This standard operating procedure establishes a risk-based approach for environmental monitoring in GMP cleanrooms, covering EM location selection, sampling procedures, monitoring frequency, alert and action limits, trending, CAPA, change control, and training.

A risk-based environment monitoring SOP helps GMP cleanrooms maintain microbiological control by defining monitoring locations, sampling methods, frequencies, alert limits, trending rules, and CAPA workflows based on contamination risk.
Use a structured risk model to prioritize monitoring points by personnel activity, airflow disturbance, proximity to Grade A, material transfer, and historical alert rate.
Align monitoring methods and frequencies with cleanroom grade, risk score, and process criticality.
Trend monitoring data continuously and define alert and action responses before excursions occur.
Link alerts to documented investigation, root cause analysis, corrective action, and effectiveness verification.
Control all EM program changes through QA-approved change control and periodic training.
| Version | Date | Description | Prepared By | Approved By |
|---|---|---|---|---|
| 1.0 | 2026-03-23 | Initial Issue | Quality Assurance | Quality Manager |
This SOP establishes a risk-based approach for Environmental Monitoring (EM) in GMP cleanrooms. The purpose is to ensure cleanroom environments remain within specified microbiological limits, provide a systematic framework for EM location selection, establish monitoring frequencies appropriate to risk levels, define procedures for sampling, analysis, and data management, and ensure compliance with EU GMP Annex 1 (2022), FDA cGMP (21 CFR 211), and ISO 14698-1.
| Term | Definition |
|---|---|
| Active Air Sampling | Volumetric air sampling using impactors or slit samplers to capture airborne microorganisms. |
| Alert Level | A specified level of microbial contamination indicating a potential drift from normal operating conditions. |
| Action Level | A specified level of microbial contamination that requires immediate corrective action. |
| Critical Zone | Grade A area where aseptic operations are performed. |
| MIDPOSI EM Location Matrix | Risk assessment tool for scoring EM location risk on a 0–100 scale. |
| Settle Plate | Nutrient agar plate exposed to the environment for passive air sampling. |
| Role | Responsibilities |
|---|---|
| Quality Assurance Manager | Overall EM program approval, change control, regulatory compliance. |
| Microbiology Manager | EM program execution, method validation, and training. |
| Microbiology Technician | Sample collection, analysis, data entry, and alert notification. |
| Production Manager | Cleanroom operation maintenance and gowning compliance. |
| Engineering Manager | HVAC maintenance and airflow pattern verification. |
| Quality Control Manager | Data review, trend analysis, and CAPA initiation. |
All EM monitoring locations shall be selected using a risk-based assessment framework.
RISK SCORE = (Personnel Activity × 0.30) + (Airflow Disturbance × 0.25)
+ (Proximity to Critical Zone × 0.20) + (Material Transfer × 0.15)
+ (Historical Alert Rate × 0.10)
Score Range: 0–100
| Risk Score | Classification | Monitoring Required |
|---|---|---|
| 80–100 | Critical | Continuous monitoring required |
| 60–79 | Haut | Fixed location monitoring (1/shift) |
| 40–59 | Modéré | Rotating monitoring (weekly) |
| 20–39 | Faible | Periodic monitoring (monthly) |
| 0–19 | Minimal | Monitoring not required unless risk changes |
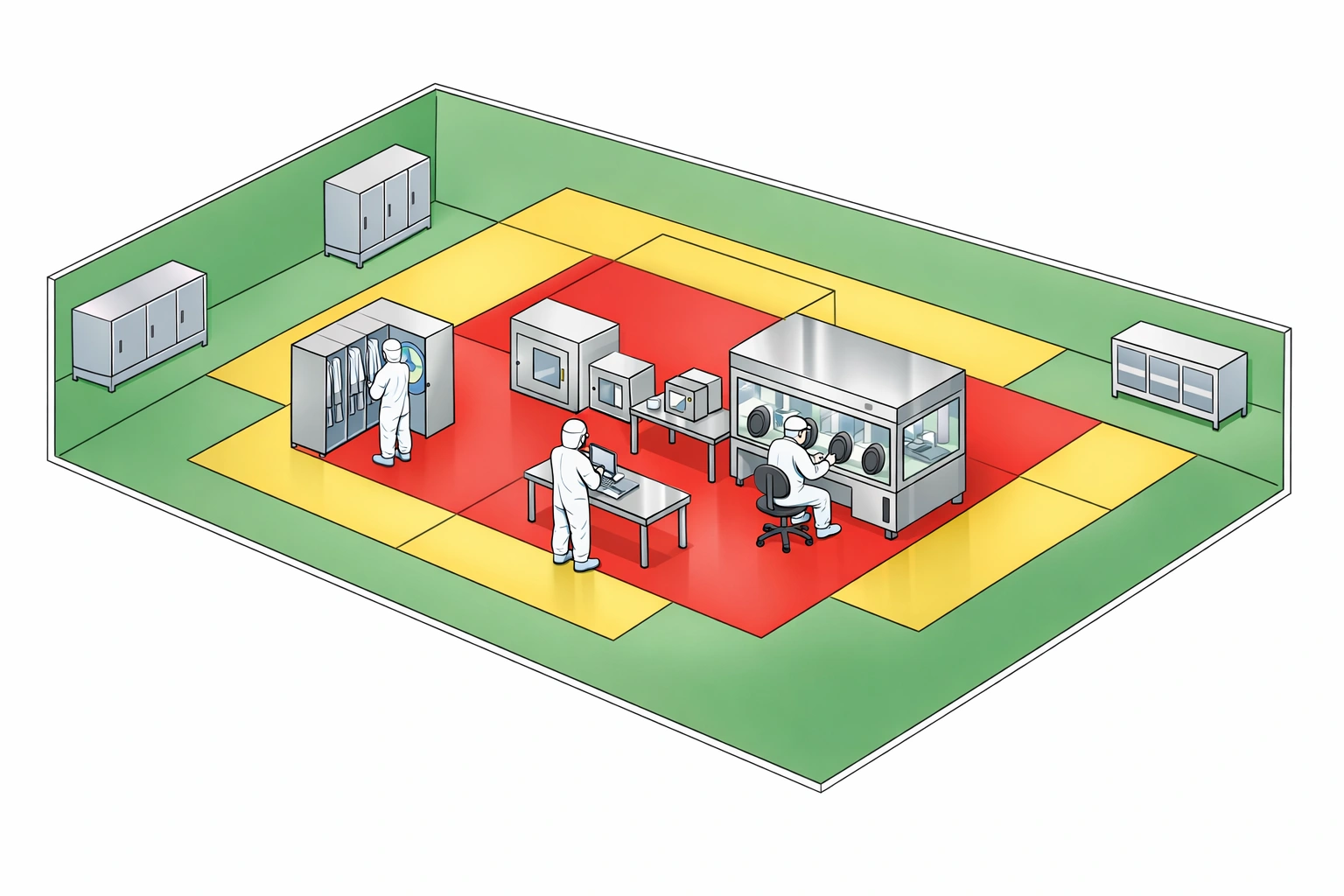
Sampling locations and frequencies should be assigned according to cleanroom grade, risk score, product exposure risk, historical alert rate, and operational traffic. Higher-grade areas require tighter frequency, stronger justification, and lower excursion tolerance.
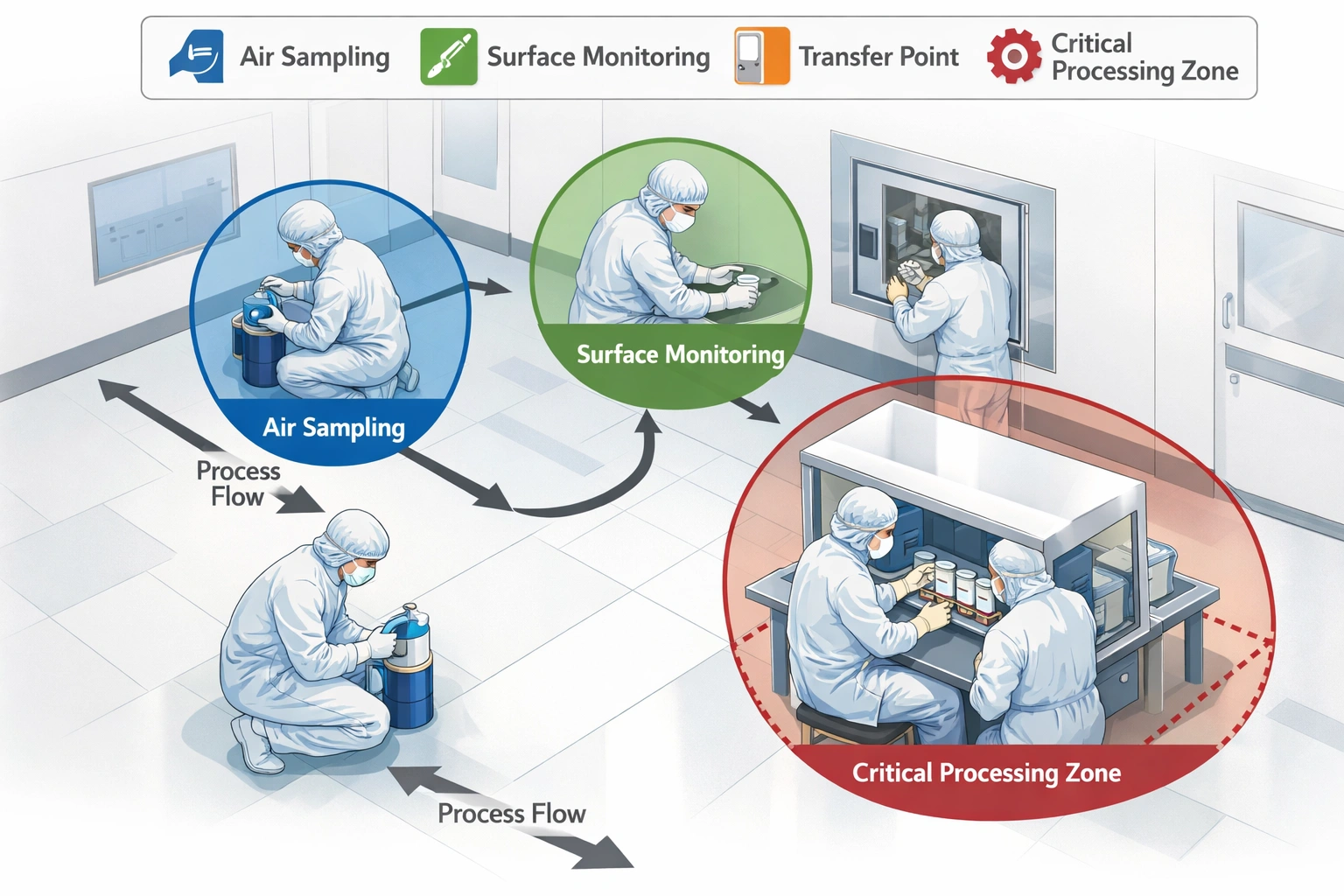
| Equipment | Specification | Placement | Flow Rate | Duration |
|---|---|---|---|---|
| SAS Sampler | 28.3 L/min, single-stage | 15–30 cm from critical surface | 28.3 L/min | 10–15 min/sample |
| Plate Type | Taille | Placement | Exposure Time | Height |
|---|---|---|---|---|
| TSA | 90mm | Directly under critical zone | 4 hours | 1.0–1.5m |
| SDA | 90mm | Adjacent to critical zone | 4 hours | 1.0–1.5m |
Monitoring frequency should be assigned according to cleanroom grade, risk score, process criticality, and historical trend performance. Critical and high-risk locations require continuous or once-per-shift review, while moderate and low-risk locations may use rotating weekly or monthly schedules.

Alert limits indicate potential drift from normal conditions and trigger review, while action levels require immediate investigation and corrective action. Limits should be aligned with cleanroom grade, sample type, historical capability, and regulatory expectations.
Data should be reviewed in real time where possible and trended by location, grade, method, organism, and excursion history. Monthly KPI dashboards should support decision-making and management review.

[ALERT TRIGGERED]
│
┌───┼───┐
│ │ │
First Second Third
Alert Alert Alert
│ │ │
▼ ▼ ▼
Document Trend Immediate
Deviation Analysis Stop
│ │ │
└───┼───┘
▼
Root Cause Analysis
│
┌───┼───┐
│ │ │
Procedural Personnel Equipment/
Training Environment
│ │ │
▼ ▼ ▼
[Corrective] [Retrain] [Maintenance/
Action Re-evaluate Qualification]
Any change affecting facility layout, equipment, personnel flow, monitoring location, risk score, or sampling method must be reviewed under change control before implementation or before production resumes.
All relevant personnel must be trained before performing EM activities. Training should cover aseptic behavior, sampling execution, data entry, alert response, CAPA workflow, and change control requirements. Refresher training should be completed at defined intervals and after significant process changes.
This layout is designed for long-form GMP and cleanroom technical content, with structured tables, SOP-style sections, KPI blocks, and image-ready modules for WordPress or Elementor publishing.


Nous vous contacterons dans un délai d'un jour ouvrable, veuillez faire attention à l'e-mail avec le suffixe "*@midposi.com".