For procurement, QA/validation, and facility teams sourcing cleanroom mops for pharmaceutical manufacturing, hospitals, and medical device facilities.

Medical cleanroom mop supplier perspective: contamination control, documentation, and scalable supply.
Related MIDPOSI guides (recommended for procurement):
Medical cleanroom mop procurement typically fails in three places: (1) mismatched ISO class vs mop design, (2) missing validation documentation, and (3) unstable batch quality over time. Your mop supplier should help you reduce these risks with consistent materials, traceability, and documented performance aligned with ISO 14644 and GMP expectations.
This guide summarizes what makes medical cleanroom mops different, how to select disposable vs reusable systems, and how to evaluate a medical cleanroom mop supplier for long-term B2B supply. (See also: Wholesale Cleanroom Mop Hub.)
Medical cleanroom mops are engineered for low particle generation, material purity, and chemical compatibility with disinfectants used in pharma, healthcare, and device manufacturing.
| Framework | What it impacts | Typical buyer teams |
|---|---|---|
| ISO14644 | Cleanroom class alignment, monitoring, particle control targets | Facilities, QA |
| GMP / Annex 1 / Validation | Cleaning process control, change control, documentation expectations | QA/Validation, Compliance |
| ISO 13485 / QMS | Supplier controls, traceability, complaint handling | Supplier Quality, Procurement |
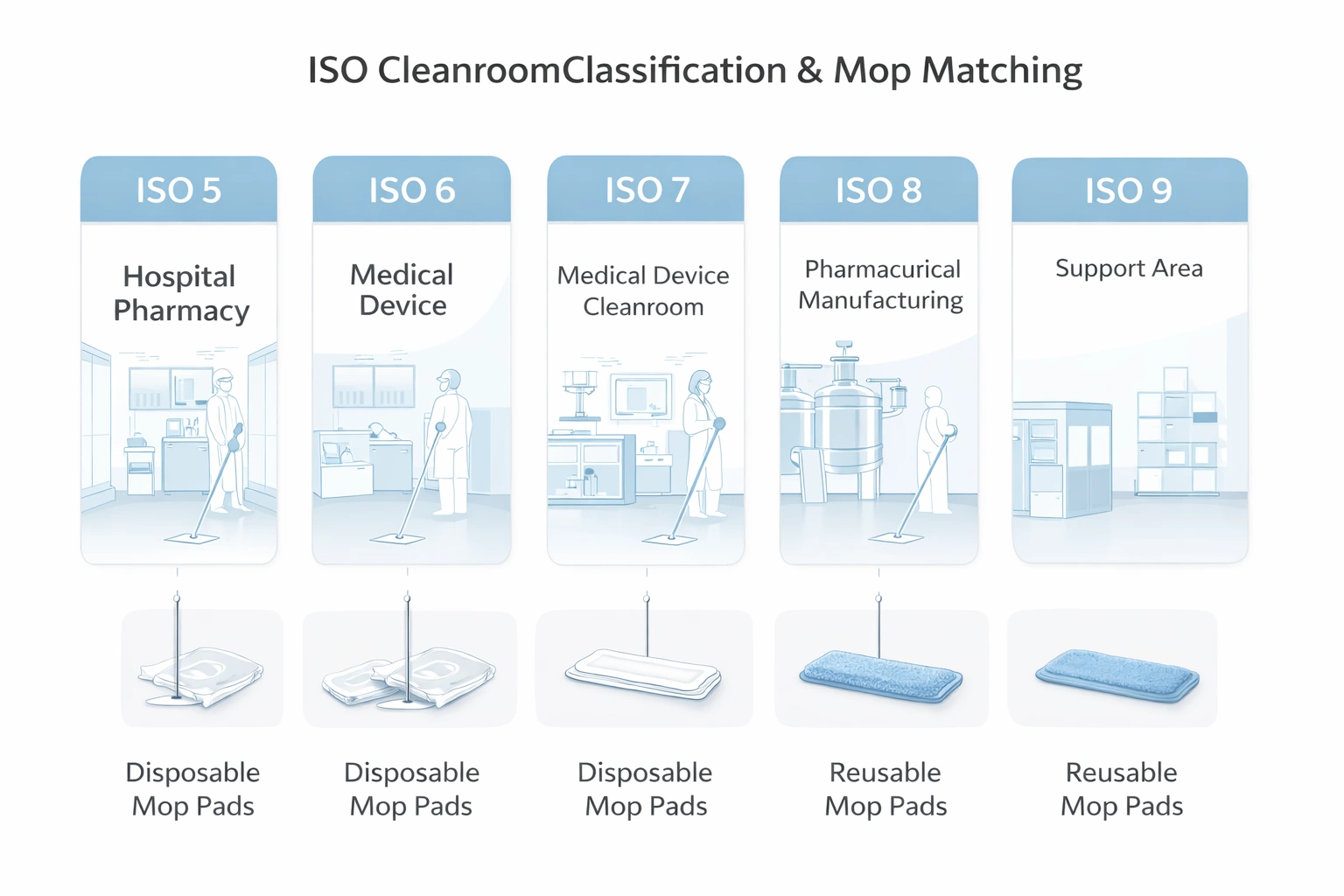
ISO 14644 mapping helps procurement align mop type, material, and validation burden to area risk.

Decision guide: evaluate cross-contamination risk, validation resources, usage volume, and TCO.
For deeper cost/compliance trade-offs, see: Mop per camere bianche monouso e riutilizzabili.

Standardized protocols reduce variability—critical for audit readiness and infection control programs.
Hospitals and healthcare environments often emphasize standardized workflows (zone separation, SOP adherence, and training). If you manage healthcare procurement, you may also want: Hospital Cleanroom Mop Selection.

Trend: visibility and accountability—digital SOPs, monitoring, and workflow traceability.
If you are sourcing mops for medical cleanrooms, build your evaluation around: ISO-class alignment, material purity/compatibility, validation documentation, and long-term supply stability.


Ti contatteremo entro 1 giorno lavorativo, fai attenzione all'email con il suffisso “*@midposi.com”.