A QA-focused technical guide for GMP material entry into Grade A/B zones—covering peel-and-pass logic, packaging integrity risks, and audit-defensible controls aligned with EU GMP Annex 1 expectations.

Sterilitet opnås under fremstilling, men de fleste forureningshændelser opstår under aseptisk overførsel—when materials move from uncontrolled areas to Grade C, then B, and finally A. Without redundant barriers, the mop packaging surface can act as a carrier for particulates and bioburden.
I praksis steril moppeemballage functions as a controlled boundary: the outer bag is stripped at the interface, while the inner bag remains sterile until the point of use. This is the operational basis of skræl-og-passér.
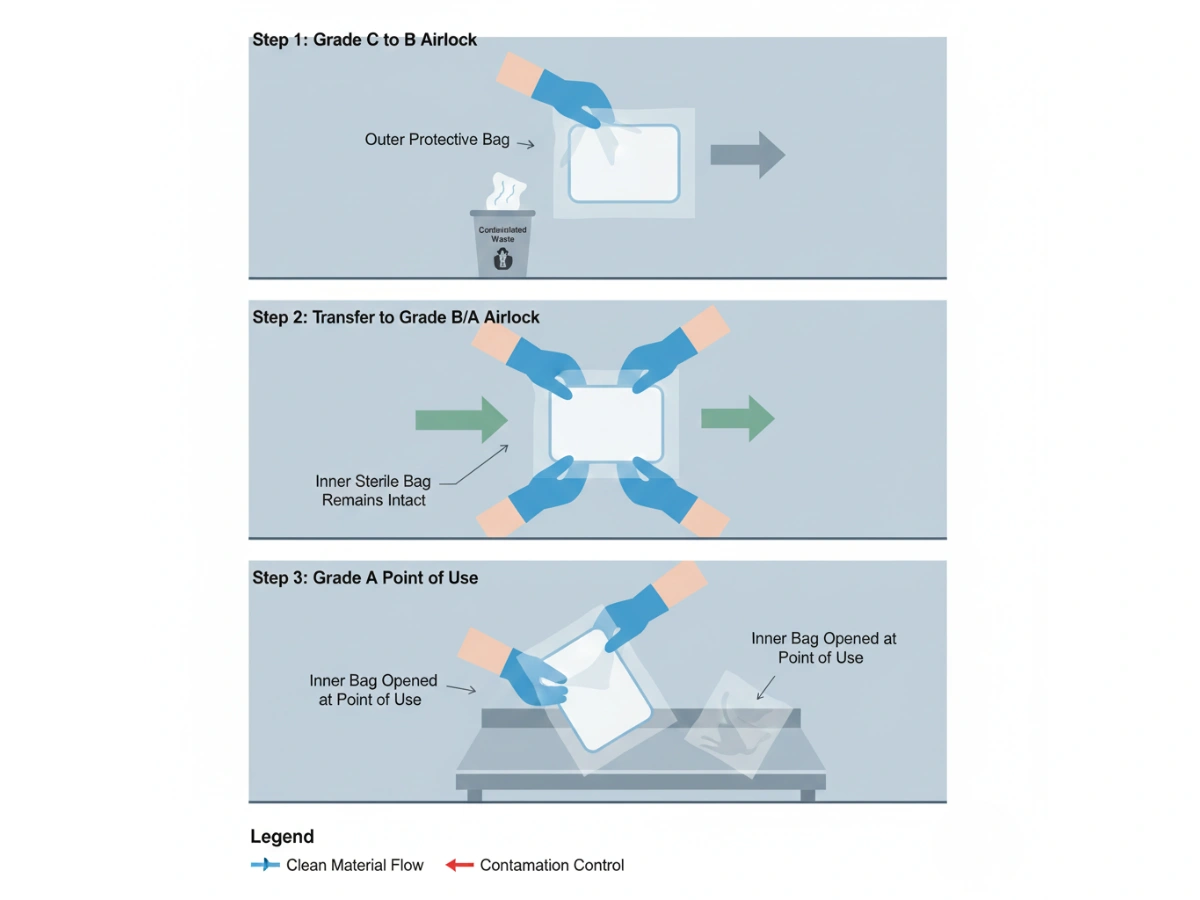

For Grade A/B operations, the core question is not “sterile at release,” but “sterile at point-of-use.” Double-bagging reduces transfer variability by removing the external surface at the boundary rather than relying on wipe-down success every time.

Use this simplified logic to align packaging selection with grade, transfer method, and validation burden. For Grade A/B, an engineered barrier is typically preferred over manual disinfection dependence.
| Beslutningsfaktor | Single-Bag Emballage | Dobbeltpose emballage |
|---|---|---|
| Overfør kontrol | Kræver valideret wipe-down og disciplin | Peel-and-pass fjerner forurening ved grænsen |
| Fejltilstand | Enkelt point of failure | Overflødige sterile barrierelag |
| Revisions forsvarlighed | Stærkt afhængig af SOP-udførelsesbeviser | Stærkere konstrueret kontrolfortælling for CCS |
| Operationel arbejdsbyrde | Højere (desinfektion, overvågning, afvigelser) | Lavere (standardiseret strip-at-boundary proces) |



Vi kontakter dig inden for 1 arbejdsdag, vær venligst opmærksom på e-mailen med suffikset “*@midposi.com”.