A QA-oriented comparison of continuous filament polyester vs split microfiber — focused on shedding risk, sterilization stress, and validation evidence for GMP environments.
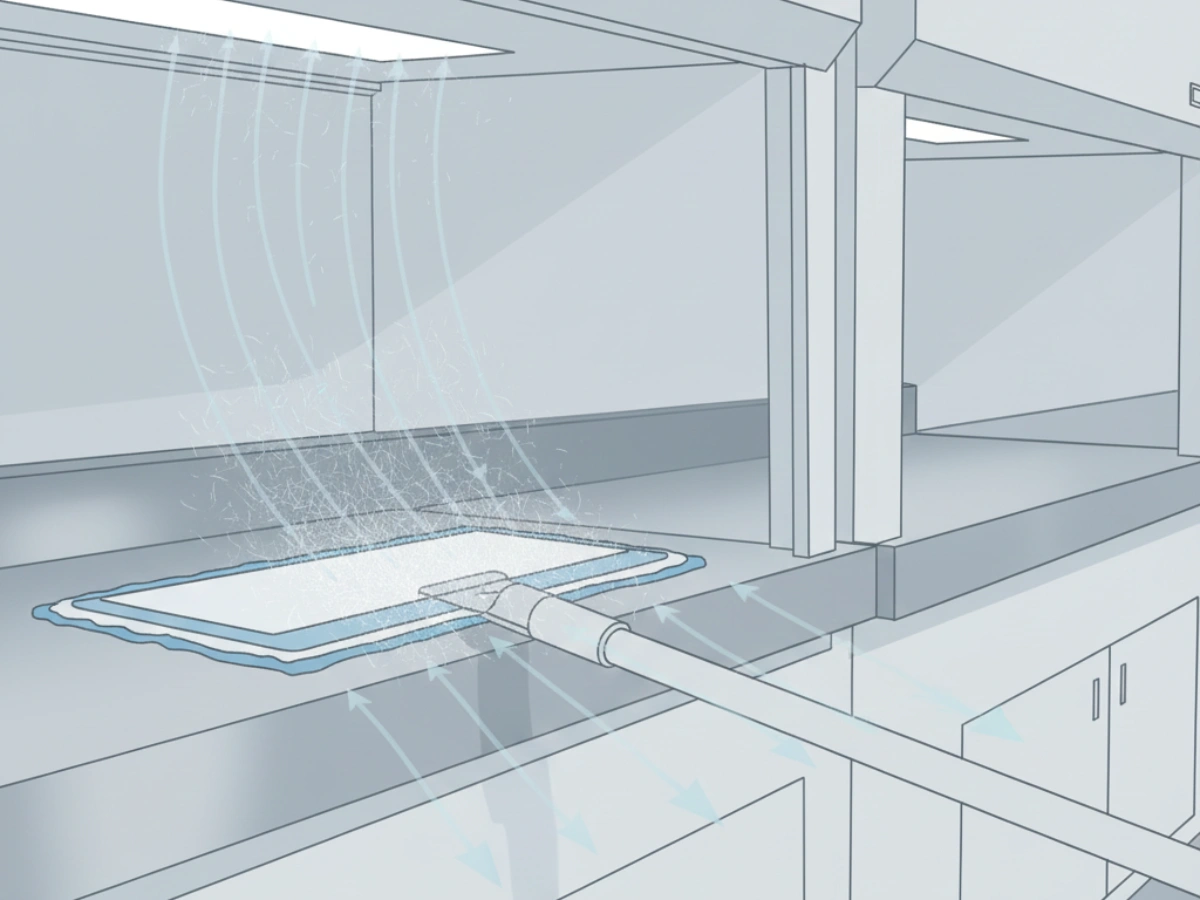
In sterile manufacturing, fibers released from cleaning tools are treated as a contamination control failure. In practice, “low lint” is not a marketing phrase — it is a validation variable that affects CCS effectiveness and deviation risk.
For QA teams, the key question is repeatability: does the mop maintain its shedding profile after the maximum allowed sterilization and handling cycles? If you need a sourcing-level framework, start from the pillar guide: pharmaceutical cleanroom mop supplier.
Linting risk is driven by fiber architecture (continuous vs split), edge construction, and the stress profile from sterilization and use. “Microfiber” is not automatically safer; split filaments can introduce more fracture points.

Even when a mop is labeled “autoclavable,” repeated steam exposure can change fiber integrity over time. For split microfiber structures, steam cycles may accelerate filament fracture and edge fatigue.

The highest shedding risk often comes from edges — especially after repeated abrasion. Laser-cutting and ultrasonic sealing are common approaches to reduce macro-linting. For a deep technical view of how these steps are controlled, see: Cleanroom Mop Head Manufacturing Process.
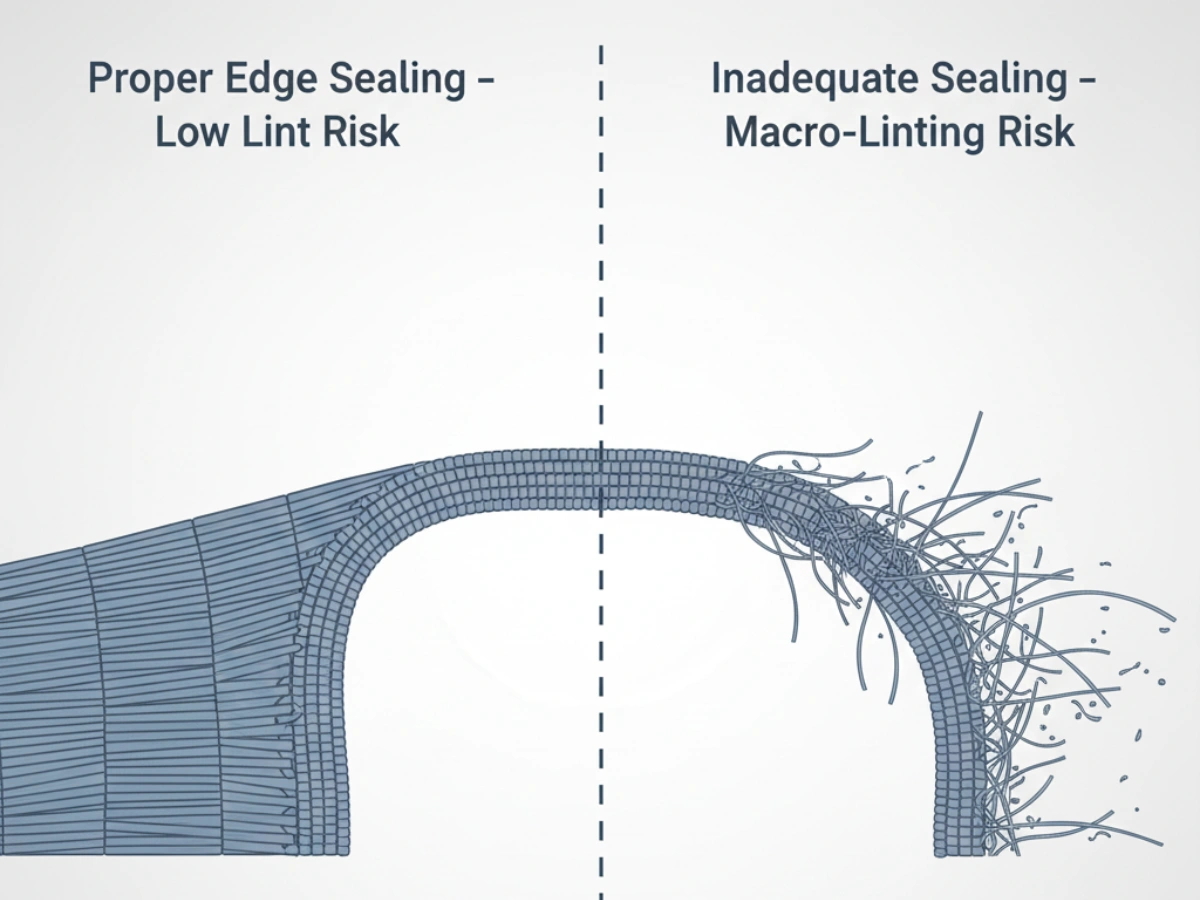
Microfiber’s split structure improves pickup efficiency but introduces more micro-ends and fracture points. If the product is processed repeatedly (steam cycles) or edges are not fused properly, linting risk rises — which is why Grade A use typically demands stronger evidence and tighter limits.
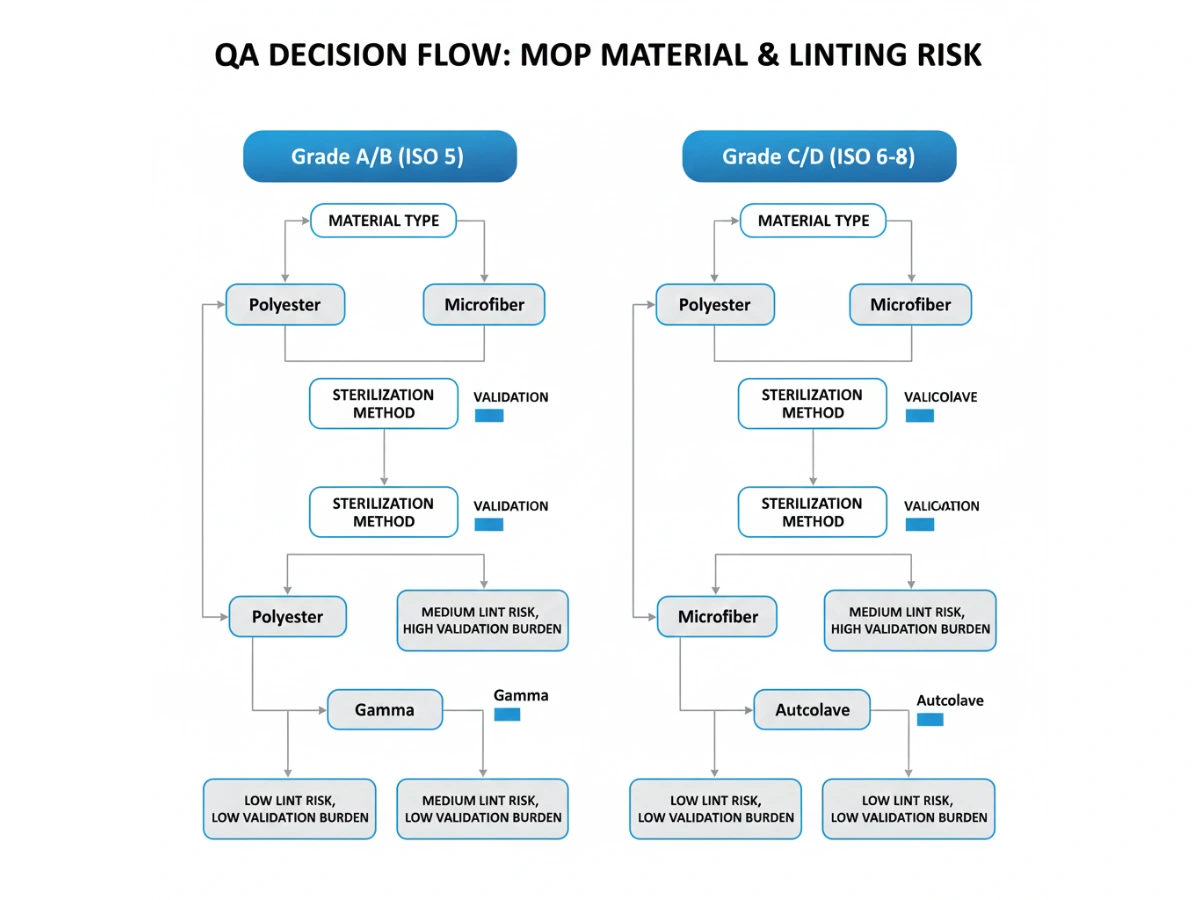
Use a decision tree that aligns grade requirement, sterilization method, and evidence availability (shedding tests, edge sealing method, cycle durability). If you are building a full supplier qualification package, cross-reference the sourcing framework here: pharmaceutical cleanroom mop supplier.


Nous vous contacterons dans un délai d'un jour ouvrable, veuillez faire attention à l'e-mail avec le suffixe "*@midposi.com".