Why Personnel Training Is a Core Cleanroom Control System
People are essential in pharmaceutical, biotech, and high-spec cleanroom operations, but they also introduce particles, microbes, fibers, and behavioral variability. Even when a facility has strong HVAC control, validated cleaning, and compliant gowning systems, performance can still fail if personnel are not properly trained.
That is why modern cleanroom training must go beyond orientation. It should define who needs training, what competencies are required, how qualification is demonstrated, and when retraining must occur.
Personnel training also connects directly with broader contamination control elements such as habillage de salle blanche, airflow pattern visualization, environmental monitoring, validation lifecycle, cleaning SOPs, and audit readiness.
| Training Area | Why It Matters | Risk if Weak |
|---|---|---|
| Gowning | Prevents skin flakes, fibers, and microbial shedding from entering controlled space | Frequent contamination events at entry stage |
| Aseptic behavior | Reduces unnecessary movement, interventions, and surface contact | Higher operator-linked contamination risk |
| Material handling | Protects clean-to-dirty flow and transfer discipline | Cross-contamination and process disruption |
| Deviation awareness | Improves early recognition and response to abnormal events | Delayed escalation and weak investigations |
| Requalification | Confirms skills remain current and repeatable over time | Competency drift and audit findings |
What GMP Expectations Require From Personnel Training
A strong cleanroom training system should reflect risk level, room classification, operator role, and process criticality. Training content should not be identical for all personnel. A sterile filling operator and a maintenance visitor do not present the same contamination risk and therefore should not receive the same qualification package.
Core expectations typically include:
- Initial induction before cleanroom access
- Role-based training by process and zone
- Practical gowning assessment
- Aseptic behavior and intervention discipline
- Understanding of contamination pathways
- Periodic retraining and requalification
- Documented approval and traceable training status
Need GMP-ready cleanroom support?
Explore Midposi solutions designed for pharmaceutical and controlled environments, including cleanroom garments and contamination-control product support.
What a Cleanroom Personnel Training SOP Should Include
An audit-ready SOP should define responsibilities, training content, qualification criteria, records, and retraining triggers. It should also clearly distinguish between theoretical training and practical qualification.
| SOP Section | What to Include |
|---|---|
| Purpose | Explain how training supports contamination control, GMP compliance, and personnel qualification |
| Portée | Define which personnel are covered: operators, QA, engineering, maintenance, contractors, visitors |
| Responsibilities | Assign ownership to QA, production, training coordinators, and department supervisors |
| Training matrix | Specify required modules by role, room grade, and access level |
| Qualification method | Describe written evaluation, gowning observation, behavior assessment, and approval process |
| Requalification frequency | Define intervals and event-based retraining triggers |
| Records | State how completion, approval, revision control, and traceability are maintained |
| Deviation response | Define retraining actions after breaches, failures, or contamination-related events |
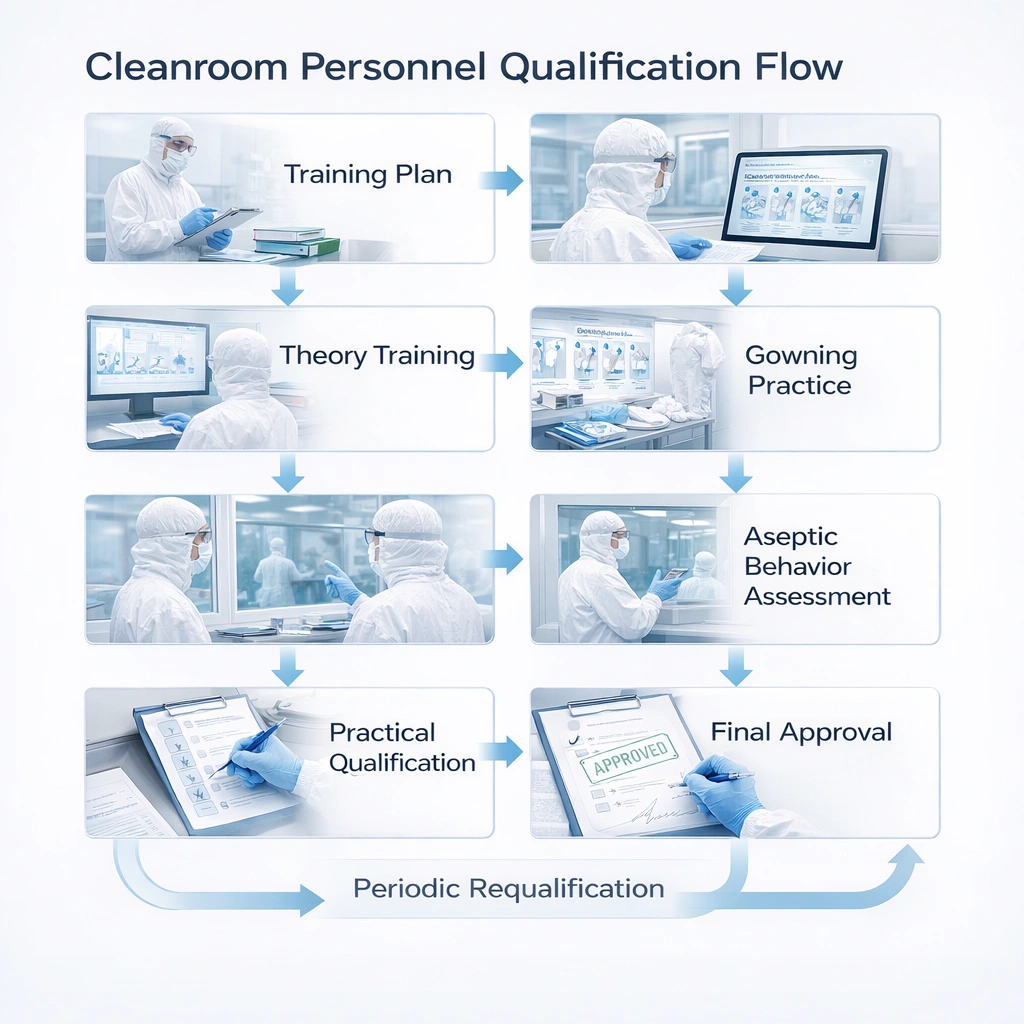
Typical Personnel Qualification Flow
Qualification should follow a consistent sequence rather than a one-time presentation. The strongest programs move from knowledge to demonstration to controlled approval.
Visual Explanation: Qualification Flow
Recommended training sequence:
- Training plan assignment
- Theory training on contamination control
- Gowning instruction and supervised practice
- Aseptic behavior observation
- Practical qualification in simulated or controlled environment
- Final approval for room access or process participation
- Periodic requalification and deviation-triggered retraining
- Do not approve personnel based only on classroom attendance
- Use observation-based signoff for gowning and movement discipline
- Separate temporary access training from full operator qualification
- Reassess after major SOP changes, room changes, or investigation outcomes
What Personnel Should Be Trained On
1. Contamination Fundamentals
Personnel should understand where contamination comes from, how humans contribute to it, and how behavior affects room control. This gives context to rules and improves compliance quality.
2. Gowning and Entry Behavior
Training should include sequencing, garment handling, glove changes, entry flow, and non-contact discipline. This should align closely with your gowning program and garment management process.
3. Aseptic Technique and Cleanroom Conduct
Operators must know how to move, reach, turn, communicate, and intervene without disrupting controlled airflow or contacting critical surfaces.
4. Room-Specific Rules
Different areas may have different restrictions, transfer protocols, and access conditions. Personnel should be qualified for the zones they actually enter.
5. Deviation and Incident Awareness
Training must explain what to do after a gown breach, glove touch, dropped item, unexpected movement, or environmental alert.
Correct vs Incorrect Personnel Behavior
One of the most effective training tools is visual contrast. Personnel understand expectations faster when they can compare acceptable behavior with common mistakes.
Visual Explanation: Correct vs Incorrect Behavior
Training visuals should compare slow controlled movement, correct hand positioning, and clean transfer technique against rushing, unnecessary turning, touching non-sterile surfaces, or blocking critical airflow.

| Correct Practice | Incorrect Practice | Why It Matters |
|---|---|---|
| Move slowly and deliberately | Move quickly or abruptly | Rapid movement increases particle generation and airflow disturbance |
| Keep hands within trained work zone | Reach across critical areas unnecessarily | Reduces contact and contamination risk |
| Follow defined transfer paths | Shortcut material or personnel flow | Protects clean-to-dirty segregation |
| Escalate possible breaches immediately | Ignore minor errors or self-correct silently | Improves investigation quality and risk control |
When Retraining and Requalification Should Be Triggered
Retraining should never be limited to an annual calendar event. It should also be triggered by operational risk signals and quality events.
- Periodic scheduled requalification
- Major SOP revision
- Room classification or process change
- Failed gowning or behavior assessment
- Human-linked EM excursion or investigation finding
- Long absence from cleanroom work
- Repeated minor deviations indicating competency drift
Visual Explanation: Training Deviation Trend
A dashboard can help QA review training-related deviations by department, operator seniority, room grade, or incident type. This makes retraining more targeted and data-based.
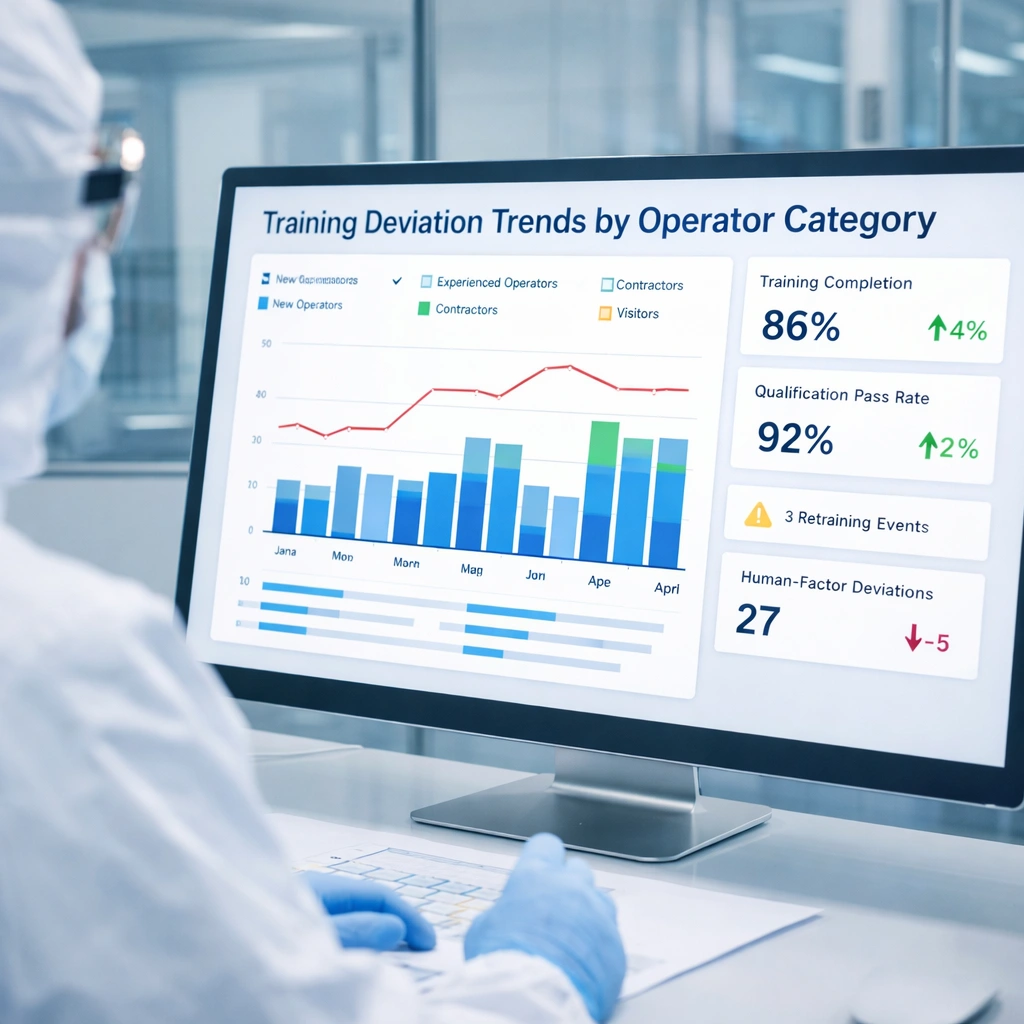
What Auditors Commonly Look For
During inspections, training systems are often reviewed not only as documents but as evidence of control. Weak traceability, generic content, or poor practical qualification can quickly undermine confidence in the broader contamination control strategy.
Auditors often ask for:
- Role-based training matrix
- Latest approved training SOPs
- Records for specific named operators
- Evidence of practical qualification
- Retraining records after deviations
- Proof that training content matches current procedures
Best-Practice Model for a High-Performance Training Program
The most effective cleanroom training systems share several characteristics:
- Risk-based content by role and cleanroom grade
- Strong integration with gowning, cleaning, environmental monitoring, and deviation systems
- Practical qualification, not just attendance records
- Routine refreshers and event-based retraining
- Visual teaching tools that show right vs wrong behavior
- Clear documentation and fast traceability during audits
Training should not be isolated from operations. It should be part of the living contamination control system.
Build a More Audit-Ready Cleanroom Environment
Upgrade your contamination control system with GMP-ready cleanroom solutions designed for pharmaceutical and controlled environments.
Questions fréquemment posées
What is cleanroom personnel training?
Cleanroom personnel training is a documented program that teaches staff how to behave, gown, move, and work within controlled environments without compromising contamination control.
Why is personnel training important in GMP cleanrooms?
Because people are a major contamination source. Proper training reduces particle shedding, poor aseptic technique, incorrect movement, and deviation risk.
Does cleanroom training need practical qualification?
Yes. Classroom training alone is not enough. Gowning, behavior, and process-specific actions should be observed and approved in practice.
How often should personnel be retrained?
Retraining is usually periodic, but should also be triggered by deviations, SOP changes, long absence, failed qualification, or contamination investigations.
Who should be included in cleanroom training?
Operators, QA personnel, supervisors, maintenance staff, contractors, and any visitor who enters controlled areas should receive appropriate training based on their access level and risk.
What documents support an audit-ready training system?
Key records include training SOPs, training matrix, attendance and qualification records, requalification history, and deviation-linked retraining documentation.
How does personnel training connect to environmental monitoring?
Environmental monitoring excursions often have a human factor component. Training helps reduce avoidable contamination events and supports better response when abnormal results occur.
Is cleanroom training only about gowning?
No. Gowning is only one part. Effective training also covers contamination awareness, aseptic behavior, movement control, transfer practices, and deviation response.




