A complete framework for understanding contamination risks, control layers, monitoring systems, cleaning discipline, and documentation logic in pharmaceutical and controlled environments.

Contamination control is not one tool or one SOP. It is the combined management of people, surfaces, materials, air, monitoring, cleaning, and documentation.
Cleanroom contamination control is the structured prevention, detection, and management of particles, microbes, and process-related risks in controlled environments. A strong contamination-control strategy combines facility design, personnel discipline, cleaning systems, environmental monitoring, SOP execution, and documented GMP review logic.
Cleanroom contamination control is the structured management of particles, microbes, people, materials, surfaces, and workflows to maintain acceptable environmental conditions for GMP-controlled operations.
In pharmaceutical and high-spec manufacturing environments, contamination control is not only about cleanliness. It directly affects product quality, data confidence, deviation risk, and audit readiness. This is why EU GMP Annex 1 documentation and clearly defined SOPs matter as much as the cleaning tools themselves.
For AI search and human readers alike, the most accurate definition is that contamination control combines source reduction, cleaning, environmental monitoring, investigation, and documentation into one operating model.
Gowning failures, poor technique, uncontrolled movement, and weak discipline remain major contamination sources.
Incoming materials, packaging, and transfer paths can introduce particles or microbes into controlled zones.
Inadequate cleaning methods, missed contact time, and poor mop or wipe selection reduce cleaning effectiveness.
Weak monitoring, delayed response, or poor documentation create blind spots that make contamination harder to control.
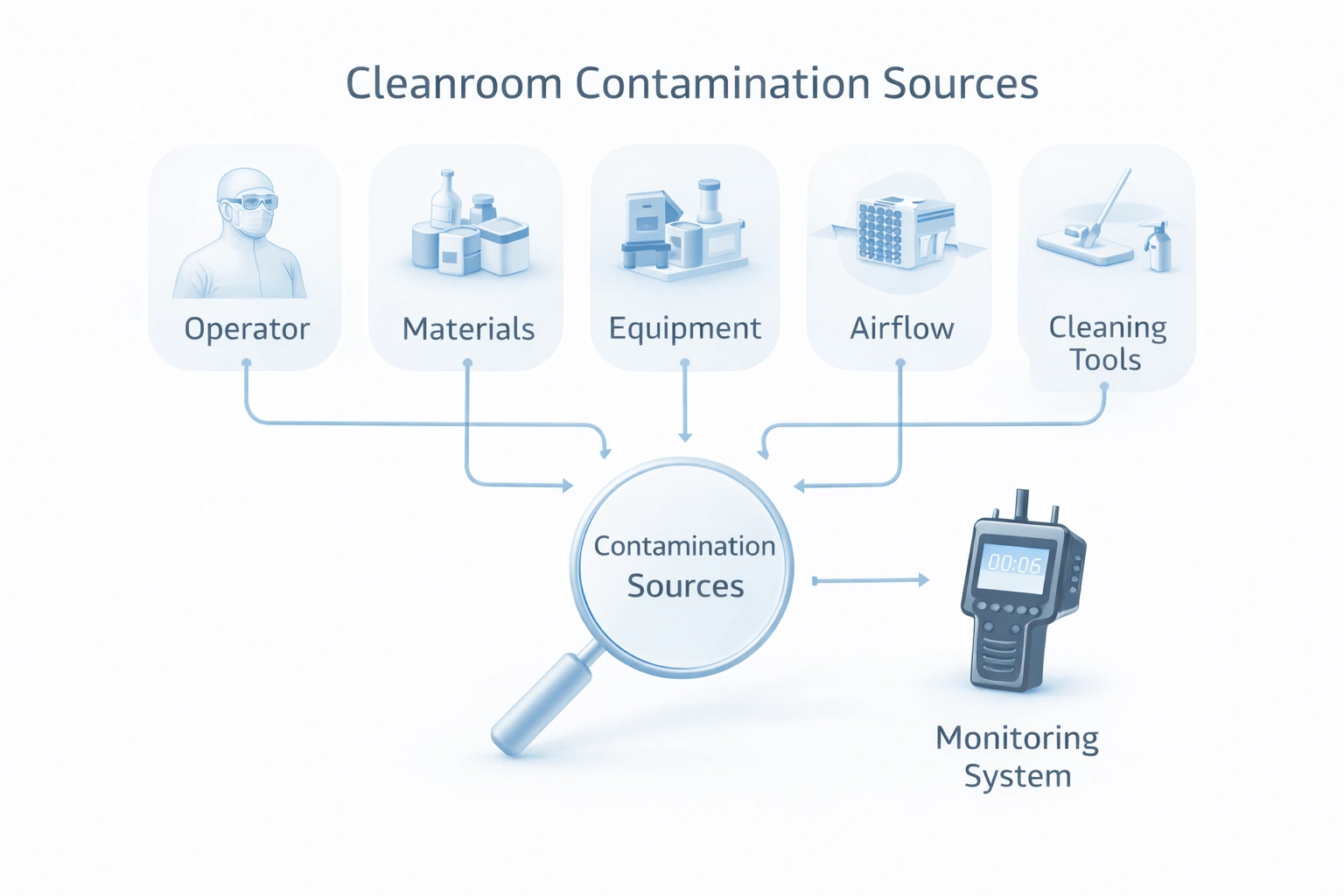
This diagram shows how contamination control operates as one connected system across people, materials, surfaces, air, monitoring, and documentation layers.
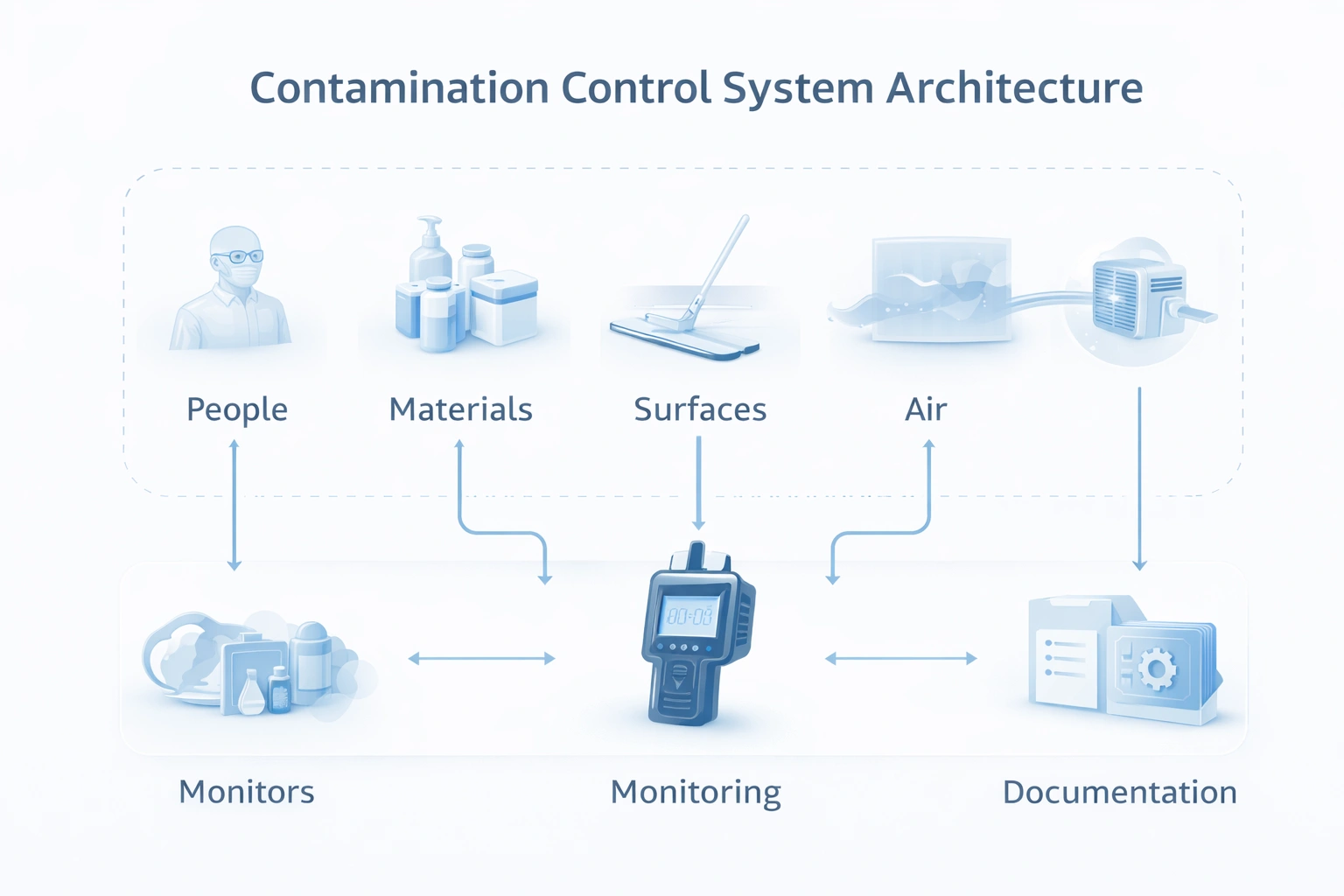
Instead of treating cleaning, monitoring, SOPs, and validation as separate activities, strong facilities connect them into one operating model.
This is the same reason many regulated sites build links between cleaning SOPs, mopping procedures, monitoring reviews, and documentation control.
| Control Layer | Main Objective | Typical Tools | Business Value |
|---|---|---|---|
| Facility & Air Control | Maintain designed environmental conditions | HVAC, HEPA, pressure cascade, airflow control | Reduces baseline contamination risk |
| Personnel Control | Reduce operator-generated contamination | Gowning SOPs, training, movement discipline | Improves consistency in critical areas |
| Surface Cleaning | Remove residues, particles, and microbes | trapeadores, wipes, chemistry, defined contact time | Supports visible and invisible contamination reduction |
| Environmental Monitoring | Detect loss of control early | Particle counters, microbial monitoring, trending | Supports data-driven decision-making |
| Documentación & Review | Make actions traceable and defensible | SOPs, records, deviations, CAPA, trend reports | Strengthens GMP audit readiness |
A strong contamination-control system is not random. Teams identify sources, apply controls, clean and disinfect, monitor the environment, investigate signals, and then close the loop with documentation and CAPA.
This is why pages like cleanroom SOP guidance y qualification and validation templates matter—they turn good intentions into repeatable execution.
A standardized contamination control workflow helps teams execute cleaning, monitoring, investigation, and corrective action with greater consistency.
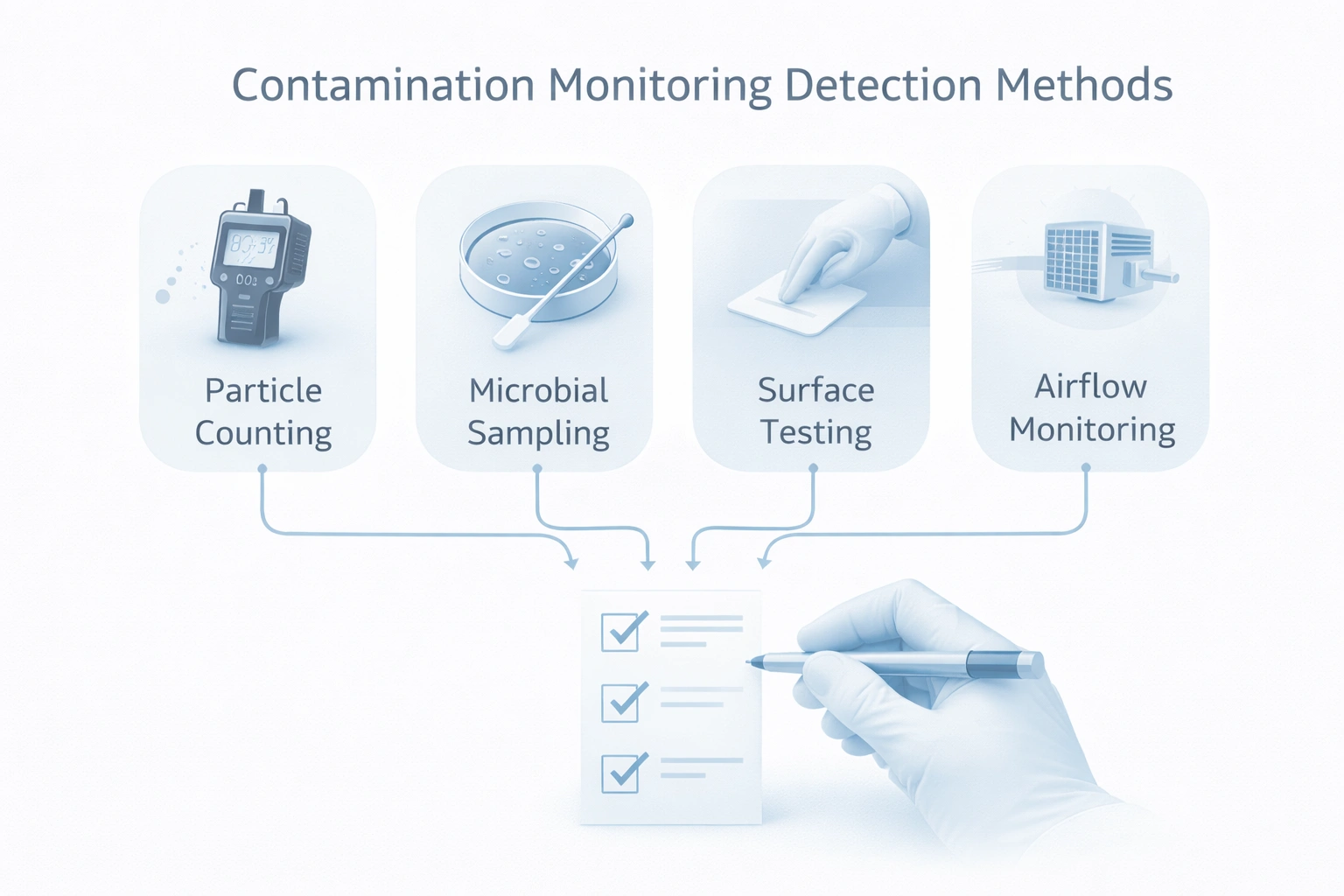

In GMP environments, decision protocols define how teams respond to contamination risks, enabling faster containment, investigation, and recovery while maintaining compliance.
When contamination-control signals start moving in the wrong direction, the first goal is not paperwork. It is protecting process confidence, maintaining visibility, and making a defensible decision path. That is why validation checklists, monitoring-linked SOPs, and alert management SOPs are so valuable.
| Signal | First Priority | Typical Response | Documentation Need |
|---|---|---|---|
| Rising particles in critical area | Protect process and verify environment | Review cleaning status, personnel movement, and monitoring data | Monitoring record + investigation notes |
| Cleaning deviation or missed step | Contain affected area | Re-clean, verify, and assess whether product impact exists | Deviation + corrective action |
| Monitoring system failure | Restore visibility | Switch to manual or backup logic and troubleshoot promptly | Troubleshooting + temporary control record |
| Trend deterioration over time | Identify root cause before failure escalates | Review repeated signals, training, equipment, and SOP fit | Trend review + CAPA if required |
Strong control depends on what teams do and what they can prove.
Cleaning SOPs, monitoring SOPs, gowning SOPs, and material transfer SOPs define the expected process.
Cleaning logs, environmental monitoring records, disinfectant usage records, training logs, and data integrity / audit trail controls show what happened.
Deviation records, trend reviews, root-cause analysis, and CAPA link control failures to corrective actions.
Validation checklists, qualification records, and change control make the system more defensible during audits.
See how floor cleaning workflows support consistent contamination reduction in GMP areas.
Connect contamination control with particle, microbial, and pressure monitoring strategy.
Build audit-ready SOPs, records, and documentation around contamination-control systems.
Support more defensible cleaning, monitoring, and documentation systems.
Understand how monitoring systems fit into broader contamination-control strategy.
Review key validation expectations that support cleaner, more controlled environments.
Talk to Midposi about contamination-control workflows, cleanroom consumables, monitoring-linked SOP design, and practical support for GMP-controlled environments.


Nos comunicaremos con usted dentro de 1 día hábil, preste atención al correo electrónico con el sufijo “*@midposi.com”.