When a pharmaceutical QA manager traces an environmental monitoring failure back to the cleaning tools, it’s rarely a surprise anymore. Industry investigations consistently link 30–60% of EM excursions to contamination introduced by cleaning equipment—and the culprit is almost always particle shedding. For facilities operating under ISO 14644-1 and EU GMP Annex 1, the choice between standard and low-lint cleanroom mops isn’t a matter of preference. It’s a documented, validated control point in your Contamination Control Strategy that determines whether your classified areas stay in spec or trigger OOS investigations, production holds, and regulatory findings.
A low lint cleanroom mop is not simply a “better” mop. It is a contamination-control device engineered to generate orders of magnitude fewer particles than conventional cleaning tools, validated through quantitative particle release testing, and selected to match the specific particle count limits and GMP requirements of your cleanroom grade. This guide explains what “low lint” means in regulatory terms, why it matters for compliance, which materials deliver validated performance, and how to specify the right mop system for each area in your facility.
What Does “Low Lint” Mean in Cleanrooms?
In cleanroom terminology, “lint” refers to fibrous particles and non-fibrous debris released from textiles during use. These particles range from visible fiber fragments (tens to hundreds of micrometers) down to sub-visible particulates below 5 µm that are captured only by optical particle counters. ISO 14644-1 classifies cleanrooms by measured airborne particle concentration at specific size thresholds—primarily ≥0.5 µm and ≥5 µm. A cleaning tool that sheds even modest quantities of lint can generate enough particles to push a classified environment out of specification, especially in ISO Class 5–7 areas where limits are tightly controlled.
“Low lint” is not a marketing descriptor—it is a quantified performance characteristic validated through standardized particle release testing. ISO 9073-10 defines a dry-state linting test for nonwoven textiles, while the Helmke Drum test (per IEST-RP-CC003.4) measures whole-garment or textile particle emission under simulated-use conditions. For renrumsmopper, particle generation testing typically follows ISO 14644-14 (assessment of equipment suitability by airborne particle concentration), where mop heads are subjected to mechanical agitation while airborne particle counters measure release rates.
Acceptable low-lint mops demonstrate particle generation well below the classification limits of their intended use area:
ISO Class 5 (3,520 particles/m³ at ≥0.5 µm; 29 particles/m³ at ≥5 µm): Mops must generate <10 particles per stroke to avoid measurable contribution to particle burden. ISO Class 6 (35,200 particles/m³ at ≥0.5 µm; 293 particles/m³ at ≥5 µm): Acceptable mops generate <50 particles per stroke. ISO Class 7 (352,000 particles/m³ at ≥0.5 µm; 2,930 particles/m³ at ≥5 µm): Mops generating <100 particles per stroke are typically qualified. ISO Class 8 (3,520,000 particles/m³ at ≥0.5 µm; 29,300 particles/m³ at ≥5 µm): Higher generation may be tolerable, but validated low-lint mops remain best practice to prevent particle trends.
- ISO Class 5 (3,520 particles/m³ at ≥0.5 µm; 29 particles/m³ at ≥5 µm): Mops must generate <10 particles per stroke to avoid measurable contribution to particle burden.
- ISO Class 6 (35,200 particles/m³ at ≥0.5 µm; 293 particles/m³ at ≥5 µm): Acceptable mops generate <50 particles per stroke.
- ISO Class 7 (352,000 particles/m³ at ≥0.5 µm; 2,930 particles/m³ at ≥5 µm): Mops generating <100 particles per stroke are typically qualified.
- ISO Class 8 (3,520,000 particles/m³ at ≥0.5 µm; 29,300 particles/m³ at ≥5 µm): Higher generation may be tolerable, but validated low-lint mops remain best practice to prevent particle trends.
By contrast, conventional janitorial mops (cotton, rayon, looped blends) shed thousands of particles per stroke. The gap is not incremental—it is structural, determined by fiber type, edge construction, and material processing.
Why Mops Are Common Lint Sources
Cleaning tools experience high mechanical stress during use—friction against floors, repeated wetting and wringing, exposure to aggressive disinfectants. For reusable systems, autoclaving or laundering cycles degrade fibers over time. Cut edges on conventional mops unravel with each use, releasing fiber ends into the cleanroom. Loosely twisted or looped fibers break under abrasion, generating both fibrous lint and microparticles from fiber fragmentation. Foam-core mops with unvalidated substrates can shed foam particles as the material degrades under chemical or thermal stress.
Engineering solves these failure modes. Continuous-filament fibers eliminate cut ends. Heat-sealed or ultrasonically welded edges prevent unraveling. Tight knit or weave density minimizes fiber pull-out. Materials are validated for chemical and autoclave resistance across hundreds of cycles. These design features, combined with particle generation testing per ISO 14644-14, provide documented evidence that the mop will not contribute measurable contamination during routine use—a requirement explicitly supported by EU GMP Annex 1’s mandate to minimize particle shedding from materials and equipment used in classified areas.

Figure 1: Particle generation comparison under simulated-use conditions. Conventional cut-edge mop (left panel) releases hundreds of fiber fragments and particulates per stroke, violating ISO Class 5–7 particle limits. Low-lint sealed-edge mop (right panel) generates <50 particles ≥0.5 µm per stroke through continuous-filament construction and heat-sealed edges, maintaining classification compliance across 100+ autoclave cycles.
Why Low-Lint Mops Are Critical for GMP Workflows
EU GMP Annex 1 (2022 revision) mandates a facility-wide Contamination Control Strategy that identifies and controls microbial, particulate, and endotoxin contamination through design, procedures, and equipment selection. Cleaning and disinfection programs must be validated, and materials used in classified areas must minimize particle generation and permit repeated application of disinfectants and sporicidal agents. Low-lint mops are not optional accessories in this framework—they are critical control points that directly affect whether your facility maintains ISO classification, passes environmental monitoring, and survives regulatory inspection.
Annex 1 and the “Visible and Non-Visible Contamination” Requirement
Annex 1 explicitly addresses both visible and sub-visible particulate contamination as risks to be controlled through the CCS. Cleanroom classification by total particles (≥0.5 µm and ≥5 µm) establishes the performance envelope; any equipment or material introduced into these areas must not compromise classification limits. Paragraph 4.10 requires materials used in cleanrooms be selected to minimize particle generation. Paragraph 4.28 mandates cleaning processes be validated to remove residues and minimize particulate contamination.
When a standard, particle-shedding mop is used in an ISO Class 6 or Class 7 support area, it introduces a contamination vector that undermines your entire environmental monitoring program. Each mopping pass releases fibers and particles that settle on equipment, walls, and floors—creating both immediate particle count excursions and long-term bioburden risks as those particles harbor microbial contamination. During regulatory inspections, auditors examine cleaning validation protocols, equipment qualification records, and EM trends. A failure to qualify cleaning tools—or worse, the use of visibly fraying, particle-generating mops—has been cited in FDA Warning Letters. One 2020 enforcement action specifically noted “fraying observed on mop heads used to clean the sterile suite” and “at least one mop head constructed of particle generating material,” with FDA concluding that the facility lacked adequate controls for cleaning equipment evaluation.
Why 30–60% of EM Failures Trace to Cleaning Tools
Environmental monitoring excursions trigger investigations, and those investigations often point back to cleaning operations. While published industry-wide statistics are limited, facility-level data consistently show that 30–60% of particulate or microbial EM failures can be traced to inadequate cleaning tool qualification, improper tool maintenance, or cross-contamination from non-sterile cleaning materials. The mechanism is straightforward: high-lint mops release particles during mopping; those particles settle; subsequent air and surface sampling detect the contamination; investigations reveal that the mop head was not validated, was used beyond its qualified life, or was incompatible with the disinfectant chemistry.
This problem is especially acute in ISO 5–7 production areas (EU GMP Grade B/C equivalents) where aseptic processing support activities—material staging, gowning, equipment prep—occur. These areas operate close to their particle limits by design. Introducing a tool that sheds even 200–300 particles per stroke can push a Class 6 environment (35,200 particles/m³ limit at ≥0.5 µm) into excursion during active cleaning or immediately afterward. The contamination is invisible to the operator but captured by continuous particle monitoring and routine surface/air sampling. The result: production holds, batch investigations, and corrective actions that could have been prevented by specifying validated low-lint mops in the first place.
Cleanrooms and the “Hidden Contamination Vector” Problem
Cleaning tools move through every room in your facility. A mop used in a Grade D material airlock may later be transferred (improperly) into a Grade C corridor, carrying particles or bioburden with it. Even within a single area, repeated use degrades mop fibers—edges fray, sealed seams separate, and foam cores crumble—transforming a qualified low-lint mop into a particle generator unless inspection and replacement protocols are enforced.
Low-lint mops, when properly validated and maintained, break this contamination cycle. Their particle generation remains stable across 100+ autoclave cycles (for reusable systems) or is eliminated entirely (for pre-sterilized disposables). Material certifications and lot traceability allow QA teams to track tool performance, identify degradation trends, and retire mops before they become contamination sources. This level of control is not achievable with standard janitorial equipment, which is why GMP facilities under Annex 1 and FDA oversight treat low-lint mops as regulated contamination-control devices rather than cleaning supplies.
B2B Decision Point: Reducing Batch Risk and Passing Audits
For procurement managers and QA directors, the value proposition of low-lint cleanroom mops is quantifiable: each prevented EM excursion avoids investigation costs (often $10,000–$50,000 in labor, testing, and documentation), eliminates production delays, and reduces the risk of regulatory findings that can lead to consent decrees or import alerts. A facility that specifies validated low-lint mops, maintains qualification records, and enforces replacement schedules demonstrates systematic contamination control—precisely what auditors expect to see during GMP inspections. The incremental cost of low-lint mops (typically 20–50% higher than standard mops) is negligible compared to the risk-adjusted cost of a single batch contamination event or a failed inspection.
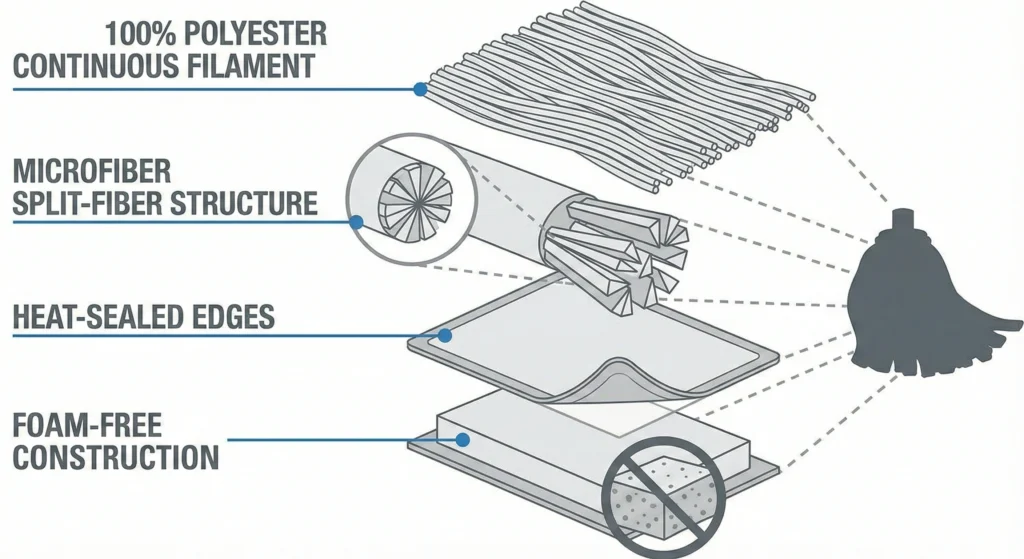
Types of Low-Lint Mop Materials
Low-lint performance is achieved through material selection, fiber construction, and edge sealing technology. Four primary material categories dominate the cleanroom mop market, each offering distinct trade-offs in absorbency, chemical resistance, durability, and particle generation.
100% Polyester Sealed-Edge Mops
Polyester knit mops constructed from continuous-filament 100% polyester yarns represent the industry standard for low-lint, high-durability cleanroom cleaning. Edges are heat-sealed or ultrasonically welded to prevent unraveling, and the tight knit structure minimizes fiber pull-out during use. Polyester is chemically inert, tolerating repeated exposure to isopropyl alcohol (70% IPA), hydrogen peroxide (3–35%), quaternary ammonium compounds, and sodium hypochlorite (bleach) at concentrations up to 1% without significant fiber degradation or color loss.
Particle generation testing per ISO 14644-14 typically shows <50 particles ≥0.5 µm per stroke for qualified polyester sealed-edge mops, supporting use in ISO Class 5–8 environments. Autoclave stability is excellent: validated products maintain dimensional stability and low-lint performance across 150–200+ sterilization cycles at 121°C. Polyester knit mops are the workhorse choice for reusable cleanroom mop programs, offering the best balance of chemical resistance, durability, and total cost of ownership for facilities with validated laundering and sterilization infrastructure.
Microfiber Low-Lint Mops (Split-Fiber Technology)
Microfiber mops use ultra-fine synthetic fibers—typically polyester/polyamide blends with diameters below 1 denier (roughly 10 micrometers)—engineered in a split-fiber construction that creates microscopic channels for particle capture. The high surface-area-to-weight ratio delivers superior absorbency (often 6–8 times the mop’s dry weight) and excellent particle trapping efficiency, making microfiber mops effective for both wet mopping with disinfectants and damp dusting to remove residual particles between disinfection cycles.
When properly constructed with sealed edges and continuous-filament fibers, microfiber mops achieve low-lint performance comparable to polyester knit systems, generating <50 particles per stroke in validation testing. However, microfiber’s split-fiber structure can be vulnerable to certain disinfectant chemistries: repeated exposure to high-concentration bleach (>1%) may cause fiber splitting to progress into fiber damage, increasing particle shedding over time. For this reason, microfiber mops are best suited to facilities using IPA, quats, and hydrogen peroxide as primary disinfectants, with bleach reserved for periodic sporicidal treatment rather than daily use.
Microfiber mops require validated laundering protocols. Improper washing—high heat, fabric softeners, or contaminated laundry equipment—can introduce particles or residues that compromise cleanroom performance. Many facilities mitigate this risk by using pre-sterilized disposable microfiber mops, which eliminate laundry complexity while preserving microfiber’s high absorbency and particle capture advantages. For a detailed comparison of microfiber vs polyester cleanroom mops, see our material selection guide.
Heat-Sealed, Laser-Cut Edge vs Cut-Edge Construction
Edge construction is the single most important factor determining whether a mop qualifies as “low lint.” Cut edges—where fabric is simply sheared and left unsealed—unravel with each use, releasing fiber ends into the cleanroom. Even a single cut edge on an otherwise high-quality mop head can generate hundreds of particles per mopping pass as the edge frays.
Heat-sealed edges use thermal welding to melt and fuse fiber ends, creating a solid border that prevents unraveling. Ultrasonic welding achieves similar results through high-frequency vibration, bonding fibers without introducing adhesives or foreign materials. Laser-cut edges combine precision cutting with edge sealing in a single step, delivering clean, particle-free borders suitable for ISO Class 5 applications. Qualified low-lint mops specify edge sealing method in their product documentation and provide particle generation data demonstrating that sealed edges deliver 60–80% lint reduction compared to cut-edge mops of equivalent material.
Foam-Free vs Foam-Core Mops

Figure 2: Edge construction determines lint generation. Cut-edge mops (left) unravel during use, releasing fiber ends into cleanrooms. Heat-sealed edges (center) use thermal welding to fuse fiber ends, creating particle-free borders. Ultrasonic-welded edges (right) bond fibers through high-frequency vibration without adhesives, delivering 60–80% lint reduction compared to cut-edge construction. Qualified low-lint mops specify edge sealing method in product documentation.
Foam-core mops incorporate a central foam substrate encased in a textile outer layer. The foam core provides structural rigidity and even fluid distribution across the mop head, which can be advantageous for applying sporicidal agents uniformly to validated surfaces. However, foam substrates vary widely in chemical resistance; not all foam formulations tolerate repeated exposure to bleach, hydrogen peroxide, or high-temperature autoclaving without degradation (crumbling, discoloration, loss of structural integrity). A degrading foam core becomes a particle source, shedding foam fragments that contaminate the cleanroom.
Foam-free mops eliminate this risk entirely. Constructed from continuous-filament textile throughout (polyester or microfiber), foam-free designs rely on fiber density and knit structure to achieve absorbency and structural integrity. For facilities using aggressive disinfectant programs or requiring 100+ autoclave cycles, foam-free construction is the safer, more predictable choice. When foam-core mops are specified, vendors must provide chemical compatibility data, autoclave validation (cycles to failure), and particle generation testing after aging to demonstrate that the foam substrate remains stable under your actual use conditions.
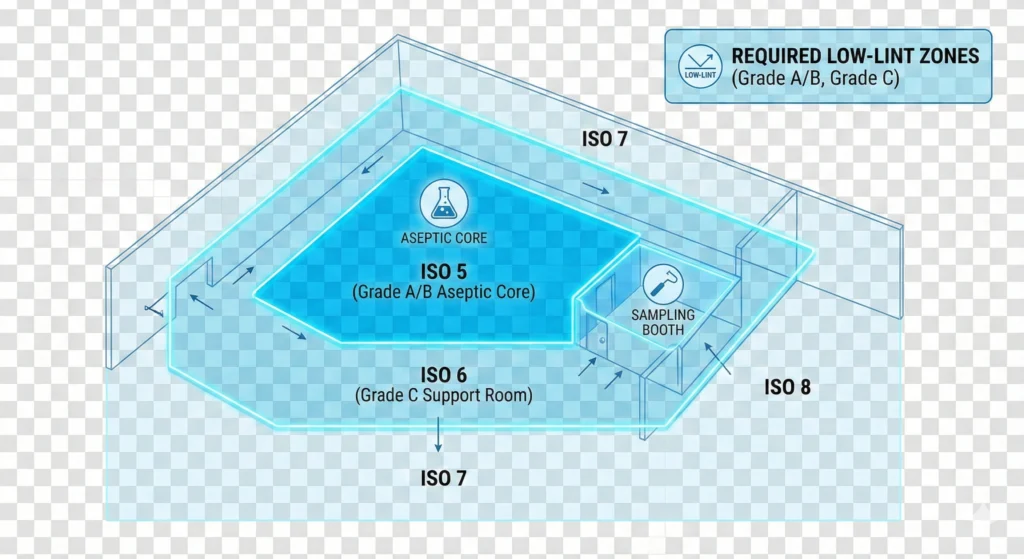
Which Areas Require Low-Lint Cleanroom Mops
Not every area in a pharmaceutical or biotech facility demands the same level of lint control. Your Contamination Control Strategy should define area-specific requirements based on ISO classification, GMP grade, product contact risk, and sterility assurance needs. The following framework guides procurement and QA teams in making defensible, risk-based decisions.
Grade A/B Aseptic Processing (ISO Class 5): Mandatory
EU GMP Grade A and Grade B environments—aseptic filling lines, lyophilization loading, vial capping under unidirectional airflow—are the most demanding cleanroom applications. Particle limits are extreme (3,520 particles/m³ at ≥0.5 µm for ISO Class 5), sterility is non-negotiable, and any contamination event can compromise entire production batches. Low-lint mops in these areas must be:
- Sterile: Either pre-sterilized disposables (gamma-irradiated, individually packaged with sterility certificates) or reusable mops autoclaved immediately before use.
- Ultra-low shedding: Particle generation <10 particles per stroke, validated per ISO 14644-14.
- Material transfer controlled: Mops must enter Grade A/B areas through validated sterilization or transfer disinfection procedures (double-ended autoclaves, sterile pass-throughs) per Annex 1 paragraph 4.22.
In practice, most facilities use pre-sterilized disposable mops for Grade A/B cores to eliminate reprocessing risk and simplify material flow. These mops are often supplied pre-saturated with sterile 70% IPA in double-pouch packaging (outer pouch removed in Grade C airlock; inner sterile pouch opened within Grade A/B area). The incremental cost is justified by the elimination of cross-contamination risk and the removal of autoclave reprocessing as a variable in contamination investigations.
ISO 5 Sampling & Weighing Rooms: High Requirement
Material sampling booths, active pharmaceutical ingredient (API) weighing rooms, and compounding suites classified as ISO Class 5 operate under the same particle limits as Grade A/B areas but may handle non-sterile materials or intermediates where sterility is not yet required. Low-lint mops are still mandatory, but the sterility requirement may be relaxed based on CCS risk assessment:
- Reusable low-lint mops (polyester sealed-edge or microfiber) with validated laundering and disinfection protocols are acceptable if the CCS justifies that sterility is not required for the specific operation.
- Partikelgenerering must still be <10–20 particles per stroke to maintain ISO Class 5 classification.
- Disinfectant compatibility with high-concentration IPA, hydrogen peroxide, and sporicidal agents is required, as these areas undergo frequent bio-decontamination.
For risk-averse facilities or those with limited laundering infrastructure, pre-sterilized disposable mops remain a pragmatic choice even when sterility is not strictly required, as they eliminate the qualification complexity of reusable systems.
Grade C/D Manufacturing (ISO Class 7/8): Recommended but Flexible
Grade C aseptic processing support areas (ISO Class 7) and Grade D final packaging or non-sterile compounding areas (ISO Class 8) operate under relaxed particle limits (352,000 and 3,520,000 particles/m³ at ≥0.5 µm, respectively). Standard high-quality cleaning mops may technically meet classification limits in these areas. However, low-lint mops remain best practice for several reasons:
- Förebyggande av kontaminering: Particles shed in Grade C/D areas can be carried into higher-grade zones via personnel gowning, material transfer, or airflow patterns, seeding contamination that manifests as EM excursions in Grade A/B areas.
- EM trendstabilitet: Using validated low-lint mops facility-wide simplifies environmental monitoring programs, reduces baseline particle counts, and makes it easier to detect true contamination events versus tool-related noise.
- Regulatory expectation alignment: Auditors expect systematic contamination control, not a patchwork of qualified and non-qualified equipment. Specifying low-lint mops across all classified areas demonstrates a mature CCS.
For budget-conscious procurement, a pragmatic compromise is to use reusable low-lint mops (polyester sealed-edge, validated for 150+ autoclave cycles) in Grade C/D areas while reserving pre-sterilized disposables for Grade A/B cores.
Support Areas, Manlocks, and Cross-Contamination Risk
Gowning rooms, personnel airlocks (manlocks), and material staging areas may be classified as ISO Class 8 or lower. While particle limits are lenient, these areas serve as contamination control buffers that protect higher-grade zones. Using low-lint mops in support areas prevents particle accumulation on gowning benches, airlock surfaces, and transfer carts that could be carried into production areas by personnel or materials.
A critical CCS control: color-coded mop segregation. Mops used in Grade D support areas must never enter Grade A/B zones. Color-coding (e.g., blue mops for production areas, red for waste handling, green for gowning rooms) combined with physical segregation (separate storage, documented procedures, operator training) prevents cross-contamination. Low-lint mops should be specified even for lower-grade areas to maintain system-wide contamination control and simplify training (one standard: all mops are low-lint and sealed-edge, regardless of area).
B2B Decision Framework: “Must Have, Recommended, Optional”
For procurement teams building mop specifications:
- Must have (non-negotiable): Grade A/B aseptic cores, ISO Class 5 sampling/weighing rooms—pre-sterilized disposable or autoclaved reusable, <10 particles/stroke.
- Strongly recommended (risk reduction): Grade C support areas (ISO Class 7), high-value or high-risk manufacturing zones—reusable low-lint with validated laundering, <50 particles/stroke.
- Recommended (best practice): Grade D packaging/compounding (ISO Class 8), support areas, manlocks—reusable low-lint, <100 particles/stroke, or accept standard cleanroom mops if CCS risk assessment justifies.
- Optional (context-dependent): Non-classified warehouse, office areas adjacent to cleanrooms—standard cleanroom mops acceptable, but low-lint preferred to prevent particle migration.
This tiered framework balances contamination control with cost, allowing facilities to allocate budget to the highest-risk areas while maintaining defensible, audit-ready specifications across the facility.
Testing & Validation Standards for Low-Lint Mops
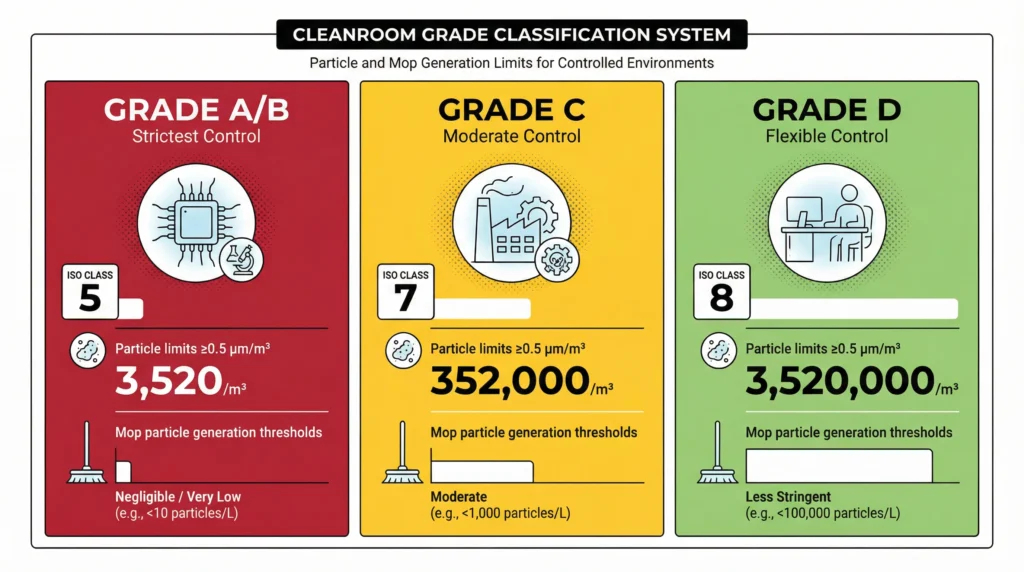
Figure 3: Area classification determines low-lint mop requirements. Grade A/B (ISO Class 5) aseptic cores demand sterile mops with <10 particles/stroke—non-negotiable for regulatory compliance. Grade C (ISO Class 7) support areas require validated low-lint mops with <50 particles/stroke to prevent contamination migration. Grade D (ISO Class 8) areas accept standard cleanroom mops but benefit from low-lint systems for EM trend stability. Color-coded framework guides procurement decisions by risk level.
Claiming “low lint” is not enough—GMP facilities require documented evidence that cleaning equipment meets quantified performance thresholds. Validation testing provides the data that supports equipment qualification, defends regulatory audits, and enables root-cause analysis when contamination events occur. Five core test categories define low-lint mop validation.

Helmke Drum Test (Particle Emission Classification)
The Helmke Drum test, standardized in IEST-RP-CC003.4, measures whole-garment or textile particle emission under simulated-use conditions. The test article (mop head, wiper, or garment) is placed inside a stainless-steel rotating drum that tumbles at 10 rpm for 10 minutes. Airborne particles released during tumbling are sampled via an isokinetic probe connected to a laser particle counter operating at approximately 1 cubic foot per minute (CFM). Particle counts are recorded at ≥0.3 µm and ≥0.5 µm, then compared to IEST-RP-CC003.3 category tables to classify the textile as Category I (recommended for ISO 1–3), Category II, or Category III (recommended for ISO 4–9).
For cleanroom mops, Helmke Drum testing validates that the mop head falls into Category I or II, confirming suitability for use in ISO Class 5–8 environments. Testing is typically performed on new mop heads and again after simulated aging (50–100 autoclave cycles or equivalent laundering) to verify that particle generation remains stable over the qualified use life. Vendors should provide Helmke Drum test reports with particle emission rates (particles/minute) and category classification; facilities conducting in-house validation can use the same protocol to qualify alternative suppliers or investigate contamination events.
ISO 9073-10 Dry-State Linting Test
ISO 9073-10 defines a dry-state linting method for nonwoven textiles, measuring the mass of lint released when a textile sample is tumbled in a standardized apparatus. While this test is widely used for wiper and garment qualification, it measures lint by weight (milligrams) rather than particle count, making it less directly applicable to cleanroom mop qualification (where particle concentration is the regulatory metric). However, ISO 9073-10 data can supplement Helmke Drum results by providing material-level characterization, especially for comparing fiber substrates or edge sealing methods during product development.
Air Particle Shedding Test (ISO 14644-14)
ISO 14644-14 provides a framework for assessing equipment suitability by measuring airborne particle concentration during equipment use. For mop validation, this translates to placing a mop head (attached to frame and handle) in a controlled cleanroom environment, performing standardized mopping strokes on a representative floor surface (epoxy or vinyl cleanroom flooring), and measuring particle generation using optical particle counters positioned downstream of the mopping path.
Test protocol details:
- Mopping pattern: Overlapping S-curves, 2 meters per pass, standardized stroke velocity (0.5 m/s).
- Downforce: Calibrated to 500 grams using a load cell or spring scale to simulate actual operator pressure.
- Mop condition: Saturated with pharmaceutical-grade water or representative disinfectant (70% IPA, hydrogen peroxide) to test wet-state shedding.
- Particle measurement: Optical particle counter sampling at 1 CFM, positioned 0.5 meters downstream, measuring ≥0.5 µm and ≥5 µm for 5 minutes post-mopping.
Acceptance criteria: Particle generation must remain below a defined threshold—typically <10 particles per stroke for ISO Class 5 use, <50 particles per stroke for ISO Class 6–7, and <100 particles per stroke for ISO Class 8. Testing is performed on new mops and after aging (25, 50, 75, 100 autoclave cycles) to validate performance stability. This test directly measures the mop’s contribution to airborne particle burden under actual use conditions, making it the gold standard for cleanroom mop qualification.
Wet Shedding Performance and Disinfectant Compatibility
Particle shedding behavior changes when mops are wetted with disinfectants. Some materials swell, stiffen, or release particles more aggressively when saturated with IPA, hydrogen peroxide, or bleach. Wet shedding validation combines chemical exposure with particle generation testing:
- Soak test: Submerge mop head in disinfectant solution at use concentration (e.g., 70% IPA, 3% H₂O₂, 0.5% bleach) for defined contact time (10–30 minutes).
- Rinse and wring: Simulate operator handling—wring out excess solution, inspect for visible fiber damage or color change.
- Particle generation re-test: Perform air particle shedding test (ISO 14644-14 protocol) with the wetted mop to measure particle release during actual cleaning.
Repeat this cycle for 10, 50, and 100 exposures to simulate cumulative chemical stress. Acceptance criteria: No mechanical failure (fiber breakage, edge separation), no absorption loss >20%, and particle generation must remain within qualified limits. This test validates that the mop’s low-lint performance is maintained across realistic disinfectant exposure and aging.
Extractables & Leachables Testing (for High-Risk Areas)
In Grade A/B environments or where mops may contact product-contact surfaces (isolator interiors, filling line decks), extractables and leachables (E&L) testing verifies that mop materials do not introduce chemical contaminants. E&L studies expose mop material to solvents (water, IPA, acidic/basic solutions) under controlled conditions, then analyze the extract using gas chromatography-mass spectrometry (GC-MS) or liquid chromatography-mass spectrometry (LC-MS) to identify and quantify leached substances. Compounds detected above threshold levels must be evaluated for toxicological risk and compatibility with product specifications.
For most pharmaceutical cleanroom mop applications, E&L testing is not required if the mop never contacts product or product-contact surfaces. However, facilities using mops inside isolators, on filling line surfaces, or in direct contact with vials/stoppers should request E&L data from vendors or conduct in-house studies. Polyester and polypropylene mop materials generally show low extractable profiles, while certain foam substrates may leach plasticizers or processing aids that require evaluation.
Autoclave Cycle Lint Stability (for Reusable Mops)
Reusable low-lint mops must maintain performance across their qualified use life—typically 50–200 autoclave cycles. Autoclave stability testing subjects mop heads to repeated sterilization at 121°C (or higher, per facility protocol), then measures dimensional stability, mechanical strength, and particle generation at defined intervals:
- Baseline (cycle 0): New mop, particle generation <50 particles per stroke.
- Mid-life (cycle 50): Dimensional change <5%, tensile strength retention >80%, particle generation <50 particles per stroke.
- End-of-life (cycle 100–200): Dimensional change <10%, no edge separation or visible fiber damage, particle generation still <50 particles per stroke.
Failure criteria: Visible degradation (fraying, discoloration, edge separation), dimensional change >10%, or particle generation exceeding qualification limits. Vendors should provide autoclave validation data showing cycles to failure; facilities can verify this data in-house by aging sample mops through their actual autoclave protocols and disinfectant exposures, then retesting particle generation per ISO 14644-14.

MIDPOSI Low-Lint Mop Recommendations
MIDPOSI offers a portfolio of validated low-lint cleanroom mops engineered to meet ISO 14644 and EU GMP Annex 1 requirements across pharmaceutical, biotechnology, semiconductor, and medical device manufacturing environments. Our product line addresses the full spectrum of cleanroom classifications, from Grade A aseptic cores to Grade D support areas, with documented particle generation performance, chemical compatibility validation, and regulatory-ready documentation.
Disposable Pre-Sterilized Mops (Grade A/B, ISO Class 5)
MIDPOSI gamma-steril polyestermoppdyna (Product Code: CMP-DS-POLY-GS)
- Material: 100% continuous-filament polyester knit, heat-sealed edges, foam-free construction.
- Sterilisering: Gamma irradiation (25–50 kGy), individually packaged in double-pouch sterile barrier system with lot-specific sterility certificates.
- Partikelgenerering: <10 particles ≥0.5 µm per stroke (validated per ISO 14644-14).
- Ansökningar: EU GMP Grade A/B aseptic filling, lyophilization loading, vial capping; ISO Class 5 sampling booths and weighing rooms.
- Förmättat alternativ: Available pre-wetted with sterile 70% IPA (CMP-DS-POLY-GS-IPA) in triple-layer packaging for direct Grade A use.
- Dokumentation: Sterility certificate (SAL 10⁻⁶), particle generation test report, material safety data sheet, gamma dose records.
MIDPOSI Gamma-Sterile Microfiber Mop Pad (Product Code: CMP-DS-MF-GS)
- Material: Polyester/polyamide microfiber blend (<1 denier), ultrasonic-sealed edges, split-fiber construction for enhanced particle capture.
- Sterilisering: Gamma irradiation, double-pouch packaging.
- Partikelgenerering: <10 particles ≥0.5 µm per stroke; superior absorbency (8× dry weight) for sporicidal agent application.
- Ansökningar: Grade A/B environments requiring high absorbency; isolator interiors; spill containment in sterile areas.
- Kemisk kompatibilitet: Validated for 70% IPA, 3–10% H₂O₂, quaternary ammonium compounds (not recommended for >0.5% bleach in single-use application).
Reusable Autoclavable Mops (Grade C/D, ISO Class 6–8)
MIDPOSI polyester återanvändbar mopp med förseglad kant (Product Code: CMP-RUS-POLY-150)
- Material: 100% polyester knit, continuous-filament yarn, heat-sealed edges on all four sides.
- Autoclave validation: 150+ cycles at 121°C with <5% dimensional change and maintained particle generation performance.
- Partikelgenerering: <50 particles ≥0.5 µm per stroke across entire use life.
- Chemical resistance: Validated for 70% IPA, 0.5–1% bleach, 3–35% H₂O₂, quaternary ammonium compounds (2,000 ppm), and phenolic disinfectants.
- Ansökningar: Grade C aseptic support areas, ISO Class 6–7 manufacturing zones, Grade D packaging and compounding areas.
- Cost efficiency: Lowest per-use cost for high-volume facilities with validated laundering infrastructure.
MIDPOSI återanvändbar mopp i mikrofiber (sealed-edge) (Product Code: CMP-RUS-MF-100)
- Material: Polyester/polyamide microfiber, ultrasonic-welded edges, split-fiber technology.
- Autoclave validation: 100+ cycles at 121°C.
- Partikelgenerering: <50 particles ≥0.5 µm per stroke; exceptional particle capture efficiency for damp dusting and residual particle removal.
- Absorptionsförmåga: 6–8× dry weight; ideal for large-area mopping and heavy disinfectant application.
- Kemisk kompatibilitet: Optimized for IPA and hydrogen peroxide programs; acceptable for periodic bleach use (≤0.5%).
- Ansökningar: ISO Class 6–8 environments prioritizing absorbency and particle capture; gowning rooms; material airlocks.
Product Selection Table by Cleanroom Grade
| Cleanroom Grade | ISO klass | Particle Limit (≥0.5 µm/m³) | Recommended MIDPOSI Product | Partikelgenerering | Sterilisering | Cost Category |
| Grade A/B | Class 5 | 3,520 | CMP-DS-POLY-GS (Gamma-Sterile Disposable) | <10 particles/stroke | Gamma, pre-sterile | Premium |
| Grade A/B | Class 5 | 3,520 | CMP-DS-MF-GS (Gamma-Sterile Microfiber Disposable) | <10 particles/stroke | Gamma, pre-sterile | Premium |
| Betyg C | Class 7 | 352,000 | CMP-RUS-POLY-150 (Polyester Reusable) | <50 particles/stroke | Autoclave in-house | Standard |
| Betyg C | Class 7 | 352,000 | CMP-RUS-MF-100 (Microfiber Reusable) | <50 particles/stroke | Autoclave in-house | Standard |
| Betyg D | Class 8 | 3,520,000 | CMP-RUS-POLY-150 (Polyester Reusable) | <50 particles/stroke | Disinfection or autoclave | Economy |
| ISO 5 Sampling/Weighing | Class 5 | 3,520 | CMP-DS-POLY-GS or CMP-RUS-POLY-150 | <10–20 particles/stroke | Per CCS requirements | Variabel |
| Support Areas / Manlocks | Class 8+ | ≥3,520,000 | CMP-RUS-POLY-150 or standard cleanroom mop | <100 particles/stroke | Disinfection | Economy |
How to Order and Validation Support
MIDPOSI provides comprehensive technical support to assist QA teams, facility engineers, and procurement specialists in selecting, qualifying, and deploying low-lint cleanroom mops:
- Sample kits: Evaluation packs containing 5–10 mop heads (mix of polyester and microfiber, reusable and disposable) for in-house pilot testing and particle generation validation.
- Technical documentation: Particle generation test reports (ISO 14644-14), Helmke Drum test results, chemical compatibility matrices, autoclave validation data, and sterility certificates (for disposables) provided with every product shipment.
- Validation protocols: Customized test protocols for facility-specific qualification studies, including recommended acceptance criteria, sampling plans, and data analysis templates.
- On-site training: Operator training sessions covering proper mop handling, disinfectant application techniques, color-coded segregation systems, and inspection procedures for identifying mop degradation.
- Regulatory documentation support: Assistance preparing justification documents for FDA audits, EMA inspections, and Contamination Control Strategy updates, including risk assessments and vendor qualification summaries.
For product catalogs, technical specifications, or to request a sample kit, visit MIDPOSI renrumsförbrukningsmaterial or contact your regional technical representative. Our team is available to discuss facility-specific contamination control challenges and recommend mop configurations optimized for your ISO classification, disinfectant program, and budget constraints.









