In the highly regulated European pharmaceutical market, EU GMP Annex 1 compliant cleanroom SOP documentation is more than a regulatory box—it is the foundation of patient safety and market longevity. For compliance officers across the EU—from the production hubs in Germany to the biotech clusters in Spain—mastering this documentation architecture is essential for January 2026 readiness.
Who Should Use This Guide
- Compliance Officers: Responsible for adherence to EMA requirements.
- Regulatory Affairs Professionals: Managing EU market authorization dossiers.
- Quality Managers: Overseeing documentation in EU-based sterile facilities.
- Validation Specialists: Ensuring cleanroom validation meets Annex 1 standards.
EU GMP Annex 1 Overview and Implementation Timeline
Revision Published
Significant updates to contamination control and environmental monitoring.
Transition Period Begins
Preparation phase for full compliance.
Full Implementation
Mandatory compliance across all EU member states.
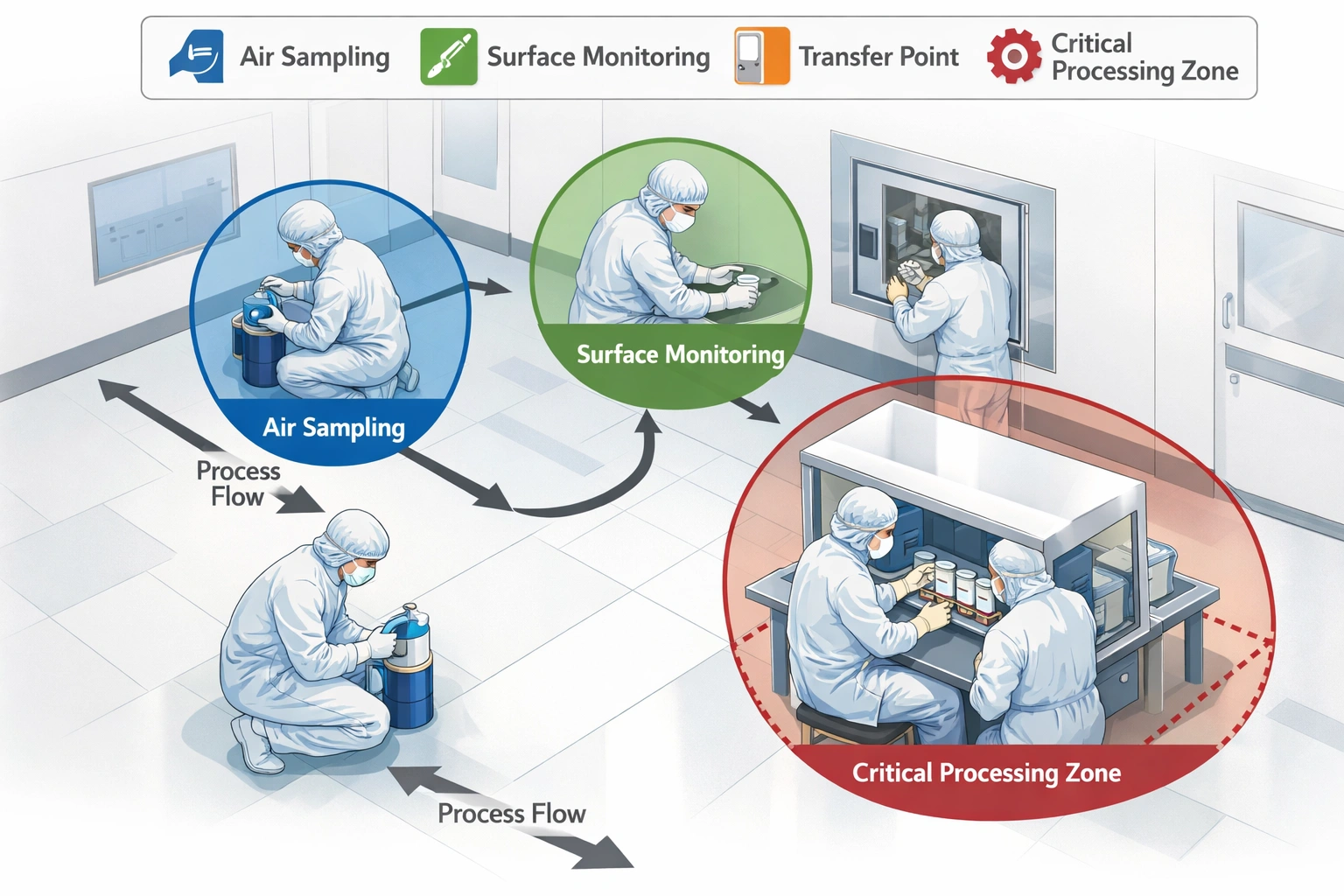
Major Updates Impacting Documentation
| Area | Revised Requirement | Impatto sulla documentazione |
|---|---|---|
| EM | Risk-based with trending | New risk assessment protocols. |
| Contamination | Persistent strategy | Enhanced cleaning SOPs. |
| Lifecycle | Full lifecycle management | Comprehensive control procedures. |
EU Documentation Architecture Requirements
European regulators expect a hierarchical approach that ensures "clear and unambiguous" instructions throughout the facility.
Level 1 & 2: Quality & System SOPs
Includes Quality Manuals and Document Control systems.

Level 3 & 4: Process SOPs and Records
Crucial cleanroom SOPs including gowning, disinfection, and batch production records.
EU Specific Requirement: Documentation must be in the official EU language of the member state where the facility is located. Consider bilingual versions for international clarity.
Cleanroom-Specific Documentation Needs
Common Inspection Deficiencies: Inadequate change control and lack of risk assessment documentation are top reasons for EU inspection findings.

| SOP Type | Specific EU Requirement | Evidence Required |
|---|---|---|
| Disinfezione | Efficacy testing | Protocolli di validazione & reports. |
| Change Control | Rischio & Impact evaluation | Formal assessments. |
| Validazione | Lifecycle verification | VMP & Summary reports. |
EU Member State Specific Considerations
While Annex 1 is harmonized, local nuances exist in major manufacturing hubs:
- Germany: Additional safety documentation and German translations.
- France: Extended traceability requirements for batch records.
- Italy & Spain: Specific training notification and competency assessment records.

Achieve EU GMP Annex 1 Compliance
Our expert team provides centralized, flexible documentation systems designed for 2026 EU regulatory success. Ensure your supply chain and SOPs meet the highest standards.






Expert Commentary: EU GMP Annex 1 Implementation
"The revised Annex 1 represents the most significant update to sterile manufacturing in decades. Regulators expect a demonstrable quality culture embedded in every document—comprehensive risk assessments are the new standard."