Indledning
Selecting the right cleanroom mop supplier is not only a purchasing decision. It affects product quality, contamination control, audit readiness, and long-term operating cost. The biggest mistake many buyers make is focusing too heavily on unit price while underestimating quality inconsistency, poor traceability, long lead times, and documentation gaps.
Supplier qualification should assess quality, technical capability, supply chain stability, regulatory compliance, and cost together, not separately. A structured weighted matrix makes the selection more defendable than choosing a supplier based on quotation alone. :contentReference[oaicite:2]{index=2}
Supplier Evaluation Dimensions
A strong evaluation model prevents subjective supplier selection. Supplier score should be calculated across five weighted dimensions: quality, technical capability, supply chain, regulatory compliance, and cost & terms. This creates a more defendable selection process than relying on price or relationships alone. :contentReference[oaicite:3]{index=3}
| Dimension | Vægt | What matters most | Typical disqualifier |
|---|---|---|---|
| Kvalitet | 30% | Low-lint validation, consistency, traceability | No validation data or unstable batch performance |
| Technical capabilities | 25% | Material expertise, documentation, customization, problem-solving | No usable technical support or no documentation package |
| Supply chain | 20% | Capacity, lead time, backup plans, geographic resilience | Single point of failure or excessive lead time |
| Regulatory compliance | 15% | QMS maturity, cGMP awareness, audit history | Weak compliance system or unresolved regulatory issues |
| Koste & terms | 10% | Total cost of ownership, MOQ, payment terms, warranty | Low price but poor total commercial fit |
Quality Assessment
Quality is the most heavily weighted dimension for a reason. If the mop itself is not consistent, validated, and traceable, then the rest of the supplier evaluation becomes much less relevant. A low-cost mop with unstable particulate performance can cost more in investigation, audit response, and contamination risk than a higher-priced but controlled product.
What to verify
- Low-lint validation and particle-performance data
- Batch-to-batch consistency
- Lot-level traceability
- Material specifications and release controls
What raises concern
- No independent validation data
- High batch variability
- Incomplete lot traceability
- Supplier claims without supporting reports

| Quality criterion | High score signal | Low score signal |
|---|---|---|
| Low-lint validation | Validated with supporting data | No validation or only marketing claims |
| Material consistency | Minimal batch-to-batch variation | Frequent quality drift |
| Batch traceability | Full lot-level traceability | Only partial or date-based control |
| Validation data availability | Complete technical package available | Missing or fragmented documents |
This section should connect naturally to Low-Lint Cleanroom Mop Materials Explained og Batch Traceability for Cleanroom Consumables.
Technical Capabilities
A cleanroom mop supplier should not only ship product. It should be able to explain why the product performs as it does, what materials are used, how the product behaves with different disinfectants, and what documentation supports its use in higher-control environments.
Technical capability includes material science expertise, R&D support, customization capability, technical documentation, and problem-solving ability. Technical weakness often shows up later as poor support during deviations, validation questions, or customer-specific requirements. :contentReference[oaicite:4]{index=4}
| Technical area | Why it matters | Preferred signal |
|---|---|---|
| Material science expertise | Supports credible material selection and performance claims | Dedicated technical or material team |
| R&D support | Helps with customization, upgrades, and issue resolution | Structured development capability |
| Customization | Important for handle systems, mop head size, sterility route, packaging | Clear customization pathway |
| Teknisk dokumentation | Critical for QA review, validation support, and audit defense | Complete and current technical package |
| Problem-solving capability | Determines how useful the supplier is when problems occur | Fast response with root-cause logic |
Supply Chain Assessment
Cleanroom consumables are often treated as simple products until supply fails. A supplier with only one production site, no backup plan, long lead times, or weak distribution coverage can become a major operational risk even if its product quality looks acceptable.
What to assess
- Production capacity vs your annual demand
- Standard and emergency lead times
- Geographic coverage and distribution model
- Backup factories, contingency plans, and continuity strategy
Typical red flags
- One site only with no backup
- Lead time above eight weeks
- No distribution flexibility
- No clear business continuity or recovery plan
Regulativ overholdelse
Regulatory compliance does not always mean the supplier needs the same certification set as a finished drug manufacturer. But it does mean the supplier should understand controlled-environment expectations, maintain a credible quality system, and be able to support audits or customer reviews with consistent documentation and records.
| Compliance area | What to review | Why it matters |
|---|---|---|
| Quality management system | QMS structure, procedures, version control, CAPA handling | Indicates process discipline |
| Regulatory experience | Years serving regulated industries and audit exposure | Shows familiarity with compliance expectations |
| Audit history | External audit findings and closure maturity | Helps evaluate risk of recurring issues |
| Traceability and records | Lot tracking, release documents, change control | Supports investigation and audit readiness |
Koste & Terms Evaluation
Price should be reviewed, but it should not dominate the decision. The supplier with the lowest unit cost may create the highest total cost if it requires excessive MOQ, carries unstable delivery, lacks documentation, or generates more deviations and review work.
| Commercial factor | What to review | Interpretation |
|---|---|---|
| Price competitiveness | Price vs market and comparable quality level | Low price is only meaningful if quality is stable |
| Payment terms | Net terms, prepayment, flexibility | Better terms reduce cash-flow pressure |
| MOQ & lead time | Commercial entry barrier and planning burden | High MOQ can hide operational inefficiency |
| Warranty / support | Defect response and replacement terms | Important for risk sharing |
| Total cost of ownership | Price + logistics + inventory + risk + documentation support | Better than comparing unit price only |
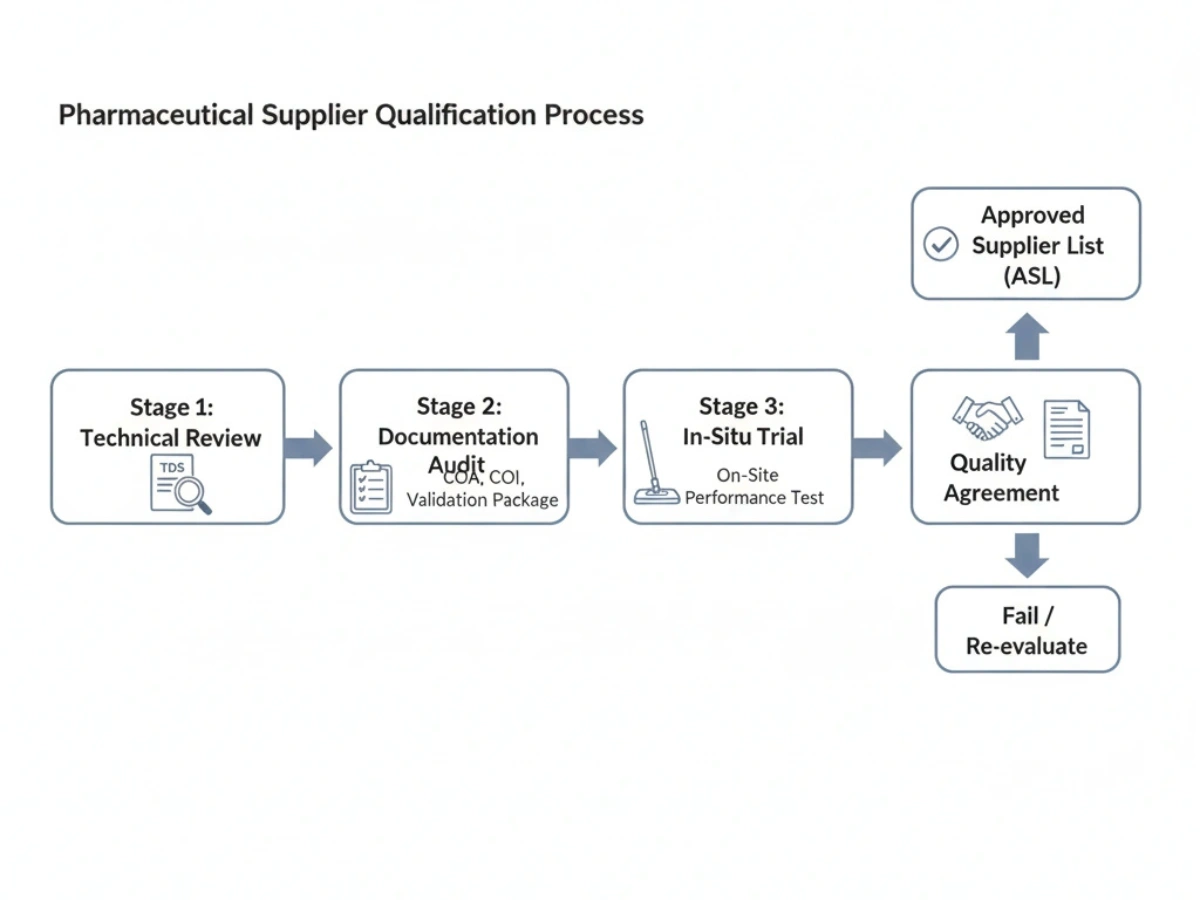
Supplier Qualification Process
A structured qualification timeline should include preparation, document review, site visit if needed, sample evaluation, and final scoring. This should be managed as a staged qualification process rather than a one-call vendor selection. :contentReference[oaicite:5]{index=5}
Week 1: Preparation
Define qualification criteria, assign reviewers, prepare supplier questionnaire, and align internal stakeholders.
Week 2–3: Document Review
Collect quality, technical, compliance, and commercial documents. Review them against evaluation criteria.
Week 4: Site Visit (if needed)
Inspect production, quality controls, documentation control, and problem-solving maturity where the risk justifies it.
Week 5: Sample Evaluation
Review actual mop samples for consistency, lint performance, usability, packaging, and documentation support.
Week 6: Final Assessment
Complete the supplier matrix, compare alternatives, decide approval status, and document rationale.
Supplier Management After Approval
Qualification is not the end of supplier control. Once approved, suppliers should be monitored through KPIs such as on-time delivery, fill rate, defect rate, response time, lead time, and audit findings. This turns supplier qualification from a one-time exercise into a managed quality relationship.
| KPI | Typical target | Why it matters |
|---|---|---|
| On-time delivery | High and stable | Protects cleaning continuity and planning |
| Fill rate | Near complete | Shows supply execution reliability |
| Defect rate | Very low | Reflects real product consistency |
| Responstid | Fast | Matters when issues or deviations occur |
| Audit findings | Minimal and closed effectively | Shows quality-system maturity |
Need Help Evaluating a Cleanroom Mop Supplier?
Get a practical supplier screening strategy based on your ISO class, application risk, documentation requirements, and procurement priorities.
- Supplier screening questions for QA and procurement teams
- Support for documentation, qualification, and sample review
- Guidance on balancing quality, lead time, and total cost
Ofte stillede spørgsmål
How many suppliers should I qualify for cleanroom mops?
In many cases, 3–5 qualified suppliers is a practical structure: preferred suppliers, approved secondary suppliers, and one backup source for contingency.
What are the most common supplier qualification failures?
The most common failures are weak low-lint validation, incomplete technical documentation, inadequate traceability, poor compliance maturity, and unstable lead time or sourcing structure.
How long does supplier qualification take?
A full qualification process often takes around 4–6 weeks depending on document readiness, sample review, and whether a site visit is needed.
Should I use domestic or international suppliers?
Both can work. Domestic suppliers may offer shorter lead times and easier communication, while international suppliers may offer scale or cost advantages.
How do I evaluate supplier technical capability?
Review material knowledge, technical documentation, customization ability, R&D support, and how the supplier responds to detailed technical questions and sample feedback.
How often should I re-qualify suppliers?
Requalification frequency depends on risk and supplier role, but every 2–5 years is common, with earlier review triggered by major quality issues or compliance events.




