Sammanfattning
In regulated cleanrooms, “mop vs wipe” is only half the decision. The other half is whether the tool is sterile at point of use—and if sterile, whether that sterility is supported by a validated steam autoclave process or a supplier-issued gamma irradiation certificate.
Primary decision rule: Use gamma irradiated sterile tools when you need a supplier-certified sterile claim at point of use (e.g., SAL 10⁻⁶ documentation), when materials are heat-sensitive, or when on-site autoclave capacity is limited. Use autoclavable mops when materials are heat-stable (often polyester systems) and you can validate and routinely control the steam process (cycle records + monitoring + BI program where applicable).
Cleaning execution rule: Use mops for large surfaces (floors/walls) and wipes for small or irregular equipment surfaces; most GMP programs use both.
Featured Snippet Answer
Autoclavable vs gamma irradiated cleanroom mops selection depends on (1) material compatibility, (2) sterility assurance documentation requirements (e.g., SAL-based sterile claim), (3) whether you can validate and routinely control an on-site steam process, and (4) total cost of ownership. In routine cleaning, use wipes for small critical equipment surfaces and mops for large floors and walls, often as a combined strategy.
Key Takeaways
- Tool choice is two-dimensional: surface geometry (mop vs wipe) and sterility method (autoclave vs gamma).
- Steam sterilization must be validated and routinely controlled; cycle time depends on load and sterilizer type.
- Gamma-irradiated tools rely on supplier certification, dose range control, and lot traceability.
- Hybrid programs are common: sterile wipes for critical zones and validated mop systems for large areas.
Introduktion
The choice between cleanroom mops and cleanroom wipes is a common decision point for pharmaceutical and medical device manufacturers. Both support contamination control, but their application areas, coverage efficiency, and sterility assurance pathways differ.
Most GMP facilities use both, but suboptimal selection increases cost (using wipes for large areas) or reduces effectiveness (using mops on irregular equipment surfaces). When sterile claims are required, the decision expands to autoclave-validated moist heat sterilization versus gamma irradiation with supplier certification and dose audits.

Grundläggande skillnader
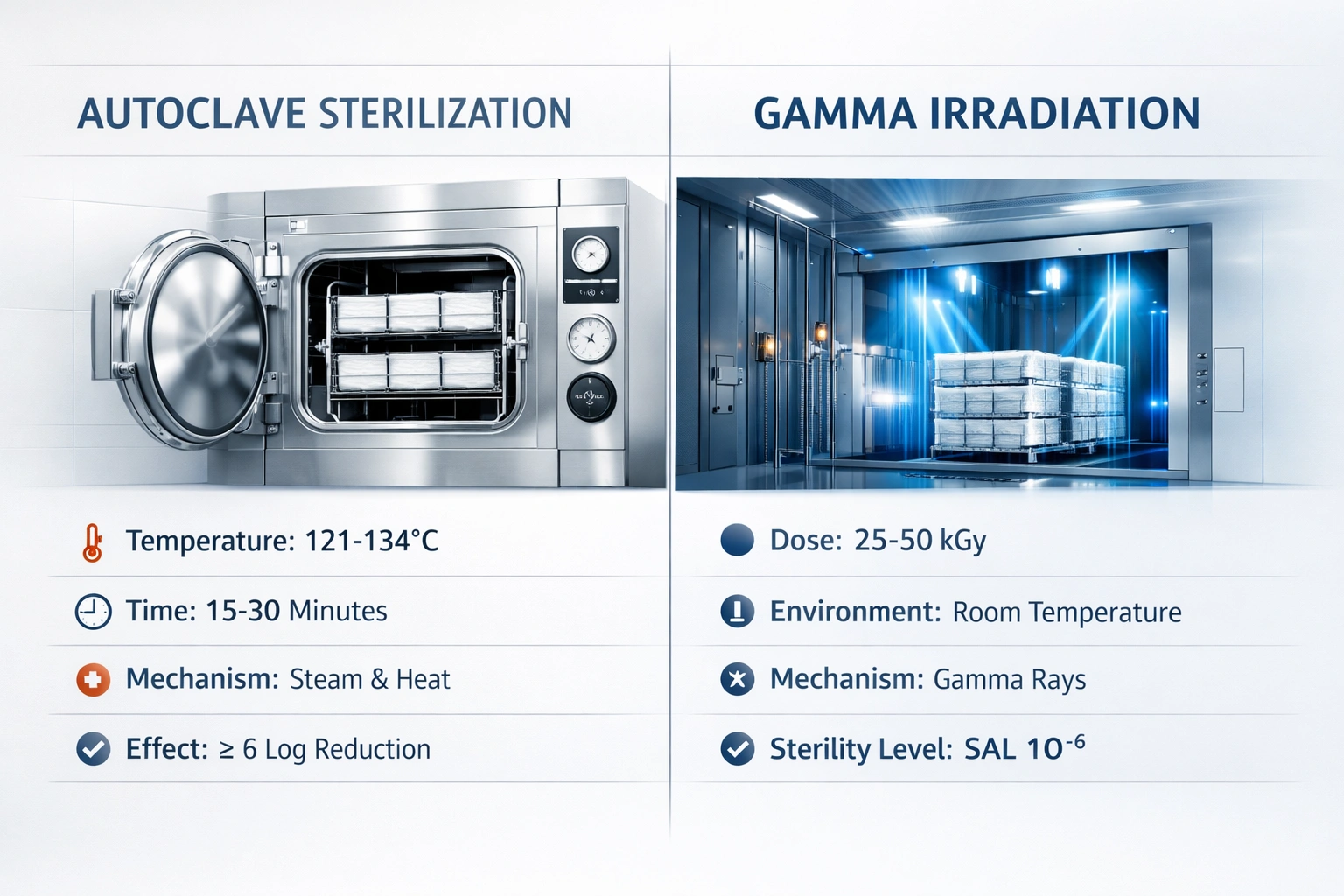
Autoclave vs Gamma Sterility Pathways
Autoclave sterilization achieves sterility through saturated steam under pressure and requires validated cycle development and routine control. Gamma sterilization uses ionizing radiation; sterile claims are supported through dose establishment/verification, dosimetry control, and routine dose audits aligned with ISO 11137 programs.
STERILIZATION METHOD CHARACTERISTICS (SUMMARY)
AUTOCLAVE (STEAM)
• Temperature: commonly 121°C or 132°C setpoints (cycle depends on sterilizer type and load)
• Exposure time: varies; minimum times depend on load/sterilizer type and must be validated
• Environment: saturated steam under pressure
• Control: mechanical + chemical indicators; biological indicators used for effectiveness monitoring as applicable
• Operational limit: chamber capacity; drying time; load configuration sensitivity
GAMMA IRRADIATION
• Sterilant: ionizing radiation (e.g., Cobalt-60 systems used industrially)
• Dose: often specified as a range; 25 kGy is a widely referenced sterilization dose, with product-specific ranges validated
• Control: dosimetry + certificate/lot traceability; ongoing dose audit expectations under ISO 11137 dose establishment programs
• Operational advantage: terminal sterilization of packaged tools at room temperature (no thermal stress)
Mopp vs Wipe egenskaper
The core cleaning-function difference is geometry and controlled contact: wipes provide direct hand pressure for full surface contact on small areas; mops provide rapid coverage for large surfaces, but contact percentage depends on mop design and technique.

MOP VS WIPE CHARACTERISTICS COMPARISON MOPS • Best for: floors, walls, large surfaces (≥ 1 m²) • Contact: typically 60–95% depending on mop type and technique • Strength: fast coverage + lower cost per square meter WIPES • Best for: equipment, workstations, spot cleaning (< 1 m²) • Contact: 100% with correct pressure application • Strength: precision + reduced cross-contamination when single-use/sterile
Core Differences Table
| Karakteristisk | Moppar | Torkar |
|---|---|---|
| Primär tillämpning | Golv, väggar, stora ytor (≥ 1 m²) | Utrustning, arbetsstationer, små ytor (< 1 m²) |
| Ytkontakt | 60–95% (depends on mop type) | 100% (with pressure application) |
| Sterility options | Autoclave validated (reusable) or sterile supplied (gamma) | Pre-sterilized (often gamma) or disinfected |
| Waste per m² | Lägre | Higher (single-use) |
Prestandajämförelse
MIDPOSI Mop-Wipe Selection Matrix
This scoring model supports consistent tool selection across surfaces and ISO grades. It can be used as part of a contamination control strategy where tool choice is justified via documented decision criteria.

MIDPOSI MOP-WIPE SELECTION MATRIX (0–100)
SCORE THRESHOLDS:
• 70–100: Highly Recommended
• 50–69: Recommended
• 30–49: Acceptable with limitations
• 0–29: Not Recommended
DECISION RULE:
Select the tool with the higher suitability score for the specific application.
Factor Scoring Criteria
| Faktor | Mop score (0–20) | Wipe score (0–20) | Scoring criteria |
|---|---|---|---|
| Områdestäckning | 0–1 m² (0) … >20 m² (20) | >10 m² (0) … <0.1 m² (20) | Match tool to area scale |
| Yttyp | Equipment (0), walls (10), floors (20) | Floors (0), walls (5), equipment (20) | Geometry and contact drive efficacy |
| ISO-klass | Risk weighting rises toward ISO 5; sterile tools may be required at point of use. | ||
Application Scenarios
| Ansökan | Område | Yta | Mopp poäng | Torka poäng | Rekommenderad |
|---|---|---|---|---|---|
| ISO 5 arbetsstation för aseptisk zon | 0,5 m² | Utrustning | 6.5 | 78,5 | Torka |
| ISO 7 corridor floor (daily) | 50 m² | Golv | 87,5 | 22.5 | Mopp |
Applikationsspecifika rekommendationer
ISO 5 Critical Aseptic Zone
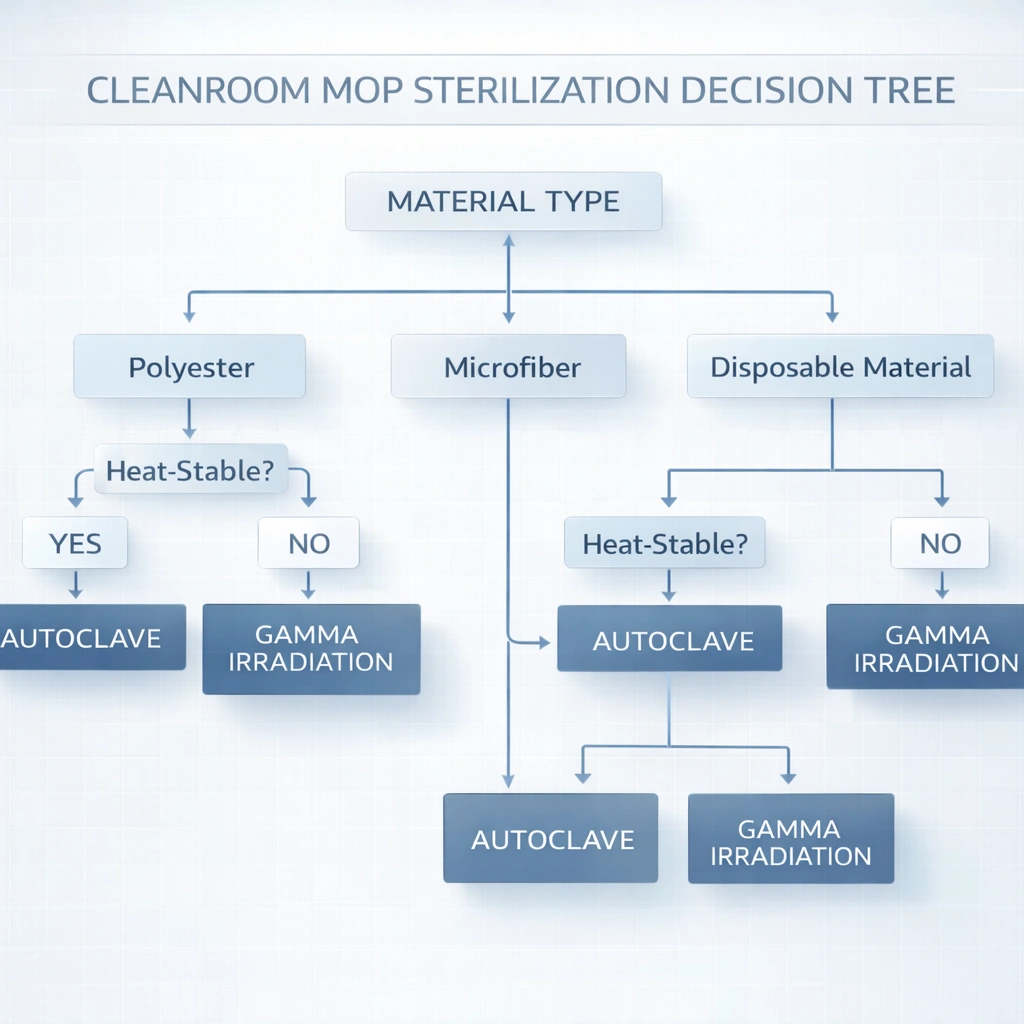
ISO 5 workflows emphasize precision cleaning and sterility assurance. Wipes are typically primary for critical equipment surfaces, while sterile mops may be used for defined floor/perimeter tasks as dictated by the site’s contamination control strategy.
ISO 7 Clean Areas

ISO 7 often supports validated reusable mop systems coupled with disinfected or sterile wipes for equipment surfaces, pass-throughs, and frequent-touch points.
Materialval
Sterilization Standards Table
For regulated decision-making, cite and align to the standards that match the sterilization method: radiation programs to ISO 11137, moist heat programs to ISO 17665, and EU sterile manufacturing expectations to Annex 1 and compendial chapters such as Ph. Eur. 5.1.1/5.1.2.
| Reference | Method scope | What it governs | Typical evidence in audits |
|---|---|---|---|
| ISO 11137-2 | Radiation | Dose setting/substantiation for sterile claims and ongoing dose audits | Certificate of irradiation, dose range controls, lot traceability, audit evidence |
| ISO 17665 | Moist heat (steam) | Process development, validation, and routine control for steam sterilization | Cycle parameters, load configuration, monitoring records, BI program evidence where applicable |
| Ph. Eur. 5.1.1 / 5.1.2 | Compendial sterile manufacturing expectations | Validated processes and biological indicator (BI) suitability/rigor | Validation strategy and BI suitability documentation aligned to process |
| EU GMP bilaga 1 | Sterile manufacture | Contamination control strategy and sterility assurance philosophy | CCS, validated procedures, monitoring/trending, deviation/CAPA integration |

Wipe Material Types

Validation note: radiation dose is typically specified as a range (not a single point), and maximum dose must be justified against material performance. For any “25–50 kGy” specification, validate that your tool materials and packaging tolerate the maximum dose without unacceptable degradation.
Kostnadsanalys
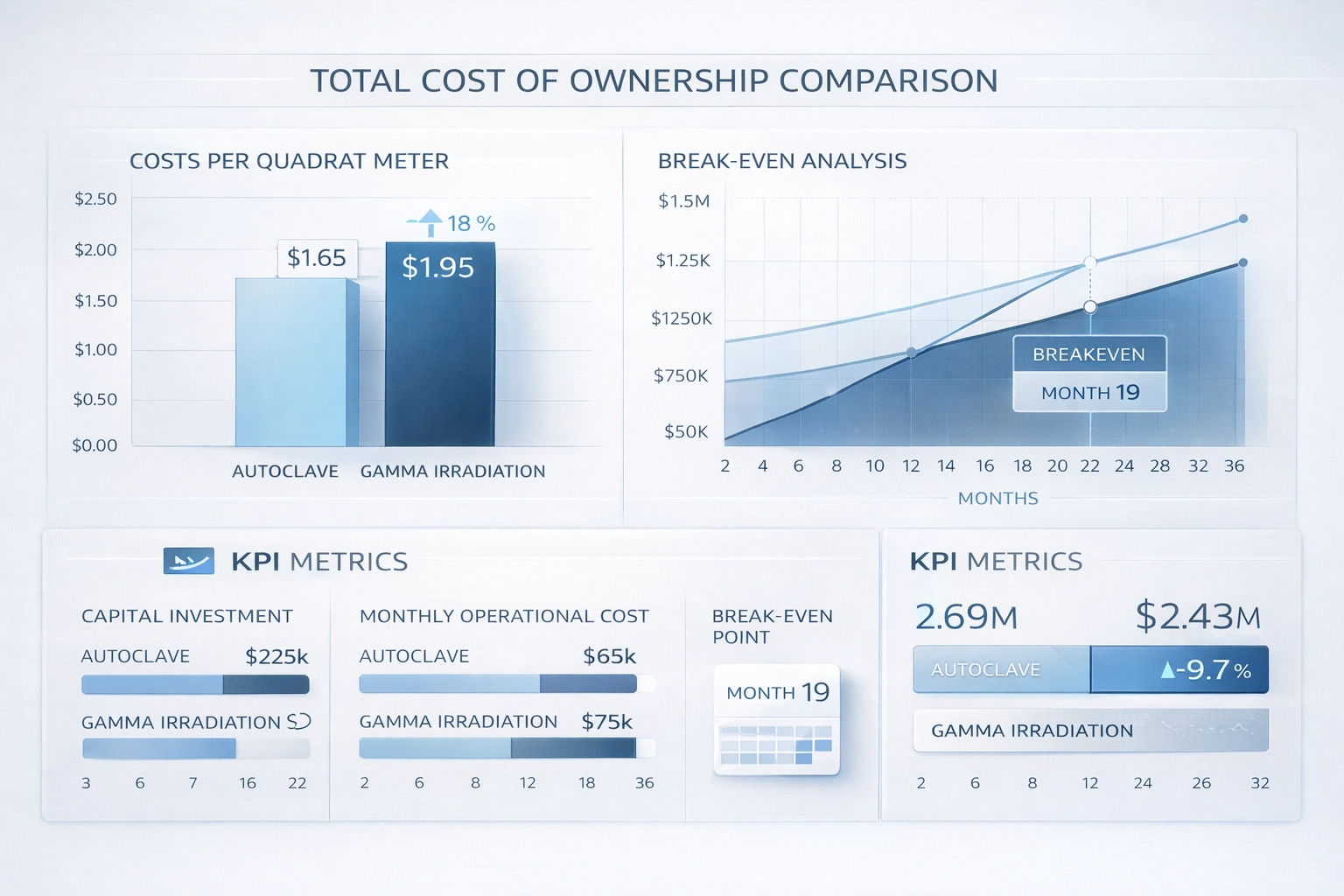
Kostnad per m² Jämförelse
COST PER m² COMPARISON (EXAMPLE MODEL)
ASSUMPTIONS
• Labor cost: $25/hour
• Mop head cost: $5 (reusable 10 times) = $0.50/use
• Wipe cost: $0.30/wipe (single-use)
• Mop covers 10 m²/use, wipe covers 0.5 m²/use
• Labor: mop 0.5 min/m², wipe 0.25 min/m²
CONCLUSION (MODEL OUTPUT)
Mop is ~4–5× more cost-effective than wipes for large surfaces.
Sterilization Cost Framework Table
Cost decision rule (sterile tools): If the fully loaded gamma program cost (including supplier qualification, incoming verification, and waste) is >3× the validated autoclave total cost, autoclave is typically the economic choice—provided the material is heat-stable and cycle capacity is available.
| Kostnadskategori | Autoclave program | Gamma program | Notes |
|---|---|---|---|
| Tool unit cost | $_____ (reusable) | $_____ (often single-use) | Include cycle life for reusables |
| Steriliseringskostnad | $_____ per cycle | $_____ per lot | Gamma typically managed via supplier certificate + incoming verification |
| Arbetskraft & handling | $_____ | $_____ | Includes staging, labeling, quarantine, release |
| Documentation burden | Cycle record + BI/monitoring program | Certificate of irradiation + lot traceability | Both require QA review and change control |
Bästa metoder
Mop Technique

Wipe Folding Technique
Common Mistakes to Avoid
Mistake: Treating supplier sterility certificates as “enough” without incoming verification and lot traceability controls.
Mistake: Overloading autoclaves or changing load configuration without reassessing cycle effectiveness.
Mistake: Using a tool material that is not proven compatible with your maximum gamma dose or repeated steam cycles.
Praktiska verktyg
Tool: Mop-Wipe Selection Decision Matrix
MOP-WIPE SELECTION DECISION MATRIX (TEMPLATE)
Application: ______________________ ISO Grade: ________ Area: __________ m²
Score each factor (0–20)
• Area Coverage: Mop ___ / Wipe ___
• Labor Efficiency: Mop ___ / Wipe ___
• Surface Type: Mop ___ / Wipe ___
• ISO Grade: Mop ___ / Wipe ___
• Cost Efficiency: Mop ___ / Wipe ___
Decision: Select the higher total score tool.
Assessors: _______________________ Date: _________________
Approver: ________________________ Date: _________________
Tool: Autoclave Sterilization Protocol
AUTOCLAVE STERILIZATION PROTOCOL (CHECKLIST)
PRE-CYCLE
☐ Mop inspected for integrity
☐ Cleaned per SOP
☐ Packaging verified (no damage)
☐ Sterilization label attached (tool ID, cycle ID)
☐ Load configuration documented
☐ Monitoring / BI plan per site validation strategy
CYCLE
☐ Temperature setpoint: ________
☐ Exposure time: ________
☐ Drying time: ________
☐ Cycle record saved (time/temperature/pressure)
☐ Any alarms investigated and resolved
POST-CYCLE
☐ Quarantine / release process followed (QA as applicable)
☐ Storage conditions maintained
☐ FIFO rotation implemented
☐ Traceability log updated
Tool: Gamma Irradiation Verification Protocol
GAMMA IRRADIATION VERIFICATION (RECEIVING CHECKLIST)
RECEIVING
☐ Supplier certificate verified
☐ Dose stated: _______ kGy (within validated range)
☐ SAL 10⁻6 sterility claim verified if required for use
☐ Lot number recorded
☐ Packaging integrity confirmed (no breach)
☐ Receipt and release logged (QA controls)
USAGE
☐ FIFO rotation maintained
☐ Expired tools not used
☐ Zone segregation (color-code / controls)
☐ Disposal documented and traceable

FAQ
När ska jag använda en mopp kontra en torkduk?
Use a mop when the surface area is ≥ 1 m² (floors/walls) and geometry is regular. Use wipes when the area is < 1 m², surfaces are irregular (equipment/workstations), or when single-use sterility at point of use is required.
Kan jag använda våtservetter för att rengöra golv?
Generally not for routine large-area floor cleaning due to cost and efficiency; use wipes for spot cleaning, corners, edges, or critical zones where the workflow requires sterile precision use.
Vilken är den mest kostnadseffektiva metoden för blandade applikationer?
A hybrid approach: mops for floors and large surfaces, wipes for equipment and small surfaces; sterile tools reserved for the highest-risk zones.
Hur väljer jag moppmaterial?
Match material to ISO grade, disinfectant chemistry, and sterilization method. Verify compatibility with repeated steam cycles or validated gamma dose ranges, and require documentation that supports low-lint and intended use.
Hur ofta ska moppar bytas?
Replace reusable mops after their validated cycle life (or sooner if damage, increased lint, or reduced absorbency appears). Disposable/sterile tools are typically single-use unless a validated reprocessing program exists.
Är förmättade våtservetter värda den extra kostnaden?
Often yes in critical zones because they standardize application, reduce prep variability, and support sterility assurance when supplied sterile.
Kan jag återanvända våtservetter?
Single-use wipes should not be reused. Reusable wipe programs require validated laundering/sterilization and clear traceability controls.
Hur verifierar jag prestanda med låg ludd?
Require supplier evidence and verify via your facility’s monitoring strategy. For critical grades, ensure the evidence aligns with your room classification and operational state. Document acceptance criteria in your SOP.
What is SAL 10⁻⁶ and why does it matter for gamma irradiated tools?
SAL (sterility assurance level) is the statistical level used for sterile claims; gamma programs commonly support SAL-based claims via validated dose establishment and ongoing dose audits, with certificates tied to lot traceability.
What documentation should I keep for audits?
For autoclave: cycle records, monitoring results, load configuration controls, and release documentation. For gamma: certificate of irradiation per lot, defined dose range, incoming verification, storage/expiry controls, and usage/disposal traceability.
Interna länkar
For related guidance and implementation resources:
Preferred Standards and References
Use official copies of standards/compendia as primary sources for validation and audit defense. Public reference links:
- ISO 11137-2: Radiation sterilization dose establishment
- ISO 17665-1: Moist heat sterilization process requirements (withdrawn; points to ISO 17665:2024)
- EDQM overview of Ph. Eur. 5.1.1 & 5.1.2
- CDC: Steam sterilization overview and monitoring
- CDC: Minimum cycle times table (steam)
- EU GMP Annex 1 (2022): Manufacture of sterile medicinal products



