Sterile cleanroom mop systems are designed for Grade A/B pharmaceutical and aseptic support areas where viable contamination control, sterile transfer, particle reduction, and documentation are critical. MIDPOSI supports sterile mop cover selection for GMP cleaning workflows, including gamma-sterilized options, sterile packaging, lot traceability, and documentation support for QA review.

In aseptic and high-control pharmaceutical environments, cleaning tools can become a source of viable contamination if they are not selected, packaged, transferred, and documented correctly.
Non-sterile cleaning tools may introduce microbial contamination into sensitive cleanroom areas, especially where aseptic support workflows are involved.
Grade A/B areas often require stricter control over cleaning material transfer, handling, packaging integrity, and disposal.
QA teams may require sterility information, lot traceability, material data, packaging details, and product compatibility information before approval.
A sterile cleanroom mop is a low-lint mop cover or mop system supplied for controlled environments where sterility, particle control, and contamination prevention are required.
In GMP and pharmaceutical facilities, a sterile mop should not be evaluated only by the mop cover. Buyers should also review packaging, lot traceability, transfer procedure, compatible frame and handle, disinfectant compatibility, and available QA documentation.
Sterile mop covers may be used as single-use sterile mops, sterile-compatible flat mop systems, or part of a broader cleanroom mopping workflow depending on the facility’s SOP, cleanroom grade, and contamination-control strategy.
Sterile and non-sterile cleanroom mops serve different risk levels. The right choice depends on the cleanroom classification, facility SOP, product risk, and whether sterile supply is required.
| ਗੁਣ | Sterile Cleanroom Mop | Non-Sterile Cleanroom Mop |
|---|---|---|
| ਨਿਰਜੀਵਤਾ ਦਾ ਭਰੋਸਾ | Sterile supply, often supported by sterilization documentation | No sterility assurance |
| Common sterilization method | Gamma irradiation or validated sterile processing depending on product type | Not sterilized |
| Grade A/B suitability | Preferred for higher-control aseptic support areas | Usually not preferred for high-risk sterile areas |
| ਪੈਕੇਜਿੰਗ | Sterile or controlled packaging, often with lot traceability | Standard cleanroom packaging |
| Common use | GMP, aseptic support, pharmaceutical and biotech workflows | Routine ISO cleanroom cleaning |
| Buyer concern | Sterility, transfer process, documentation and contamination risk | Particle control, cost and routine cleaning performance |
For sterile mop covers, buyers should understand the basic sterile supply chain from production and packaging to sterilization, shipment, cleanroom transfer, and use.
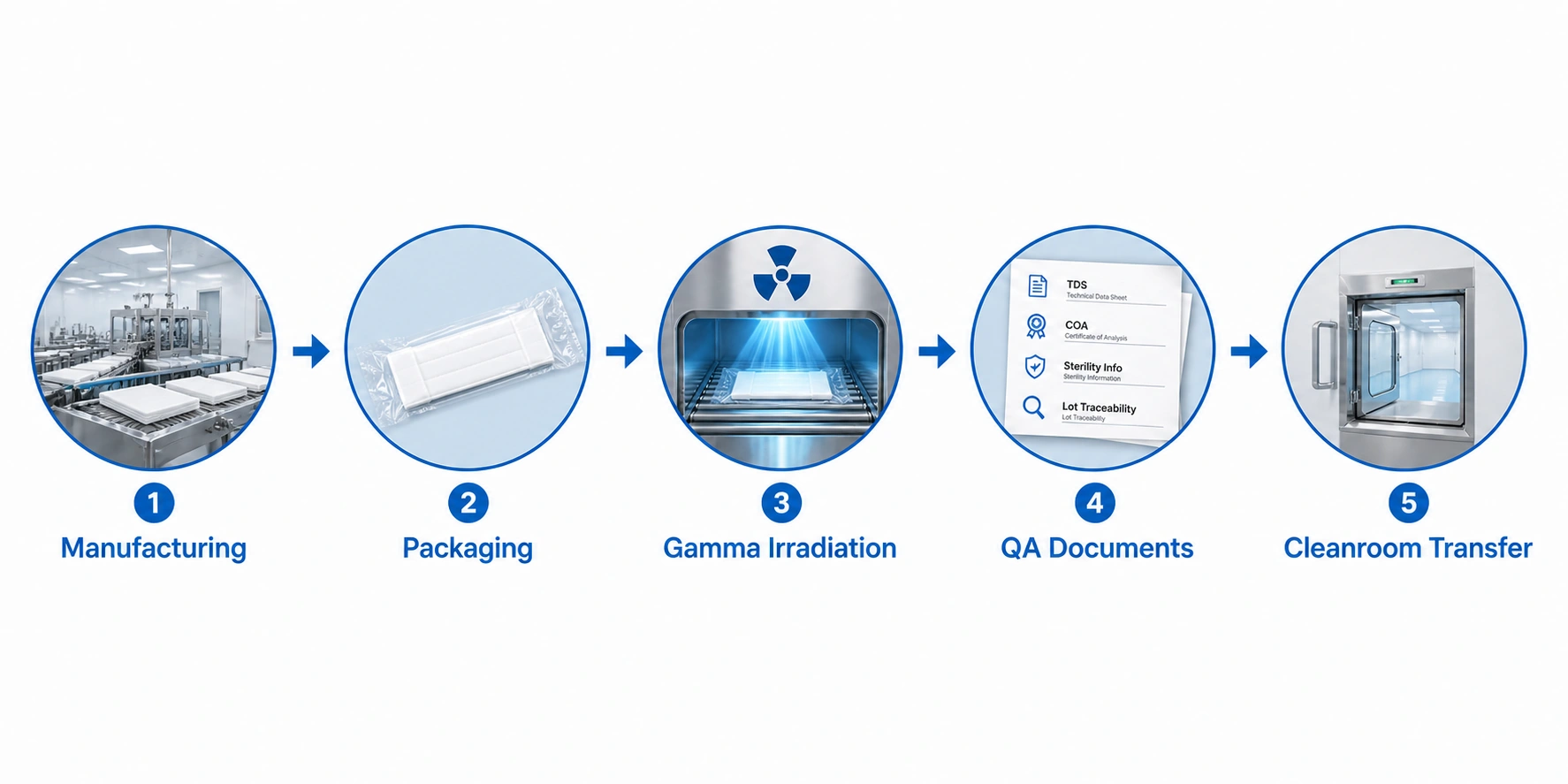
Mop cover material, edge construction, size, and cleanroom suitability are confirmed before packaging.
Packaging should support cleanroom transfer, lot traceability, and internal receiving inspection.
Gamma-sterilized products may be supplied with sterilization-related documentation for QA review.
Facilities should follow their SOP for sterile transfer, unpacking, use, disposal, or reprocessing.
Sterile cleanroom mops are most relevant where microbial control, aseptic handling, or sterile cleaning materials are required by facility procedures.
| Area / Application | Sterile Mop Recommendation | Reason |
|---|---|---|
| Grade A aseptic support | Strongly recommended if required by SOP | Higher viable contamination risk and stricter sterile workflow control |
| Grade B background area | Recommended for high-control workflows | Supports sterile material handling and contamination-control review |
| Isolator / RABS support area | Recommended where sterile cleaning tools are specified | Supports aseptic intervention control and sterile transfer procedures |
| Grade C/D manufacturing area | Case-by-case | May use sterile or non-sterile mops depending on product risk and SOP |
| Warehouse or non-critical support area | Usually optional | Non-sterile cleanroom mops may be sufficient for routine controlled cleaning |
For GMP cleaning programs where environmental control and contamination prevention are important.
For support zones around aseptic operations where sterile-compatible tools may be preferred.
For controlled surface cleaning in production and laboratory support areas.
For cleanroom areas requiring repeatable workflow, low-lint materials, and documentation.
Polyester and microfiber sterile mop covers serve different cleaning priorities. Buyers should compare lint control, residue pickup, absorption, chemical compatibility, and SOP fit before selection.

| ਗੁਣ | Polyester Sterile Mop Cover | Microfiber Sterile Mop Cover |
|---|---|---|
| Main advantage | Low-lint performance and chemical resistance | Surface contact and residue pickup |
| Typical use | Pharma and GMP controlled areas | Routine cleanroom surface cleaning |
| ਕਣ ਕੰਟਰੋਲ | Strong when properly specified | Depends on construction |
| Liquid absorption | Moderate to good | ਮਜ਼ਬੂਤ |
| Buyer concern | Lint, chemical compatibility, sterile packaging | Pickup, saturation control, material compatibility |
Sterile cleaning programs may use single-use sterile mop covers, reusable autoclavable systems, or hybrid configurations depending on risk level, cleaning frequency, and cost-control requirements.
| Option | ਲਈ ਵਧੀਆ | Key Advantage | Buyer Consideration |
|---|---|---|---|
| Single-use sterile mop | Grade A/B or high-control areas | Reduces reuse-related contamination concerns | Higher per-use cost and waste handling |
| Reusable autoclavable mop | Facilities with validated sterilization process | Lower long-term operating cost | Requires lifecycle and reprocessing control |
| Hybrid system | Reusable frame with disposable mop cover | Balances cost and contamination control | Compatibility must be confirmed |
Sterile mop sourcing often requires more than price and product photos. QA and sourcing teams may need documents that support material review, traceability, sterility review, and internal qualification.

Technical data sheet covering material, structure, size, application area, and recommended use.
Sterilization-related information may be requested for sterile or gamma-sterilized mop options.
Lot traceability helps QA teams connect product batches with receiving inspection and internal records.
Material information helps assess lint control, chemical compatibility, and SOP fit.
Packaging information helps evaluate cleanroom transfer, double-bagging needs, and handling workflow.
Certificate of analysis or certificate of conformity may be discussed depending on product and order needs.
Tell us which sterile mop option you are evaluating and whether you need TDS, material data, sterility information, lot traceability, packaging details, or sample support.
A sterile mop should not be evaluated only as a mop cover. Buyers should also confirm whether the frame, handle, and bucket workflow are compatible with their cleaning SOP and cleanroom requirements.
Low-lint sterile mop cover selected according to risk level, material preference, and cleaning area.
Compatible frame for stable floor, wall, or ceiling contact during controlled cleaning.
Smooth, cleanroom-compatible handle for reach, control, and repeatable operator movement.
Bucket or wringer workflow to support saturation control and separation of fresh and used liquid.
Sterile cleanroom mops can support GMP cleaning SOPs by improving traceability, workflow consistency, surface contact, saturation control, and documentation review.
Confirm mop material, size, packaging, lot traceability, and compatibility with the facility’s frame and handle.
Use approved disinfectants and preparation methods defined by the facility cleaning procedure.
Apply a consistent wetting level to support repeatable surface coverage and contact time.
Follow a defined cleaning direction and area-zoning procedure to reduce cross-contamination risk.
Single-use sterile mops should be disposed of according to facility requirements; reusable systems require validated reprocessing.
Buyers should choose sterile cleanroom mops by comparing cleanroom grade, sterile requirement, mop material, workflow design, chemical compatibility, and documentation needs.
Sterile cleanroom mops should be selected according to the facility’s contamination-control risk, surface type, operator workflow, and documentation requirements.
MIDPOSI can help B2B buyers compare sterile mop cover options, flat mop systems, bucket workflows, and disposable or reusable configurations.
| ਲੋੜ | Recommended Direction |
|---|---|
| Grade A/B or aseptic support area | Sterile mop cover with documentation review |
| Sterile transfer required | Sterile packaging, lot traceability, and packaging evaluation |
| Gamma-sterilized option required | Request sterilization-related information and certificate details |
| Routine ISO cleanroom | Non-sterile cleanroom mop may be enough |
| Low-lint priority | Polyester mop option |
| Strong residue pickup | Microfiber mop option |
| Floor / wall / ceiling cleaning | Flat mop system with compatible handle |
Tell us your cleanroom class, application area, sterile requirement, mop material preference, and workflow needs. MIDPOSI can help recommend a suitable sterile cleanroom mop system for your facility.
Explore related MIDPOSI resources for cleanroom mop systems, flat mop systems, disposable mop covers, microfiber mop materials, bucket workflows, and buyer guidance.
Learn how mop covers, frames, handles, buckets, and workflows work together as a complete cleanroom mop system.
View Mop System →Explore flat mop pads, frames, handles, and bucket workflows for controlled floor, wall, and ceiling cleaning.
View Flat Mop System →Learn how disposable cleanroom mop covers can reduce reuse-related contamination concerns.
View Disposable Mops →Compare microfiber mop options for residue pickup, surface contact, and cleanroom cleaning.
View Microfiber Mop →Learn how bucket and wringer workflows support liquid separation and saturation control.
View Bucket Workflow →A practical buyer guide for comparing mop materials, frames, handles, bucket workflows, and documentation needs.
Read Buyer Guide →These answers are designed for cleanroom buyers, QA teams, contamination-control specialists, pharmaceutical sourcing teams, and facility managers.
A sterile cleanroom mop is a low-lint mop cover or mop system used in controlled environments where sterility, particle control, and contamination prevention are required.
SAL 10⁻⁶ refers to a sterility assurance level where the probability of a viable microorganism remaining is extremely low. If a sterile mop is supplied with SAL-related documentation, buyers should confirm the sterilization method, lot information, and certificate details with the supplier.
Gamma-sterilized mop covers are commonly considered for Grade A/B support areas when sterile cleaning materials are required by facility SOP. Final approval should always be based on the pharmaceutical facility’s internal QA review.
Buyers should check whether the mop is supplied in sterile packaging, whether double-bagging is available, whether lot traceability is provided, and whether the packaging supports cleanroom transfer procedures.
Common documents may include TDS, material information, lot traceability, sterility-related documentation, packaging details, and product compatibility information for internal QA review.
Many sterile cleanroom mop covers are single-use, but some facilities use reusable autoclavable systems or hybrid configurations depending on SOP and contamination-control requirements.
Yes. Sterile mop covers can be used with compatible flat mop frames when the size, attachment method, and cleaning workflow are suitable for the facility’s SOP.
Polyester is often selected for low-lint performance and chemical compatibility, while microfiber is often selected for stronger surface contact and residue pickup.
If you are sourcing sterile cleanroom mops for pharmaceutical manufacturing, biotechnology facilities, laboratories, medical device production, or ISO cleanrooms, MIDPOSI can help you compare sterile mop covers, flat mop systems, disposable options, microfiber and polyester materials, and bucket-based workflows.


ਅਸੀਂ ਤੁਹਾਡੇ ਨਾਲ 1 ਕੰਮਕਾਜੀ ਦਿਨ ਦੇ ਅੰਦਰ ਸੰਪਰਕ ਕਰਾਂਗੇ, ਕਿਰਪਾ ਕਰਕੇ ਪਿਛੇਤਰ ਵਾਲੀ ਈਮੇਲ 'ਤੇ ਧਿਆਨ ਦਿਓ “*@midposi.com”.