Hospital Cleanroom Mop Selection Best Practices
A practical, compliance-first guide to choosing mop materials, defining ISO zone cleaning programs, and building validation-ready protocols for healthcare facilities.

Cover image: mop system selection in healthcare controlled environments.
Introduction
Selecting the right cleanroom mop for healthcare facilities is a core control point for contamination prevention, infection control, and inspection readiness. A hospital-grade mop program must align with ISO classification targets, disinfectant compatibility, and validation documentation practices—especially for high-risk zones.
If you need a full product overview for procurement and program design, start from our wholesale cleanroom mop guide.
Hospital Cleanroom Classifications
ISO classification sets particle limits and drives the mop material and change-out strategy. As zones become more critical, programs shift toward low-lint microfiber surfaces, individual packaging, and tighter use-life control.
| Classe ISO | Typical Hospital Areas | Mop Program Focus |
|---|---|---|
| OIN 5 | Compounding / sterile fields | Ultra-low lint, strict change-out |
| ISO 7 | OR support, processing rooms | Routine SOP + zone segregation |
| ISO 8 | Corridors, supply, buffer zones | Durability + cost control |
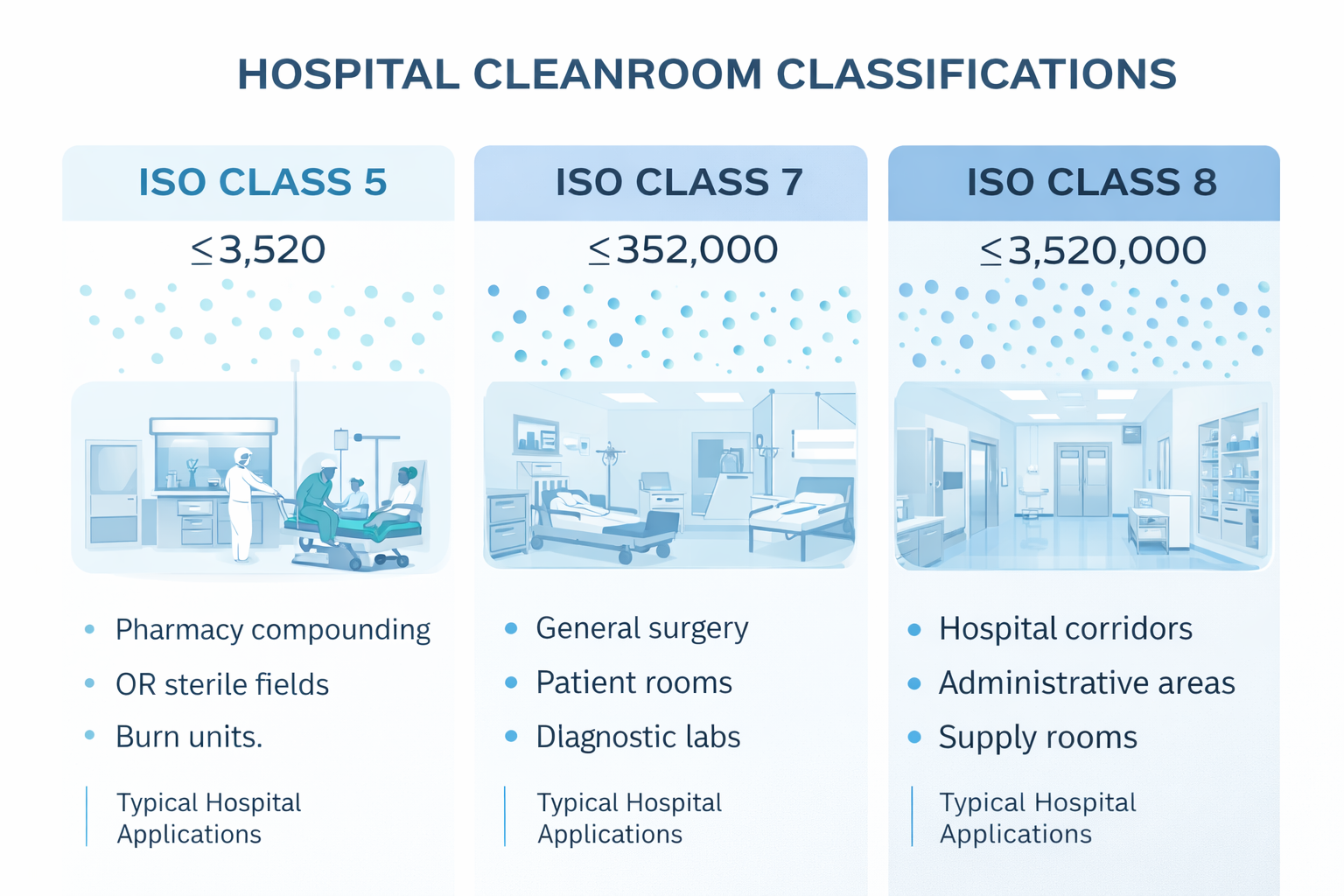
ISO zone illustration for hospital use: align mop materials, packaging, and change frequency to the target class.
Mop Types for Healthcare Programs
Disposable Head Mops
Best for high-risk zones, rapid changeover, and cross-contamination control.
- Reduces laundering validation load
- Supports strict zone segregation
- Ideal for outbreaks, isolation, and critical areas
Reusable / Launderable Mops
Best for large-area programs where validated laundering is established.
- Lower unit cost over lifecycle
- Requires validated wash/dry cycle and segregation
- Ideal for ISO 7–8 support and non-critical zones

Decision-support view: match risk level, validation burden, and change frequency to the right mop strategy.
Hospital Cleaning Protocol Implementation
3-Bucket System (Workflow)
- Dirty bucket — initial soil capture
- Rinse bucket — reduce carryover
- Clean/disinfectant bucket — final controlled application
Operational Rules That Reduce Deviations
- Clean-to-dirty flow with zone segregation
- Head change-out at defined triggers (soil, time, zone)
- Handle disinfection and documented checks
- Training + competency sign-off for operators

Protocol scene: 3-bucket workflow with controlled application and reduced cross-contamination risk.
Conformité & Documentation Requirements
Validation-Ready Document Set
- Material and lot traceability
- Cleaning efficacy evidence (program-specific)
- Change control for supplier/material updates
- Training records and SOP revision history
Risk Control in Daily Operations
- Zone-based color coding and storage separation
- Defined change-out frequency by ISO zone
- Documented checks (visual, ATP/monitoring where used)
- Audit-ready procurement specs & acceptance criteria
Tip: Align mop selection with your ISO/GMP framework first, then standardize SKUs by zone and workflow to reduce deviations.
Future Trends: Smart Cleanroom Cleaning
Healthcare facilities are moving toward tighter digital traceability, more frequent monitoring, and workflow automation. Future-ready programs emphasize standardization, training discipline, and data-driven oversight—without complicating daily execution.
- Digital SOP execution + checklists
- Zone-based accountability with traceable batches
- Smarter stocking and rapid-response readiness

Concept image: smart cleaning programs focus on traceability, monitoring, and consistent execution.
Conclusion
A hospital cleanroom mop program succeeds when ISO targets, zone risk, and operational discipline are designed together. Disposable strategies reduce validation overhead in critical areas, while reusable systems perform well when laundering and segregation are validated and enforced.
Build your purchasing specs around ISO/GMP alignment, then standardize SKUs and SOPs for day-to-day reliability—so your program stays effective, auditable, and scalable.



