MIDPOSI disposable cleanroom mops are designed for pharmaceutical cleanrooms, ISO-controlled environments, laboratories, medical device production and contamination-sensitive manufacturing areas where facilities require low-lint cleaning tools, simplified mop replacement workflows and stronger contamination-control consistency.

A disposable cleanroom mop is a single-use or limited-use cleanroom mop cover designed for GMP and ISO-controlled environments where facilities need low-lint cleaning tools, simplified replacement workflows and reduced reuse-related contamination concerns.
Disposable cleanroom mops are commonly used in pharmaceutical manufacturing, medical device production, laboratory cleanrooms, biotechnology facilities and semiconductor support environments.
Disposable cleanroom mops are single-use or limited-use mop covers designed for controlled environments where repeated mop washing, storage, inspection and reuse may create additional contamination-control concerns.
Unlike general janitorial mop heads, disposable cleanroom mop covers should be selected for low-lint performance, material compatibility, surface contact, packaging format, frame compatibility and suitability for cleanroom cleaning SOPs.
They are commonly used with compatible クリーンルーム用フラットモップシステム、 クリーンルームモップハンドル, and defined cleaning procedures for floors, walls, ceilings and controlled surfaces.
Disposable mop covers are often selected when the facility wants a cleaner, simpler and more controlled alternative to repeated mop washing, storage, inspection and reuse.
Single-use mop covers help reduce contamination concerns associated with previous use, incomplete washing, uncontrolled storage or repeated handling.
Disposable workflows can make it easier to define one-area-one-mop-cover procedures and reduce uncertainty during routine cleanroom cleaning.
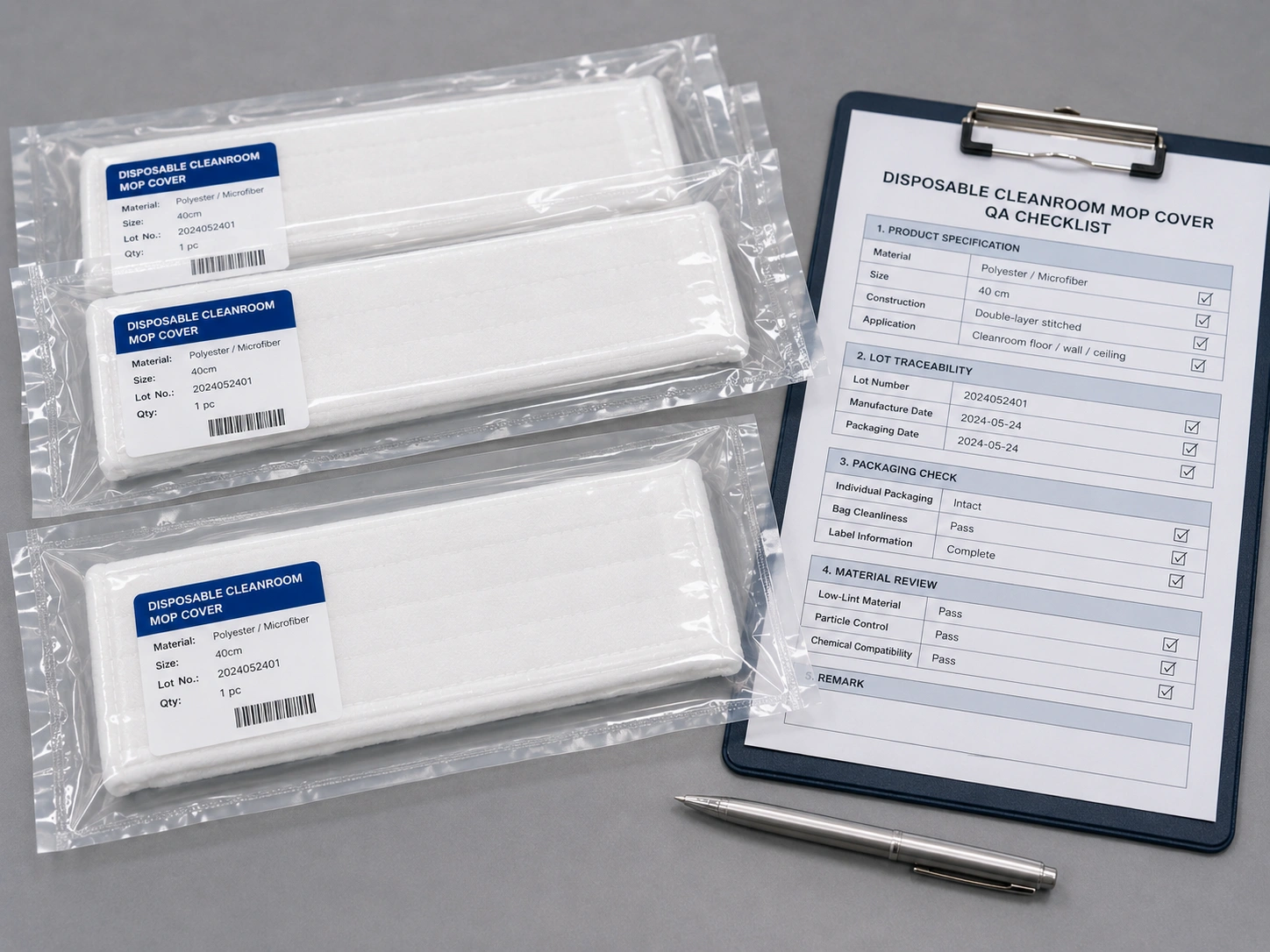
Clean packaging, lot traceability and consistent product specifications can help procurement and QA teams review cleanroom consumables more efficiently.
A disposable mop cover works best when it is matched with the correct flat mop frame, cleanroom handle, bucket or wringer workflow, disinfectant SOP and area zoning procedure.

Disposable low-lint mop cover for cleanroom floor, wall, ceiling and controlled surface cleaning.
安定した表面接触と効率的な洗浄範囲をサポートする互換性のあるフレーム。
Smooth handle suitable for controlled-environment cleaning and repeatable operator movement.
Defined cleaning sequence for zone control, disinfectant use and mop cover replacement.
Disposable cleanroom mops are suitable for controlled-environment applications where reuse control, zone separation, packaging traceability and cleaning documentation are important.
Supports controlled cleaning workflows in GMP facilities, formulation areas, filling support areas and QA-managed environments.
Supports surface cleaning where particles, residues and production-area cleanliness must be controlled.
Suitable for routine cleaning of laboratory cleanrooms, controlled corridors and process-adjacent areas.
Useful where low-lint cleaning tools are required for controlled floors, walls and work-adjacent surfaces.
Supports consistent wet mopping and removal of residues from cleanroom floors.
適切なハンドルを備えたフラット モップ システムは、垂直およびオーバーヘッドの清掃作業をサポートできます。
The right choice depends on contamination-control goals, cleaning frequency, sterilization capability, waste policy, documentation needs and total operating cost.
| 選択要素 | 使い捨てクリーンルームモップ | 再利用可能なクリーンルームモップ |
|---|---|---|
| 再利用に伴う汚染リスク | モップ カバーは使用後、または定義された使用サイクル後に交換されるため、低くなります。 | Depends on washing, sterilization, inspection and storage control |
| SOPのシンプルさ | 1 エリアまたは 1 セッションの使用を定義しやすくなりました | ライフサイクル追跡と再処理手順が必要 |
| コスト構造 | 1回あたりの消耗品コストが高い | 再処理が適切に管理されている場合、使用あたりのコストが削減されます |
| 検証ワークロード | 再利用とライフサイクルに関する懸念の一部が簡素化される可能性があります | Requires validation of cleaning, sterilization, storage and reuse limits |
| ベストフィット | High-control areas, contamination-sensitive workflows and simplified tool control | 強力な再処理管理を備えた定期的な清掃エリア |
Disposable mop covers can be integrated into controlled cleaning procedures by defining when a cover is attached, where it is used, how it is removed and when a fresh cover is required for the next area.
| ワークフローのステップ | 目的 | クリーンルーム管理値 |
|---|---|---|
| 新しいモップカバーを準備します | Use a clean disposable cover before cleaning a defined area | 追跡可能で管理されたツールの使用をサポート |
| 対応フレームに取り付けます | カバーを平らなモップフレームに安定した面接触で固定します。 | 洗浄の一貫性と適用範囲を向上させます |
| 担当エリアの清掃 | モップカバーは施設の清掃順序に従って使用してください。 | エリアゾーニングとSOP制御をサポート |
| 取り外して処分する | 定められた使用サイクル後に使用済みのカバーを取り外します | 再利用に関連した汚染の懸念を軽減するのに役立ちます |
| 次のエリアに交換 | 次のゾーンまたは洗浄ステップでは新しいカバーを使用してください | 管理エリア間のより明確な分離をサポート |
使い捨てのクリーンルーム モップのオプションを比較する場合は、以下の表を実際の調達ガイドとして使用してください。
| 要件 | 推奨フォーカス | なぜそれが重要なのか |
|---|---|---|
| GMP施設の清掃 | 糸くずの出にくい使い捨てモップカバー、ドキュメントサポート付き | QAレビューと汚染管理の期待をサポート |
| 無菌領域のサポート | Sterile cleanroom mop option if required by internal SOP | Helps reduce reuse-related contamination concerns in higher-control areas |
| クリーンルームの床のモップ掛け | 適度な吸水性と接触面を備えたフラットモップカバー | 洗浄範囲と湿潤の一貫性を向上させます |
| 壁や天井の掃除 | 適切なフラットフレームとハンドルに合わせた使い捨てモップカバー | 垂直および頭上の表面洗浄をサポート |
| 消毒剤の適合性 | 施設洗浄用薬品との適合性を確認 | 素材の劣化や脱落の懸念を軽減 |
| トレーサビリティ | Lot information, packaging details and supplier documentation | Supports purchasing, QA and internal approval workflows |
For B2B purchasing, disposable cleanroom mops should not be evaluated by price alone. Procurement and QA teams should also review material, packaging, compatibility, lot traceability and available supplier documentation.

大量注文を行う前に、購買チームはサプライヤーに次の詳細を確認する必要があります。
Tell us your cleanroom class, application area, mop frame size, preferred packaging format and whether sterile options are required. MIDPOSI can help recommend a suitable disposable cleanroom mop solution for your workflow.
Continue exploring MIDPOSI cleanroom mop resources for system selection, GMP facility use and B2B procurement guidance.
Learn how mop covers, frames, handles, buckets and workflows work together as a complete cleanroom mop system.
クリーンルームモップシステムを見る→Compare cleanroom mop heads and mop covers for low-lint cleaning, disposable workflows and frame compatibility.
モップヘッドガイドを見る→Learn how bucket and wringer workflows support liquid separation and saturation control.
バケットのワークフローを表示 →These answers are written for purchasing managers, QA teams, cleanroom supervisors and contamination-control buyers.
A disposable cleanroom mop is a single-use or limited-use mop cover designed for controlled environments where particles, residues and reuse-related contamination risks must be controlled.
Disposable cleanroom mops are designed for controlled environments where low-lint performance, contamination control and defined replacement workflows are important. Regular janitorial mops are not designed for GMP or ISO cleanroom cleaning requirements.
Disposable cleanroom mops are often better for reducing reuse-related contamination concerns and simplifying SOP control. Reusable mop systems may be more economical when washing, sterilization, storage and lifecycle validation are well managed.
Not always. Some facilities use reusable cleanroom mops with validated reprocessing procedures, while others prefer disposable mop systems to simplify contamination-control workflows and reduce reuse-related risk.
Yes. Disposable cleanroom mops can be used in GMP facilities when the product is suitable for the application and properly qualified according to the facility’s internal cleaning SOP and QA requirements.
No product should be described as eliminating all cross-contamination risk by itself. Disposable mop covers can help reduce reuse-related cross-contamination risk when used correctly in a defined cleanroom cleaning workflow.
No. Disposable means single-use or limited-use; it does not automatically mean sterile. If sterile supply is required, buyers should request sterile packaging and sterility-related documentation from the supplier.
Replacement frequency should be defined by the facility SOP. Common approaches include one cover per area, one cover per cleaning session, or one cover per defined zone to support contamination-control separation.
Buyers should check material, low-lint performance, frame compatibility, packaging format, disinfectant compatibility, sterility requirements, lot traceability, MOQ and available supplier documentation.
MIDPOSI supports pharmaceutical, laboratory, medical device and controlled-environment buyers with disposable cleanroom mop solutions for floor, wall, ceiling and contamination-control cleaning workflows.
MIDPOSI supports B2B buyers with disposable cleanroom mop options for GMP, ISO, pharmaceutical, laboratory, medical device, electronics and controlled-environment cleaning applications.


1営業日以内にご連絡いたします。末尾が次のメールに注意してください 「*@midposi.com」.