer: Valideringspakke (partikeldata, sterilitets-/biobelastningsrapporter, kompatibilitetsmatrix), brugervejledning og teknisk specifikationsark
- Pilottest gør det muligt for faciliteterne at validere ydeevnen under faktiske driftsforhold før fuld indkøb
Valideringspakkeindhold (Leveres med alle ordrer):
- Partikelgenereringstestrapport: ISO 14644-1-kompatibel test, der dokumenterer partikler ≥0,5 µm pr. m² moppet
- Sterilitetscertifikat (for gamma-bestrålede produkter): Leveret dosis, biologiske indikatorresultater, SAL 10⁻⁶ validering
- Bioburden baseline rapport (for ikke-sterile produkter): Kolonidannende enheder (CFU) pr. moppepude, testet i henhold til USP <61> og <62>
- Validering af materialekompatibilitet: Kompatibilitetsmatrix, der ikke viser nogen nedbrydning eller øget partikeldannelse efter kontakt med facilitetsspecificerede desinfektionsmidler (IPA, hydrogenperoxid, kvaternære ammoniumforbindelser, chlordioxid)
- Undersøgelse af ekstraherbare og udvaskbare materialer: Demonstrerer <10 ppm udvaskbare efter desinfektionsmiddelkontakt (GC-MS-analyse pr. ICH Q3C)
- Overensstemmelsescertifikat: Verifikation af partispecifik overensstemmelse med partikel-, biobelastnings- og sterilitetsspecifikationer
Teknisk rådgivning:
- MIDPOSI-kontamineringskontrolspecialister giver facilitetsspecifikke anbefalinger baseret på ISO-klassificering, EM-fejlhistorik, operationel skala og TCO-optimering
- Understøttelse af regulatoriske indsendelser: Valideringsoversigter formateret til FDA, EMA eller PMDA regulatoriske ansøgninger
- Brugerdefinerede valideringsundersøgelser tilgængelige (kontaktresistenstestning, specifik desinfektionsmiddelkompatibilitet, udvidede partikelgenereringsundersøgelser)
Kontakt for priser, prøver og teknisk support:
- E-mail: [email protected]
- Responstid: Pristilbud og valideringspakker leveret inden for 12 timer efter anmodning
- Leveringstid: Lagervarer sendes inden for 48 timer; brugerdefinerede konfigurationer (specifikke dimensioner, tilpasset desinfektionsmiddelmætning) sendes inden for 2 uger









