In pharmaceutical manufacturing, contamination control failures traced to cleaning tools represent one of the most preventable sources of environmental monitoring excursions and regulatory observations. A 2025 FDA warning letter specifically cited “non-sterile wipes within the ISO 5 aseptic processing area” and “failure to establish an adequate system for cleaning and disinfecting” as CGMP violations that jeopardized batch sterility. The root cause: reusable cleaning tools carrying bioburden and particle contamination from prior use, laundered inadequately, and introduced into aseptic zones without validated reprocessing.
Disposable cleanroom mops eliminate this contamination pathway entirely. Every mopping cycle begins with a fresh, validated mop pad—no cross-contamination risk from previous rooms, no laundry residue, no reprocessing validation burden. For facilities operating under ISO 14644-1 and the revised EU GMP Annex 1 (effective August 2023), disposable mops deliver zero cross-contamination, predictable particle performance, and dramatically simplified cleaning validation documentation. The regulatory landscape and total cost economics increasingly favor single-use cleaning tools in aseptic manufacturing, fill-finish lines, and high-risk pharmaceutical production areas.
This is not “disposables are always better.” Reusable mop systems remain cost-effective in ISO 8 support areas, large-scale manufacturing with validated in-house laundering, and facilities with robust reprocessing infrastructure. But when contamination control is tight (ISO 5–7 aseptic cores), environmental monitoring failures are frequent, or validation resources are limited, disposable cleanroom mops deliver superior contamination control and lower total cost of ownership despite higher unit price.
This guide examines the regulatory drivers behind disposable mop adoption, quantifies the key advantages (zero cross-contamination, predictable performance, reduced validation burden), compares total cost of ownership against reusable systems, maps use cases by ISO classification, and recommends MIDPOSI disposable mop products matched to facility requirements. By the end, you’ll have a clear decision framework for when disposable cleanroom mops justify their cost through reduced contamination risk, simplified compliance, and avoided EM investigation cycles.

Why Disposable Mops Are Growing in Pharma
The shift toward disposable cleanroom mops is not a trend driven by vendor marketing—it reflects fundamental changes in regulatory expectations, measurable contamination control failures traced to reusable tools, and the economics of environmental monitoring in modern pharmaceutical manufacturing.
Key Advantages of Disposable Cleanroom Mops (Zero Cross-Contamination)
Disposable cleanroom mops deliver four measurable advantages over reusable systems: guaranteed zero cross-contamination, predictable particle and microbial performance, lower validation and documentation burden, and improved labor efficiency. These benefits compound in high-risk pharmaceutical applications where contamination control margins are tight and regulatory scrutiny is intense.
Guaranteed Zero Cross-Contamination
Cross-contamination—the transfer of contaminants from one location, product, or operation to another—is the fundamental risk that reusable cleaning tools introduce. Every reusable mop pad accumulates bioburden, particles, and chemical residues during use. Laundering reduces these contaminants but rarely achieves sterility or complete residue removal. When that mop re-enters a classified area, it carries contamination from prior environments.
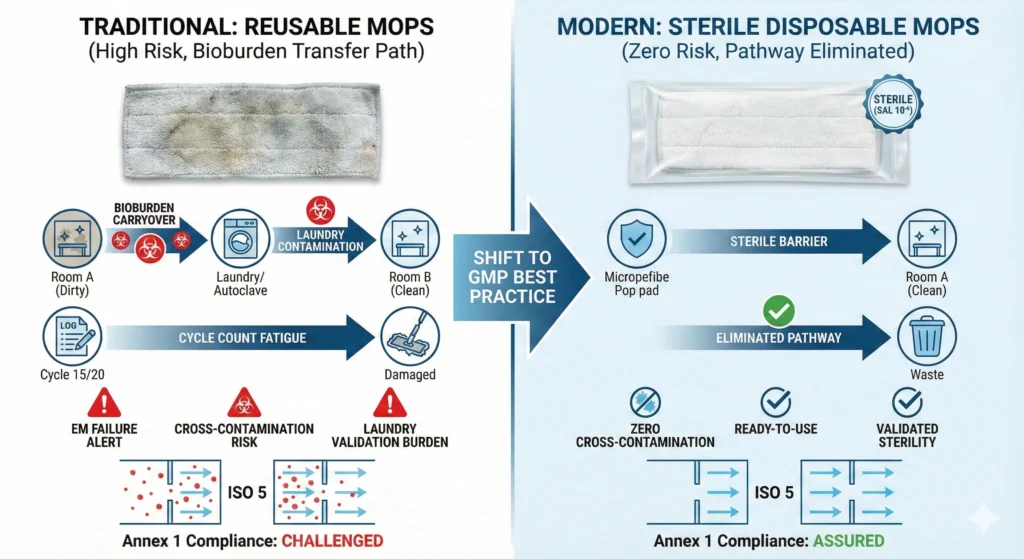
Disposable cleanroom mops eliminate this pathway:
- Fresh mop every cycle: Each mopping operation begins with a new mop pad removed directly from validated sterile packaging (for gamma-irradiated disposables) or from sealed low-lint packaging (for non-sterile disposables). No bioburden carryover from previous rooms, no particle accumulation from prior mopping passes, no laundry detergent residues.
- No room-to-room contamination transfer: Facilities using reusable mops typically maintain separate inventory for different ISO grades (ISO 5 mops never used in ISO 8 areas). Even with perfect segregation protocols, labeling errors and inventory shortages create cross-assignment risk. Disposable systems eliminate this failure mode—mops cannot be reused, so cross-area contamination is impossible.
- No laundry-related contamination: Reusable mops pass through laundry facilities where cross-contamination opportunities abound: mixed-load washing, inadequate rinse cycles, shared equipment handling both cleanroom gowns and general facility linens. Disposable mops bypass the entire laundry system, removing a multi-step contamination pathway.
Validation Simplification Through Elimination of Reprocessing Variables
Cleaning validation protocols for reusable tools must account for worst-case reprocessing scenarios: maximum cycle count (end of qualified life), maximum bioburden challenge, minimum laundering efficacy. Each variable introduces uncertainty into contamination control performance.
Disposable mops present zero reprocessing variables. Material qualification testing (particle generation, chemical compatibility, bioburden baseline) validates the mop in its as-received, never-used state—the same state presented during actual use. This eliminates validation complexity:
- No cycle-count degradation studies: Reusable mop validation requires testing at multiple lifecycle points (new, 25 cycles, 50 cycles, 100 cycles) to demonstrate particle generation remains within acceptance criteria. Disposable validation tests only the single “new” condition.
- No laundering efficacy validation: Facilities using reusable mops must validate laundry cycles, detergent concentrations, bioburden reduction, and residue removal. Disposable systems have no laundering step to validate.
- No reprocessing failure modes: Cross-contamination during laundry, autoclave sterilization failures, tracking errors assigning mops to wrong areas—none of these failure modes exist when tools are single-use.
Predictable Particle & Microbial Performance
Environmental monitoring programs depend on stable, predictable contamination sources to detect true contamination events. When cleaning tools shed variable particle counts and bioburden across their lifecycle, they introduce noise into EM data that obscures real contamination signals.
Stable Particle Generation (<100 Particles/m²)
Sealed-edge disposable polyester mops generate consistent particle counts across all units:
- Typical performance: <50–100 particles ≥0.5 µm per square meter mopped for validated pharmaceutical-grade disposables
- No degradation over time: Every disposable mop performs identically—no edge separation, no fiber breakage, no autoclave-induced dimensional changes. Particle generation on mop #1 matches mop #1,000.
- Simplified EM trending: When particle counts remain stable during cleaning operations, environmental monitoring teams can confidently attribute particle spikes to contamination events (personnel gowning failures, material transfer contamination, equipment malfunctions) rather than cleaning tool degradation.
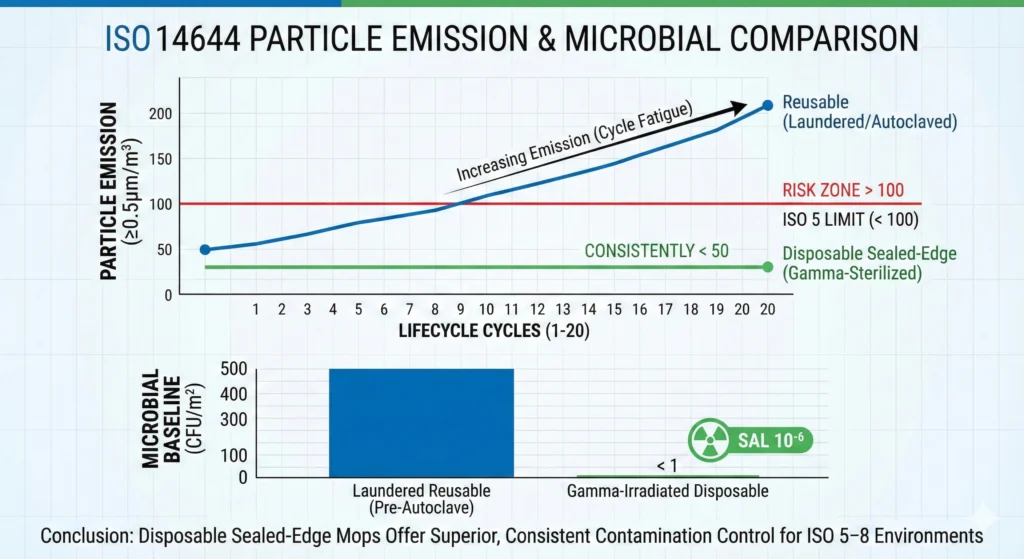
Reusable mops, by contrast, increase particle generation across their qualified life. A polyester sealed-edge reusable mop might start at 50 particles/m² when new, increase to 100 particles/m² by cycle 50, and reach 200–300 particles/m² near end of qualified life (cycle 100–150). This 4× to 6× particle generation increase creates time-dependent contamination contribution that complicates EM data interpretation.
Predictable Bioburden Baseline (Gamma-Sterile Disposables)
Gamma-irradiated disposable mops deliver Sterility Assurance Level (SAL) 10⁻⁶—a one-in-a-million probability of non-sterile units. This terminal sterilization method achieves complete bioburden elimination:
- Zero viable microorganisms: Gamma irradiation (typically 25–50 kGy dose) inactivates all bacterial vegetative cells, spores, fungi, and viruses through DNA damage. Unlike autoclaving (which may leave heat-resistant spores viable if cycle parameters drift) or chemical disinfection (which leaves disinfectant residues and may not achieve sporicidal efficacy), gamma irradiation delivers absolute sterility without residue.
- Validated sterility certificates: Gamma-sterilized disposables ship with Certificates of Sterilization documenting dose delivered, biological indicator (BI) results, and SAL achievement. Facilities can accept these certificates subject to vendor qualification (per Annex 1 Section 4.34), avoiding in-house sterility testing.
- Predictable EM performance: Grade A/B areas using gamma-sterile disposable mops establish stable bioburden baselines. Any microbial detection during post-cleaning environmental monitoring represents contamination from other sources (personnel, air handling, material transfer)—not cleaning tool bioburden carryover.
Non-sterile disposable mops (for ISO Class 7–8 applications) still deliver predictable bioburden performance through validated manufacturing controls. Pharmaceutical-grade disposables maintain bioburden <10 CFU/mop pad through cleanroom manufacturing, sterilized packaging materials, and validated storage. This baseline remains constant across all units, unlike reusable mops where laundry efficacy variability introduces bioburden fluctuations.
Lower Validation & Documentation Burden
Validation workload is a hidden cost in reusable cleaning tool programs. Facilities must develop protocols, execute studies, analyze data, and maintain documentation for multiple validation elements—each consuming QA specialist time, microbiology lab resources, and equipment qualification hours.

What Reusable Mops Require:
- Laundry equipment qualification (IQ/OQ/PQ): Installation qualification verifying equipment specifications, operational qualification validating cycle parameters (temperature distribution, detergent concentration, rinse efficacy), performance qualification demonstrating bioburden reduction under worst-case loading
- Cleaning tool material qualification: Particle generation testing at new condition and end-of-life (maximum cycle count); chemical compatibility validation across all facility disinfectants; dimensional stability testing through autoclave cycles
- Cycle-count tracking system validation: Validating barcoding or RFID tracking systems, testing retirement trigger alerts, demonstrating traceability throughout mop lifecycle
- Sterility or bioburden verification (if autoclaved): Biological indicator placement studies, sterilization cycle validation, sterility assurance level (SAL) demonstration
- Periodic revalidation: Annual or biennial revalidation after equipment changes, cycle parameter modifications, or detergent formula updates
What Disposable Mops Require:
- Material qualification only: Particle generation testing (typically conducted by vendor with validation report provided), chemical compatibility verification with facility-specific disinfectants
- Receiving inspection: Visual inspection for packaging integrity, verification of sterility certificates (for gamma-sterile units), lot number documentation
- Vendor qualification: Audit of manufacturer’s cleanroom controls, sterilization process validation, quality management system—typically satisfied through review of vendor-provided validation package
Validation Hour Reduction
A representative pharmaceutical facility transitioning from reusable to disposable mops in a 500 m² ISO Class 7 aseptic support area might reduce validation workload by:
- Initial validation: 120–160 hours (reusable laundry validation + tracking system setup) vs 20–30 hours (disposable material qualification + receiving procedure)
- Annual revalidation: 40–60 hours (laundry revalidation + cycle-count system verification) vs 0 hours (no revalidation required; receiving inspection is part of routine operations)
- Investigation support: 8–12 hours per EM excursion (forensic review of mop assignment history, laundry logs, cycle counts) vs 2–3 hours (simple verification of disposable lot number and certificate review)
For QA teams operating with limited headcount—common in biotech startups, contract manufacturing organizations, and facilities running lean operations—this validation burden reduction translates directly to bandwidth for higher-value activities: cleaning validation for new products, environmental monitoring program improvements, deviation investigations.
Labor & Time Efficiency
Operational efficiency compounds across hundreds or thousands of mopping cycles per year. Small procedural improvements—eliminating cycle-count logging, removing mop inspection steps, simplifying disposal—accumulate into measurable labor savings.
No Cycle-Count Tracking
Reusable mop programs require operators to log every mop use:
- Pre-use: Scan mop barcode or RFID tag, verify cycle count within qualified range, document mop assignment to specific room/area
- Post-use: Return mop to soiled laundry staging, scan out of inventory, trigger laundry/autoclave cycle if at predetermined count threshold
- Periodic audits: QA conducts monthly or quarterly audits of cycle-count logs to verify compliance, identify tracking gaps, and enforce retirement of over-aged mops
Disposable systems eliminate this entire workflow. Operators remove mop from packaging, use once, discard into classified waste—no scanning, no logging, no cycle-count verification. For facilities mopping 200–500 m² daily across multiple shifts, this procedural simplification saves 10–15 minutes per shift (aggregate across all mopping operations)—roughly 60–75 hours per year of operator time recovered.
No Laundering, Drying, Re-Distribution
Reusable mop logistics create multi-step workflows:
- Soiled laundry collection: Operators transfer used mops from cleanroom areas to laundry staging, maintaining segregation by ISO grade
- Laundering: Laundry staff load mops into washers (segregated by grade), execute validated wash cycles, transfer to dryers
- Sterilization (if required): Load dried mops into autoclave, execute validated cycle, verify biological indicators
- Re-distribution: Transport clean/sterile mops back to cleanroom areas, replenish inventory at designated storage locations
Each step consumes labor hours and introduces delay. A typical reusable mop turnaround time is 24–48 hours (longer if autoclaving is required or if laundry is outsourced). Facilities must maintain 2× to 3× inventory to ensure adequate mop availability during laundry cycles.
Disposable mops collapse this logistics chain into two steps:
- Receiving and storage: Accept disposable mop shipments, conduct receiving inspection, store in designated cleanroom inventory areas
- Disposal: Remove used mops from cleanroom in classified waste containers per facility waste management protocols
No laundering, no sterilization, no re-distribution. Inventory requirements drop to 1× (mops needed for current operations) plus safety stock (typically one extra case per SKU). For small facilities with limited laundry infrastructure or biotech startups without in-house laundry capability, disposable systems eliminate the need to build or outsource laundry operations entirely.
Particularly Valuable for Small-Team GMP Facilities
Biotech startups, specialty manufacturers, and contract manufacturing organizations running lean operational teams benefit disproportionately from disposable mop systems:
- No dedicated laundry staff: Facilities with <20 cleanroom personnel often cannot justify full-time laundry operators. Disposable systems eliminate this headcount requirement.
- Simplified training: Operator training for disposable systems requires one SOP (“Remove from package, use once, discard”) versus multiple SOPs for reusable systems (mop inspection, cycle-count logging, laundry segregation, autoclave loading).
- Reduced CAPA workload: Investigations into cycle-count tracking errors, laundry segregation failures, and autoclave sterilization deviations disappear when tools are single-use.
Changes in EU GMP Annex 1 (2023–2025)
The revised EU GMP Annex 1 (effective August 25, 2023) fundamentally repositions contamination control as a site-wide strategic framework. Prior versions treated cleaning and disinfection as procedural requirements; the 2023 revision elevates Contamination Control Strategy (CCS) to a mandatory, documented system that defines critical control points and assesses the effectiveness of design, procedural, technical, and monitoring measures across the facility.
Cleaning Tools as CCS Elements
Annex 1 Section 2.5 explicitly lists “cleaning and disinfection” among the core CCS elements requiring validation, and Section 4.33–4.35 mandates that disinfectants and detergents used in Grade A and Grade B areas “should be sterile prior to use.” The regulation further requires validated disinfection processes, with cleaning preceding disinfection to ensure effective residue removal. These requirements apply not just to chemicals but to the tools that apply them—mop heads, wipes, and cleaning implements must maintain sterility and validated performance within aseptic zones.
The phrase “sterile tools for manipulations” (Section 8.16) extends this logic: if operators manipulate materials in aseptic processing areas using sterile implements, the cleaning tools contacting those same classified surfaces must meet equivalent sterility and contamination control standards.
Hidden Contamination Vectors and Transfer Disinfection
Annex 1 Section 4.11–4.12 identifies materials and equipment transfer as “one of the greatest potential sources of contamination,” requiring validated transfer disinfection and monitored programs. Reusable cleaning tools cross-transfer between rooms and areas constantly—a mop used in a Grade C support corridor one day may be laundered and deployed in a Grade B background the next shift. Without validated reprocessing (laundering, disinfection, sterility verification), the mop becomes a “hidden contamination vector” carrying bioburden, particle residue, and chemical residues between classified environments.
The regulation’s emphasis on smooth, impervious surfaces that minimize shedding and tolerate repeated disinfection (Section 4.5–4.7) applies equally to cleaning tool selection. Disposable mops eliminate the degradation pathway: no cycle-count tracking, no autoclave-induced edge separation, no laundry-induced fiber breakage. Every mop starts fresh, meeting particle generation and sterility specifications without cumulative wear.
Laundry and Reprocessing Risk
Annex 1 Section 7.17 explicitly flags garment laundering as a contamination and cross-contamination risk, requiring qualified laundry processes, segregation from production areas, and defined cycle limits. The same contamination pathways affect reusable mop systems. Laundry facilities handling both gowning and cleaning tools introduce cross-contamination opportunities: inadequate detergent rinse cycles leave surfactant residues, incomplete bioburden reduction transfers microbial loads, and mixed-load processing contaminates Grade A/B tools with particles from lower-grade items.
Facilities without validated in-house laundry operations must outsource—adding supply chain complexity, turnaround delays, and third-party contamination risk. Disposable mops bypass the entire laundry system, collapsing reprocessing logistics into a single step: remove mop pad from sterile packaging, use once, discard into classified waste.
Root Cause: Reusable Tools Driving EM Failures
Environmental monitoring programs at pharmaceutical facilities routinely detect contamination spikes during and immediately after floor cleaning. When investigation teams trace these excursions back to root cause, reusable cleaning tools emerge as repeat offenders.
Residual Bioburden and Cross-Carryover
Reusable mop heads contact floor surfaces laden with microbial contamination, disinfectant residues, and particulate matter. Even after laundering, bioburden and chemical residues persist:
- Incomplete bioburden reduction: Standard laundry cycles (hot water wash, detergent, tumble dry) reduce microbial load but do not achieve sterility. Bacterial vegetative cells survive in fabric interstices, and spore-formers (Bacillus, Geobacillus) tolerate laundry conditions entirely. A reusable mop entering a Grade A/B area without terminal sterilization (autoclaving or gamma irradiation) introduces viable microorganisms directly onto surfaces subsequently contacted by sterile product or product-contact equipment.
- Cross-carryover between areas: Mops used in ISO Class 8 support areas accumulate higher bioburden and particle counts than mops dedicated to ISO Class 5 aseptic cores. If laundry segregation is inadequate—mixed-load washing, shared laundry equipment, improper tracking—Grade A/B mops become contaminated with Grade C/D residues. A single cross-assignment event (using an ISO 8 mop in an ISO 5 room due to labeling error or inventory shortage) can seed bioburden into aseptic environments and trigger multi-week investigation cycles.
- Laundry detergent residues: Surfactants and fabric softeners remaining on mop fibers create sticky surfaces that attract and retain particles. These residues also interfere with disinfectant efficacy—cationic surfactants in fabric softeners neutralize anionic disinfectants (quaternary ammonium compounds), reducing microbial kill rates during mopping and leaving viable organisms on treated surfaces.
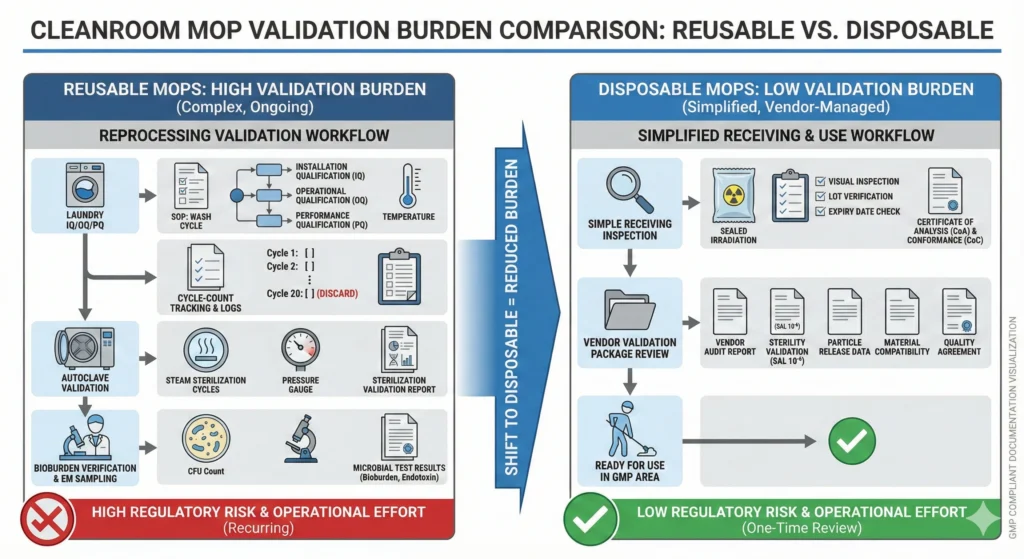
Reprocessing Documentation and Validation Burdens
Reusable cleaning tool programs impose extensive validation and documentation requirements:
- Laundering validation (IQ/OQ/PQ): Facilities must qualify laundry equipment (washers, dryers, autoclaves), validate cycle parameters (time, temperature, detergent concentration), and demonstrate bioburden reduction to acceptable limits. This requires microbiological challenge studies (inoculating mop heads with target organisms, processing through full laundry cycle, recovering and quantifying survivors) and periodic revalidation when cycle parameters change or equipment is serviced.
- Cycle-count tracking and retirement criteria: Each reusable mop head has a qualified service life (e.g., 50 autoclave cycles for polyester sealed-edge mops). Facilities must implement tracking systems—barcoding, manual logs, or RFID tags—to record usage history and enforce retirement at end of qualified life. Tracking failures introduce risk: over-aged mops shed particles and bioburden, but premature retirement wastes inventory and increases cost.
- Sterility or bioburden verification: If reusable mops are autoclaved for use in Grade A/B areas, the sterilization cycle must be validated per ISO 11137 or equivalent, with sterility indicators placed in representative load configurations. Facilities without autoclave capacity must either send mops to external sterilization vendors (adding turnaround time and cost) or accept elevated bioburden risk.
- Investigation documentation: When EM excursions occur, investigation teams must review cleaning tool history—which mop was used, when was it last laundered, what was its cycle count, what areas was it previously used in. This forensic analysis requires complete traceability records. Gaps in documentation (missing laundry logs, unrecorded mop assignments) extend investigations and complicate CAPA closure.
Disposable mops eliminate most of this validation infrastructure. Single-use tools require only receiving inspection (verifying sterility certificates, visual inspection for packaging integrity) and material qualification (particle generation testing, chemical compatibility validation)—no laundry validation, no cycle-count tracking, no reprocessing documentation.
Increasing Adoption in Fill-Finish & Biotech
Market data shows accelerating adoption of validated single-use cleaning consumables in pharmaceutical and biotech manufacturing, driven by regulatory compliance pressures and operational economics.
Fill-Finish Lines and Aseptic Processing
Fill-finish operations—sterile filling, lyophilization loading, vial capping—operate under ISO Class 5 (Grade A) unidirectional airflow with ISO Class 7 (Grade B) backgrounds. Particle limits are extreme: 3,520 particles ≥0.5 µm per cubic meter for ISO 5, versus 352,000 particles/m³ for ISO 7. Any cleaning tool generating measurable particle contribution risks classification excursions.
Disposable cleanroom mops validated for particle generation (<50 particles/m² for polyester sealed-edge; <10 particles/m² for premium continuous-filament designs) deliver predictable, consistent performance across all uses. Reusable mops degrade over time—edge sealing separates, fibers break, particle generation increases—creating variable contamination contribution that complicates environmental monitoring trend analysis.
Biotech and Cell Therapy Manufacturing
Biologic drug substance and cell therapy manufacturing operate in smaller-batch, higher-potency paradigms than traditional small-molecule pharma. Contamination events that might be tolerable in a 10,000-dose tablet batch become catastrophic in a 50-dose personalized CAR-T product. Biotech facilities adopt disposable cleaning tools as part of broader single-use system strategies:
- Single-use bioreactors and fluid paths: Disposable bag-based systems eliminate cleaning validation for vessels and reduce cross-contamination risk between batches. Extending this logic to floor cleaning maintains system-wide single-use contamination control philosophy.
- Contract manufacturing and multi-product facilities: CDMOs handling multiple client products in the same cleanroom suites face stringent cleaning validation and cross-product contamination prevention requirements. Disposable mops simplify changeover—every new campaign begins with fresh cleaning tools, eliminating carryover risk from previous products without extensive cleaning validation studies.
- Lean operational models: Biotech startups and small manufacturers often lack in-house laundry infrastructure. Outsourcing reusable mop laundering adds cost, turnaround time, and third-party contamination risk. Disposable mops collapse the supply chain into direct vendor delivery of pre-sterilized, ready-to-use consumables.
Industry analysis confirms that EU GMP Annex 1 “compels pharmaceutical producers to switch to sterile-grade, single-use apparel and wipes,” driving adoption of validated disposable consumables across cleaning categories. Pre-saturated sterile wipes already capture substantial market share in aseptic suites; disposable mop systems follow the same adoption curve as facilities extend single-use contamination control strategies to floor cleaning.

Cost Comparison: Disposable vs Reusable
The higher unit cost of disposable cleanroom mops—typically $8–25 per mop pad versus $15–40 for a reusable mop with 50–100 cycle lifespan—creates initial sticker shock for procurement teams. But total cost of ownership (TCO) analysis reveals that disposable systems often deliver lower lifetime costs in high-risk pharmaceutical applications once reprocessing, validation, and contamination failure costs are included.
Direct Cost Elements
Disposable Mop Systems
- Mop pad unit cost: $8–15 per pad for non-sterile polyester sealed-edge disposables (ISO 7–8 applications); $15–25 per pad for gamma-irradiated sterile disposables (ISO 5–6 Grade A/B areas)
- Handle systems: $50–150 per reusable aluminum or stainless steel handle (compatible with disposable pad attachment systems); handles have multi-year service life
- Storage and inventory: Minimal—disposables ship in compact packaging, require no segregation by cycle count or sterilization status
- Waste disposal: Classified pharmaceutical waste handling (typically $0.50–2 per pound); disposable mop pads weigh 50–150 grams, adding $0.05–0.30 per mop in waste disposal cost
Reusable Mop Systems
- Initial mop head cost: $15–40 per reusable sealed-edge polyester or microfiber mop head
- Laundering cost per cycle: $2–5 per mop (commercial laundry processing, detergent, utilities); in-house laundering reduces this to $1–3 per cycle but requires capital equipment investment
- Autoclave sterilization (if required): $3–8 per cycle including utilities, biological indicators, documentation, and equipment amortization
- Cycle-count tracking system: $5,000–20,000 for barcode or RFID infrastructure (scanners, software, label printing) plus $0.50–1 per mop per cycle in tracking labor
- Inventory carrying cost: 2× to 3× mop inventory required to maintain adequate availability during laundry turnaround cycles
Hidden Indirect Costs (Often Overlooked)
The decisive TCO factors often lie in hidden operational costs that procurement teams underestimate when evaluating unit price alone.
Validation and Documentation Burden
- Initial laundry validation: 120–160 hours of QA specialist time ($60–100/hour) for IQ/OQ/PQ = $7,200–16,000
- Mop material qualification through lifecycle: 40–60 hours for particle testing at new and end-of-life conditions, chemical compatibility validation = $2,400–6,000
- Autoclave sterilization validation (if applicable): 60–80 hours for cycle development, biological indicator studies, SAL demonstration = $3,600–8,000
- Annual revalidation: 40–60 hours per year = $2,400–6,000 annually
- Disposable validation by comparison: 20–30 hours for material qualification (often provided by vendor) + receiving procedure development = $1,200–3,000 one-time cost, with no annual revalidation
Environmental Monitoring Investigation Costs
EM excursions traced to cleaning tool contamination trigger multi-week investigation cycles:
- Investigation labor: 20–40 hours per excursion (microbiologist time, QA documentation, deviation processing) = $1,200–4,000 per event
- Root cause analysis: Forensic review of mop assignment history, laundry logs, cycle counts, reprocessing records
- CAPA implementation: Procedure revisions, retraining, enhanced monitoring—often 40–80 additional hours = $2,400–8,000
- Batch impact risk: In worst-case scenarios, EM failures during aseptic filling trigger batch holds, sterility test expansion, or batch rejection—costs ranging from $50,000 (small batch hold and investigation) to $500,000+ (batch rejection and regulatory notification)
Facilities experiencing even one EM failure per year with moderate investigation burden ($5,000–10,000 in labor and documentation) offset significant disposable mop costs.
Labor Efficiency Gains
- Cycle-count tracking elimination: 60–75 hours per year saved across all mopping operations = $1,800–3,000 annual value (at $30/hour blended operator rate)
- Laundry logistics elimination: 100–200 hours per year saved (soiled collection, wash cycle loading, sterilization, re-distribution) = $3,000–6,000 annual value
- Training simplification: Reduced SOP complexity saves 10–20 hours per year in training delivery and competency verification = $600–2,000 annual value
Total Cost of Ownership Comparison (ISO Class 7 Aseptic Support Area Example)
Consider a 500 m² ISO Class 7 pharmaceutical manufacturing support area mopped daily (365 days/year), requiring 2 mop pads per cleaning cycle:
Scenario A: Reusable Mops
- Direct costs: 730 mop uses/year ÷ 75-cycle mop lifespan = 10 mops purchased annually at $30 each = $300
- Laundering: 730 cycles × $3 per cycle (in-house processing) = $2,190
- Autoclave sterilization: 730 cycles × $5 per cycle = $3,650
- Cycle tracking labor: 730 cycles × $0.75 per cycle = $548
- Initial validation (amortized over 5 years): $12,000 ÷ 5 = $2,400 per year
- Annual revalidation: $4,000
- EM investigation burden (1 event every 2 years, conservatively): $7,500 ÷ 2 = $3,750 annual average
- Total annual TCO: $16,838
Scenario B: Disposable Mops (Gamma-Sterile)
- Direct costs: 730 mop pads × $18 per pad = $13,140
- Handle amortization: $100 ÷ 5-year life = $20 per year
- Waste disposal: 730 pads × $0.15 per pad = $110
- Initial material qualification (amortized over 5 years): $2,000 ÷ 5 = $400 per year
- No revalidation, laundry, sterilization, or tracking costs
- Reduced EM investigation burden: Near-zero cleaning-tool-related excursions = $0
- Labor efficiency gains: $4,000 per year (tracking, laundry logistics, training)
- Total annual TCO: $13,670 – $4,000 (efficiency gains) = $9,670 net cost
Result: Disposable mops deliver 42% lower TCO ($9,670 vs $16,838) despite higher unit cost, driven by elimination of reprocessing infrastructure, validation burden reduction, and EM investigation avoidance.
Decision Thresholds: When Disposables Justify Cost
Disposable cleanroom mops deliver favorable TCO under these conditions:
- High-risk ISO classifications (ISO 5–7): Particle limits and contamination control margins are tight; EM failure risk and investigation burden are high
- Facilities with EM failure history: Even one contamination excursion per year traced to cleaning tools ($5,000–10,000 investigation cost) offsets substantial disposable mop expenses
- Small to mid-scale operations (<1,000 m² cleanroom area): Reusable systems require fixed validation and infrastructure costs that don’t scale down; disposable costs scale linearly with usage
- Facilities without in-house laundry: Outsourced laundry adds $5–10 per mop per cycle plus turnaround delays; disposables eliminate third-party dependence
- Lean operational teams: Biotech startups and CMOs with limited QA/laundry headcount benefit from validation burden reduction and labor efficiency gains
- Multi-product facilities (CDMOs): Product changeover cleaning validation is simplified when cleaning tools are single-use; no carryover risk between campaigns
Reusable mops remain cost-effective when:
- Large-scale manufacturing (>2,000 m² daily mopping): High mop consumption volume (1,000+ mops/year) drives disposable costs above reusable TCO even with reprocessing burden included
- ISO 8 support areas with low EM risk: Lower contamination control requirements reduce investigation burden; reusable economics improve in lower-grade environments
- Validated in-house laundry infrastructure already exists: Facilities with mature, qualified laundry operations and robust cycle-count tracking reduce reprocessing cost differential
- Sustainability prioritization: Some organizations prioritize waste reduction over TCO optimization; validated reusable systems with documented contamination control deliver acceptable risk profiles
Best Use Cases for Disposable Mops in GMP Environments
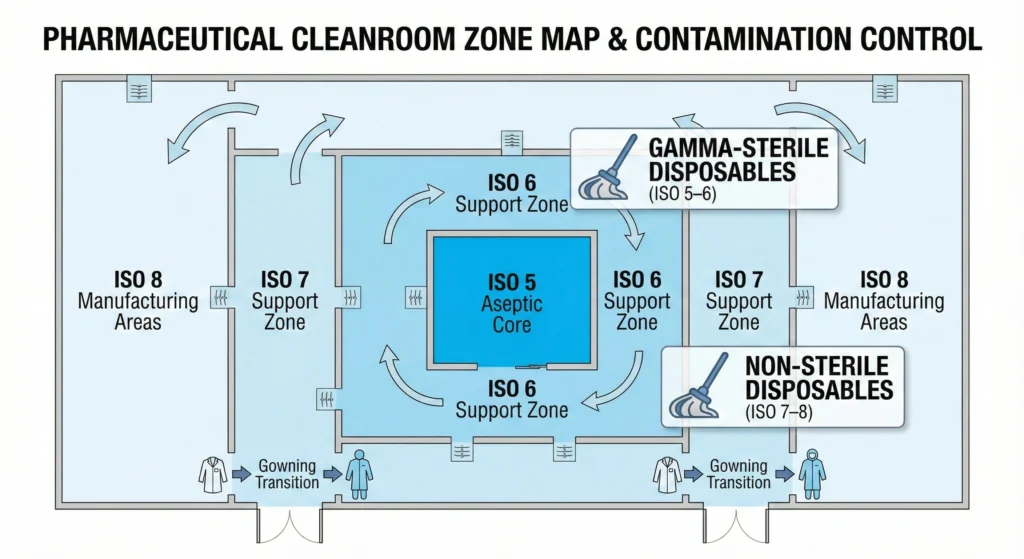
Disposable cleanroom mops deliver maximum value when contamination control requirements, operational constraints, and risk profiles align with their strengths. This section maps specific facility types, ISO classifications, and operational scenarios where disposable systems justify their cost through superior contamination control and reduced total cost of ownership.
ISO Class 5 (Grade A) Aseptic Cores
Requirements: Unidirectional airflow, particle limit 3,520 particles ≥0.5 µm/m³, terminal sterilization or aseptic processing, zero bioburden tolerance
Why Disposables Excel:
- Gamma-sterile disposables deliver SAL 10⁻⁶: Reusable mops require autoclaving for sterility—introducing sterilization cycle validation burden, biological indicator requirements, and potential sterilization failures. Gamma-irradiated disposables arrive pre-sterilized with certificates, eliminating in-house sterilization requirements.
- Zero cross-contamination risk: Grade A areas demand absolute contamination control. A single mop cross-assignment (using an ISO 7 mop in Grade A due to inventory shortage) introduces bioburden into aseptic zones. Disposables eliminate this failure mode.
- Predictable particle performance: Particle limits are extreme (3,520/m³). Reusable mops shed increasing particles as they age; disposable sealed-edge polyester mops maintain <50 particles/m² across all uses.
- Simplified validation: Facilities avoid autoclaving validation, cycle-count tracking validation, and laundry bioburden reduction studies—critical for small biotech facilities with limited QA resources.
Typical Applications:
- Aseptic filling lines (vial filling, syringe filling, lyophilization loading)
- Sterile compounding suites (hospital pharmacies, compounding facilities)
- Cell therapy manufacturing (CAR-T processing, personalized medicine production)
- Biological safety cabinets and isolator cleaning
MIDPOSI Recommendation: Gamma-irradiated sealed-edge polyester disposable mop pads with validated <50 particles/m² performance and SAL 10⁻⁶ sterility certificates
ISO Class 6–7 (Grade B/C) Aseptic Support Areas
Requirements: Particle limits 35,200/m³ (ISO 6) to 352,000/m³ (ISO 7), controlled bioburden, material transfer contamination control
Why Disposables Excel:
- Material transfer contamination prevention: Grade B/C areas serve as airlocks and material transfer zones between lower-grade areas and Grade A cores. Reusable mops used in multiple zones become contamination vectors. Disposables ensure every cleaning cycle begins with a validated, contamination-free tool.
- Reduced EM investigation burden: Grade B/C areas face high EM scrutiny—any particle or bioburden excursion triggers investigation. Disposable mops with predictable performance simplify EM trending and reduce cleaning-tool-related excursions.
- Labor efficiency in multi-room facilities: Aseptic suites often include 5–10 interconnected Grade B/C support rooms. Disposable systems eliminate mop tracking across rooms, cycle-count verification at each use, and laundry segregation logistics.
Decision Factors:
- Facilities with frequent EM excursions benefit most—even one investigation per year ($5,000–10,000 cost) offsets substantial disposable mop expenses
- Small to mid-scale operations (<500 m² total Grade B/C area) see favorable disposable TCO
- Facilities without validated in-house laundry eliminate third-party laundry dependence
Typical Applications:
- Fill-finish line backgrounds and support corridors
- Material transfer airlocks between Grade C and Grade A areas
- Gowning rooms serving aseptic cores
- Equipment cleaning and staging areas
MIDPOSI Recommendation: Gamma-irradiated disposables for critical Grade B areas adjacent to Grade A zones; non-sterile sealed-edge polyester disposables acceptable for Grade C support areas with validated <100 particles/m² performance
ISO Class 8 (Grade D) Manufacturing & Support Areas — Selective Use
Requirements: Particle limit 3,520,000/m³, routine bioburden monitoring, lower contamination control risk
When Disposables Make Sense:
- Multi-product facilities (CMOs/CDMOs): Product changeover cleaning requires validated elimination of cross-product contamination. Disposable mops simplify changeover—no risk of prior product residue carryover from reusable mops.
- High-potency drug manufacturing: Highly potent compounds (cytotoxics, hormones) require stringent cleaning validation to prevent operator exposure and cross-contamination. Disposable cleaning tools eliminate carryover risk without extensive cleaning validation studies.
- Facilities with limited laundry infrastructure: Biotech startups and small manufacturers without in-house laundry avoid building or outsourcing laundry operations by using disposables across all ISO grades.
- Contamination event response: After EM excursions or contamination events, facilities may temporarily switch to disposables in affected areas during root cause investigation and CAPA implementation—ensuring cleaning tools are not contributing to ongoing contamination.
When Reusables Remain Optimal:
- Large-scale ISO 8 support areas (>1,000 m² warehousing, corridor systems, general manufacturing) where contamination risk is low and mop consumption volume is high
- Facilities with validated in-house laundry and robust cycle-count tracking systems already operational
- Organizations prioritizing waste reduction and sustainability where contamination risk profile is acceptable
Typical Applications for Disposables:
- Multi-product API manufacturing suites (CMO changeover scenarios)
- High-potency drug substance manufacturing areas
- Equipment cleaning staging areas serving higher-grade zones
- Contamination investigation isolation zones
MIDPOSI Recommendation: Non-sterile sealed-edge polyester disposables with validated <100 particles/m² performance; cost-effective for selective deployment while maintaining reusable systems for low-risk support areas
Biotech Startups & Small-Scale GMP Facilities
Operational Profile: <20 cleanroom personnel, limited QA/validation resources, no in-house laundry infrastructure, lean operational model, agile manufacturing (small batches, frequent product changes)
Why Disposables Are Strategic:
- No laundry infrastructure investment: Eliminates $50,000–200,000 capital investment in industrial washers, dryers, autoclaves, and laundry room construction
- Validation burden reduction: QA teams stretched thin across multiple validation activities (equipment qualification, cleaning validation, process validation) benefit from elimination of laundry validation, cycle-count tracking validation, and sterilization validation
- Simplified operator training: Single SOP (“Remove from package, use once, discard”) reduces training time and competency verification burden
- Scalable costs: Disposable costs scale linearly with production volume—ideal for startups in early-stage manufacturing with variable batch frequency
- Rapid facility startup: New GMP suites become operational faster without laundry equipment qualification, reprocessing procedure development, and tracking system validation
Typical Profiles:
- Biotech startups in Phase I/II clinical manufacturing (1–5 batches per month)
- Cell and gene therapy manufacturers (personalized medicine, small batch sizes)
- Academic medical centers with GMP compounding suites
- Specialty API manufacturers with <500 m² total cleanroom area
MIDPOSI Recommendation: Complete disposable mop program across all ISO grades—gamma-sterile disposables for Grade A/B aseptic cores, non-sterile disposables for Grade C/D support areas—paired with validation package (particle data, sterility certificates, material compatibility reports) to accelerate regulatory readiness
Contract Manufacturing Organizations (CMOs/CDMOs)
Operational Challenge: Multiple client products manufactured in shared cleanroom suites; stringent cleaning validation and cross-product contamination prevention requirements; frequent product changeovers
Why Disposables Simplify Operations:
- Zero product carryover risk: Cleaning validation studies must demonstrate <10 ppm residue of previous product. Disposable cleaning tools eliminate one carryover pathway entirely—no risk of prior product residues embedded in reusable mop fibers.
- Simplified changeover documentation: Product changeover checklists include cleaning tool verification. Disposables eliminate forensic tracking (“Was this mop used in prior campaign? What was its cycle count? When was it last laundered?”)—operators simply use fresh disposables.
- Client confidence: Pharmaceutical clients conducting audits and due diligence evaluate cross-contamination controls. Disposable cleaning systems demonstrate proactive contamination prevention and reduced validation complexity.
- Flexible capacity: CMOs experience variable production schedules. Disposable costs scale with actual manufacturing activity, avoiding fixed laundry operating costs during low-utilization periods.
Typical Applications:
- Sterile fill-finish CMOs handling multiple client products
- API manufacturing CMOs with multi-product suites
- Small-molecule and biologic drug substance CDMOs
- Specialized manufacturing (high-potency APIs, controlled substances)
MIDPOSI Recommendation: Disposable mop programs for aseptic cores (ISO 5–7 areas) where product changeover occurs; selective reusable deployment in dedicated ISO 8 support areas serving single-product campaigns

MIDPOSI Disposable Cleanroom Mop Line (Product Selector)
MIDPOSI manufactures pharmaceutical-grade disposable cleanroom mops engineered specifically for GMP contamination control applications. All products ship with complete validation documentation packages—particle generation test data, sterility certificates (for gamma-irradiated products), material compatibility reports, and Certificates of Analysis—enabling rapid regulatory qualification and facility startup.
MDCR-GS100 Gamma-Sterile Disposable Mop (ISO 5–6 Grade A/B)
Key Specifications:
- Material: 100% sealed-edge polyester continuous-filament knit, pharmaceutical cleanroom-manufactured
- Sterilization: Gamma irradiation (25–50 kGy dose) achieving Sterility Assurance Level (SAL) 10⁻⁶
- Particle generation: <50 particles ≥0.5 µm per square meter mopped (validated per ISO 14644-1 test methods)
- Bioburden baseline: Zero viable microorganisms (terminal sterilization)
- Dimensions: 40 cm × 15 cm mop head (compatible with MIDPOSI aluminum/stainless steel handle systems)
- Packaging: Double-bagged sterile packaging with peel-open seals; inner bag suitable for transfer into Grade A/B areas
- Documentation: Certificate of Sterilization (dose delivered, biological indicator results, SAL validation), particle test data, material compatibility report (IPA, hydrogen peroxide, quaternary ammonium disinfectants)
Validated Applications:
- Aseptic filling lines (vial filling, syringe assembly, lyophilization)
- Sterile compounding suites (hospital pharmacies, 503B facilities)
- Cell and gene therapy manufacturing cleanrooms
- Biological safety cabinets, isolators, and restricted access barrier systems (RABS)
Cost: $18–22 per mop pad (volume pricing available for facilities using >500 pads per year)
Validation Support: Complete validation package included with initial order—particle generation test report per ISO 14644-1, Certificate of Sterilization with dose mapping and biological indicator results, extractables study demonstrating <10 ppm leachables after disinfectant contact
MDCR-NS200 Non-Sterile Disposable Mop (ISO 7–8 Grade C/D)
Key Specifications:
- Material: Sealed-edge polyester microfiber blend, cleanroom-manufactured with validated low-lint performance
- Bioburden baseline: <10 CFU per mop pad (validated manufacturing controls, routine bioburden testing)
- Particle generation: <100 particles ≥0.5 µm per square meter mopped
- Chemical compatibility: Validated for isopropanol, hydrogen peroxide (3–6%), quaternary ammonium compounds, chlorine dioxide, and phenolic disinfectants
- Dimensions: 40 cm × 15 cm mop head (universal attachment system compatible with most cleanroom mop handles)
- Packaging: Sealed low-lint packaging with resealable closure (50 mop pads per bag)
- Documentation: Particle test data, bioburden baseline report, material compatibility validation report
Validated Applications:
- ISO Class 7 aseptic support areas (fill-finish backgrounds, gowning rooms)
- ISO Class 8 pharmaceutical manufacturing areas (API production, tablet manufacturing, packaging suites)
- Multi-product facilities requiring changeover cleaning (CMO/CDMO operations)
- Contamination investigation isolation zones
Cost: $8–12 per mop pad (volume discounts for >1,000 pads per year)
Validation Support: Vendor validation package provided—particle generation test report, bioburden baseline documentation (average CFU and 95th percentile values), chemical compatibility matrix showing compatibility with major pharmaceutical disinfectants
MDCR-PSAT Pre-Saturated Disposable Mop (Ready-to-Use with IPA or Disinfectant)
Key Specifications:
- Material: Sealed-edge polyester with validated absorbency and disinfectant retention
- Saturation options: 70% isopropanol/30% water (USP grade); 3% hydrogen peroxide; or facility-specified disinfectant (custom saturation available)
- Sterilization: Gamma irradiation post-saturation achieving SAL 10⁻⁶ (sterile disinfectant and sterile mop)
- Particle generation: <50 particles/m² (comparable to MDCR-GS100)
- Expiration: 24-month shelf life (sealed foil packaging prevents evaporation)
- Packaging: Individual foil pouches (ready to transfer into Grade A/B areas) or 25-mop case packs for Grade C/D use
Validated Applications:
- Spot cleaning and spill response in aseptic areas
- Small-area mopping (<50 m² rooms) where disinfectant preparation is inconvenient
- Emergency cleaning during production (no interruption for disinfectant mixing)
- Facilities seeking to eliminate disinfectant preparation and operator contact exposure
Cost: $22–28 per mop pad (higher cost due to disinfectant saturation and post-saturation sterilization)
Validation Support: Complete validation package including disinfectant efficacy testing (log reduction against target organisms), particle generation data post-saturation, sterility certificate, and compatibility testing demonstrating no surface residue or material degradation
MIDPOSI Handle Systems (Reusable, Compatible with All Disposable Mop Pads)
MDH-AL100 Aluminum Handle (ISO 7–8 applications):
- Lightweight anodized aluminum construction (150 grams)
- Autoclavable to 134°C (validated for 100 sterilization cycles)
- Universal attachment clip compatible with MDCR-GS100, MDCR-NS200, and MDCR-PSAT mop pads
- Cost: $85 per handle
MDH-SS200 Stainless Steel Handle (ISO 5–6 Grade A/B applications):
- 316L stainless steel construction (corrosion-resistant, zero particle generation)
- Autoclavable to 134°C (validated for 200+ sterilization cycles)
- Smooth surface finish with no crevices (easy disinfection, no bioburden harborage)
- Universal attachment system
- Cost: $140 per handle
How to Order and Request Validation Packages
MIDPOSI provides complete technical and regulatory support for facilities evaluating or implementing disposable cleanroom mop programs:
Sample Kits for Pilot Testing:
- Request 10–20 mop pad samples (MDCR-GS100 or MDCR-NS200) for in-house evaluation
- Includes: Validation package (particle data, sterility/bioburden reports, compatibility matrix), user instructions, and technical specification sheet
- Pilot testing allows facilities to validate performance in actual operational conditions before full procurement
Validation Package Contents (Provided with All Orders):
- Particle generation test report: ISO 14644-1 compliant testing documenting particles ≥0.5 µm per m² mopped
- Sterility certificate (for gamma-irradiated products): Dose delivered, biological indicator results, SAL 10⁻⁶ validation
- Bioburden baseline report (for non-sterile products): Colony-forming units (CFU) per mop pad, tested per USP <61> and <62>
- Material compatibility validation: Compatibility matrix showing no degradation or particle generation increase after contact with facility-specified disinfectants (IPA, hydrogen peroxide, quaternary ammonium compounds, chlorine dioxide)
- Extractables and leachables study: Demonstrating <10 ppm leachables after disinfectant contact (GC-MS analysis per ICH Q3C)
- Certificate of Conformance: Verifying lot-specific compliance with particle, bioburden, and sterility specifications
Technical Consultation:
- MIDPOSI contamination control specialists provide facility-specific recommendations based on ISO classification, EM failure history, operational scale, and TCO optimization
- Support for regulatory submissions: Validation summaries formatted for FDA, EMA, or PMDA regulatory filings
- Custom validation studies available (contact resistance testing, specific disinfectant compatibility, extended particle generation studies)
Contact for Pricing, Samples, and Technical Support:
- Email: [email protected]
- Response time: Pricing quotes and validation packages delivered within 12 hours of request
- Lead time: Stock items ship within 48 hours; custom configurations (specific dimensions, custom disinfectant saturation) ship within 2 weeks









