Che cos'è uno straccio per camera bianca sterile?
UN sterile cleanroom mop is a low-lint mop supplied in sterile packaging for controlled cleaning workflows where sterile presentation is required. It is commonly used in GMP cleanrooms, pharmaceutical manufacturing, biotechnology facilities, aseptic support areas, medical device production, laboratories, and other controlled environments.
A sterile cleanroom mop is a cleanroom-compatible mop pad or mop cover that has been sterilized and packaged for controlled use. Buyers should review the mop material, sterility information, packaging format, lot traceability, documentation, and SOP suitability before purchasing.

What Does “Sterile Cleanroom Mop” Mean?
A sterile cleanroom mop is a mop designed for controlled environments and supplied in a sterile format. The mop material is usually selected for low-lint performance, controlled wiping, cleanroom compatibility, and use with defined cleaning or disinfection procedures.
Sterile cleanroom mops are different from regular mops because they are not only evaluated by absorbency or cleaning appearance. Buyers also review sterility status, packaging, material construction, traceability, documentation, and suitability for internal SOPs.
Why Sterile Cleanroom Mops Matter
In GMP and controlled cleanroom environments, cleaning tools can become a contamination source if they shed fibers, introduce particles, or are not handled according to SOP. Sterile cleanroom mops help support controlled workflows where sterile presentation and cleaning discipline are important.
Sterile Presentation
Supports workflows where the mop must enter controlled areas in a sterile or validated presentation format.
Low-Lint Cleaning
Helps reduce fiber-shedding concerns during floor, wall, ceiling, and controlled-surface cleaning.
QA Review Support
Packaging, sterility, product specifications, and traceability details help buyers review suitability before use.
Sterile Cleanroom Mop vs Non-Sterile Cleanroom Mop
Sterile and non-sterile cleanroom mops can both be used in controlled environments, but they serve different risk levels, SOP requirements, and procurement strategies.
| Punto di confronto | Mop sterile per camere bianche | Mocio per camere bianche non sterile |
|---|---|---|
| Stato di sterilità | Fornito sterile o irradiato con raggi gamma a seconda delle specifiche del prodotto | Not supplied as sterile |
| Uso tipico | Higher-control workflows, sterile support areas, QA-driven applications | Routine controlled-area cleaning where SOP allows |
| Confezione | Often individually packed or sterile-packed for controlled presentation | Standard controlled packaging or bulk packaging depending on product |
| Buyer Review Focus | Sterility information, packaging format, lot traceability, material, and documentation | Material, lint level, packaging, cost, application area, and replacement workflow |
| Cost Consideration | Usually higher due to sterilization and packaging requirements | Usually more cost-efficient for routine cleaning where sterile presentation is not required |

Where Are Sterile Cleanroom Mops Used?
Sterile cleanroom mops are used in environments where cleaning tools must support contamination control and sterile presentation. The final selection depends on the facility SOP, cleanroom grade, area risk, product type, and QA requirements.
Typical application areas include:
- Pharmaceutical manufacturing cleanrooms
- Aseptic processing support areas
- Biotechnology production rooms
- Medical device clean manufacturing areas
- Laboratories with controlled cleaning requirements
- Cleanroom floors, walls, ceilings, and controlled support areas
How Sterile Cleanroom Mops Are Commonly Reviewed
Buyers should review sterile cleanroom mops as part of a cleaning system, not only as a mop head. The mop material, frame compatibility, packaging, sterility information, bucket workflow, and documentation all matter.
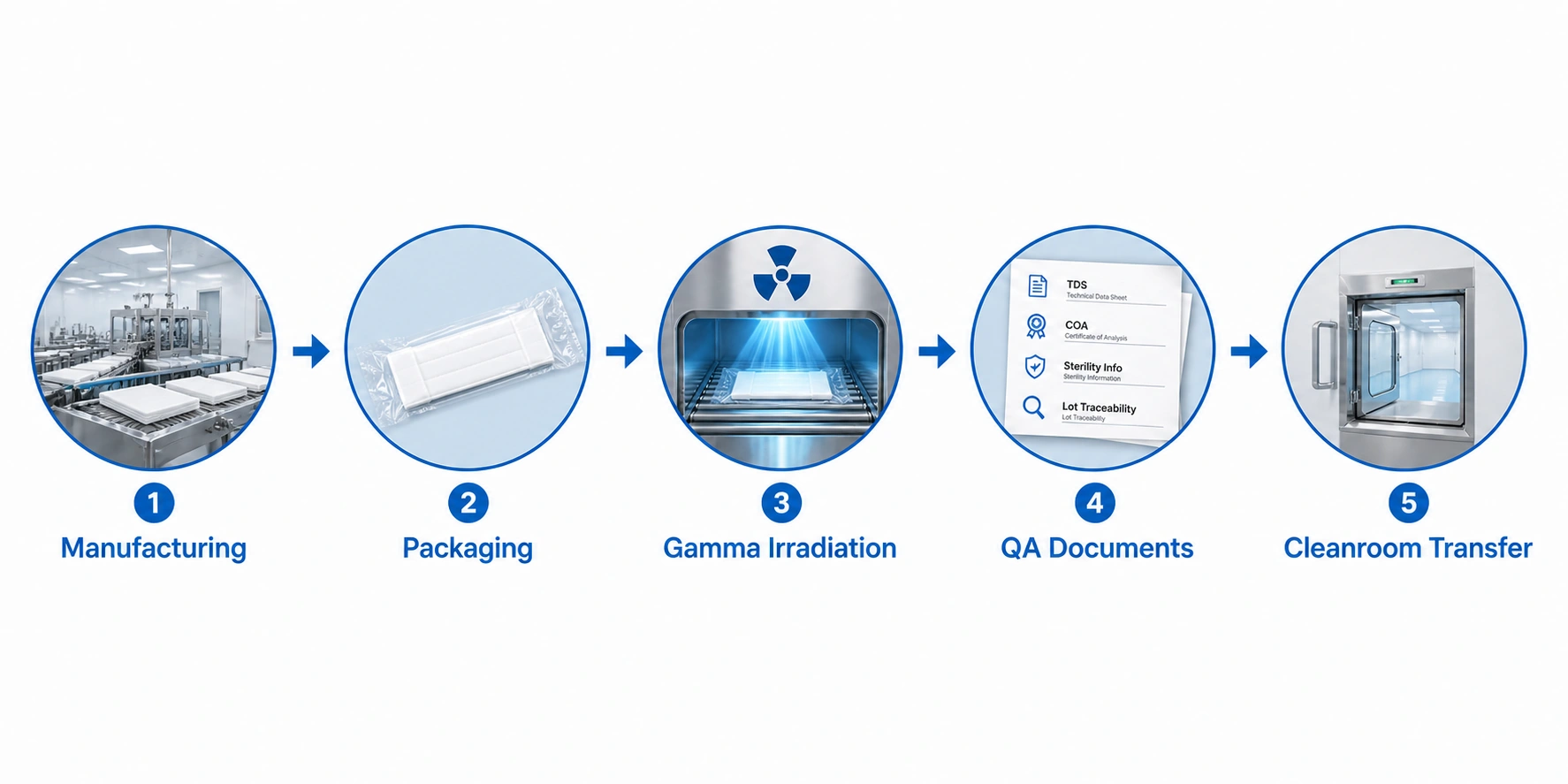
Sterility, Gamma Irradiation and Packaging
Many sterile cleanroom mop products are supplied after sterilization and packed for controlled presentation. For buyers, the most important question is not only whether the mop is sterile, but whether the sterility-related information and packaging format match the facility’s internal SOP and QA review requirements.
Buyers should ask:
- Is the mop supplied sterile or non-sterile?
- What sterilization method is used?
- Is the product individually packed or bulk packed?
- Is the sterile version double-bagged or vacuum packed?
- Can sterility-related information be provided for QA review?
- How is lot or batch traceability handled?
Sterile Cleanroom Mop Selection Guide
The table below helps procurement managers, QA teams, cleanroom supervisors, and distributors compare sterile mop requirements before sample testing or bulk purchase.
| Buyer Requirement | Messa a fuoco consigliata | Perché è importante |
|---|---|---|
| GMP facility cleaning | Low-lint sterile mop pad with clear product specification | Supports controlled cleaning and QA review |
| Sterile presentation | Sterile packaging, lot information, and handling suitability | Helps align product with facility SOP |
| Floor, wall, and ceiling cleaning | Compatible flat mop frame and cleanroom handle | Improves reach, surface contact, and operator workflow |
| Controllo della contaminazione crociata | Disposable sterile mop cover or controlled replacement workflow | Reduces reuse-related contamination concerns |
| Supplier qualification | TDS, packaging details, sterility information, sample record, and quotation | Supports procurement and QA approval process |
What Documents Should Buyers Request?
Sterile cleanroom mop buyers should request documentation that supports material review, sterility review, packaging review, and purchasing control.
Scheda Tecnica
Product size, material, construction, weight, packaging, and intended cleanroom application.
Informazioni sulla sterilità
Sterile status, sterilization method, packaging format, and related information for QA review.
Dettagli dell'imballaggio
Inner pack, sterile pack, carton quantity, carton size, storage notes, and label information.
Tracciabilità del lotto
Product code, lot number, batch reference, or carton-level traceability information where available.
Esempio di record di approvazione
Approved model, mop weight, packaging format, sterile status, and test feedback before bulk purchase.
Supplier Support
MOQ, lead time, quotation, repeat-order communication, and B2B distributor support.

MIDPOSI Sterile Cleanroom Mop Options
MIDPOSI supports B2B buyers with sterile and non-sterile cleanroom mop options for pharmaceutical cleanrooms, biotechnology facilities, laboratories, medical device manufacturing, and other controlled environments.
Serie di mop bianchi per camere bianche
- 40g light option
- 55g medium option
- 65g heavy-duty option
- Versioni sterili e non sterili
- Costruzione in poliestere a basso rilascio di fibre
B2B Support
- Supporto campione
- Supporto alle specifiche
- Packaging details
- Quotation and MOQ communication
- Supporto al distributore e all'acquirente della struttura
Quando gli acquirenti dovrebbero scegliere i mop sterili per camere bianche?
Sterile cleanroom mops are not required for every controlled area. Buyers should choose sterile mops when their SOP, QA team, cleanroom grade, contamination-control strategy, or customer requirements call for sterile presentation.
Scegli i panni sterili quando:
- The area requires sterile presentation.
- The SOP specifies sterile or gamma-irradiated cleaning tools.
- The facility wants stronger replacement control.
- Il flusso di lavoro di pulizia viene utilizzato nelle aree GMP a rischio più elevato.
- QA requires sterility-related information for review.
Non-sterile may be suitable when:
- La SOP accetta mop pad non sterili per camere bianche.
- The application is routine controlled-area cleaning.
- Sterile presentation is not required.
- The buyer needs a more cost-efficient disposable option.
- The area is lower risk under internal procedures.
FAQ About Sterile Cleanroom Mops
What is a sterile cleanroom mop?
A sterile cleanroom mop is a low-lint mop supplied sterile for controlled cleaning workflows where sterile presentation, contamination control, and QA review are required.
Where are sterile cleanroom mops used?
Sterile cleanroom mops are commonly used in GMP cleanrooms, pharmaceutical manufacturing, biotechnology facilities, aseptic support areas, medical device production, laboratories, and other controlled environments.
Are sterile cleanroom mops required in all GMP cleanrooms?
No. Sterile cleanroom mops are required only when the facility SOP, cleanroom grade, contamination-control strategy, or QA requirements specify sterile presentation or sterile cleaning tools.
Qual è la differenza tra i panni per camere bianche sterili e non sterili?
Sterile cleanroom mops are supplied sterile or gamma-irradiated depending on product specification. Non-sterile cleanroom mops are not supplied sterile and are usually used in routine controlled-area cleaning where SOP allows.
What documents should buyers request for sterile cleanroom mops?
Buyers should request product specifications, material information, sterility information, packaging details, lot traceability information, sample approval records, and supplier qualification support where required.
Does a sterile cleanroom mop guarantee GMP compliance?
No. A sterile cleanroom mop does not guarantee GMP compliance. It supports GMP cleaning only when selected, qualified, documented, and used according to the facility’s approved SOP and QA system.
Need Sterile Cleanroom Mops for GMP Cleaning?
Contact MIDPOSI to discuss sterile cleanroom mop options, sterile packaging, low-lint mop materials, sample availability, documentation support, and B2B supply details.



