Einführung
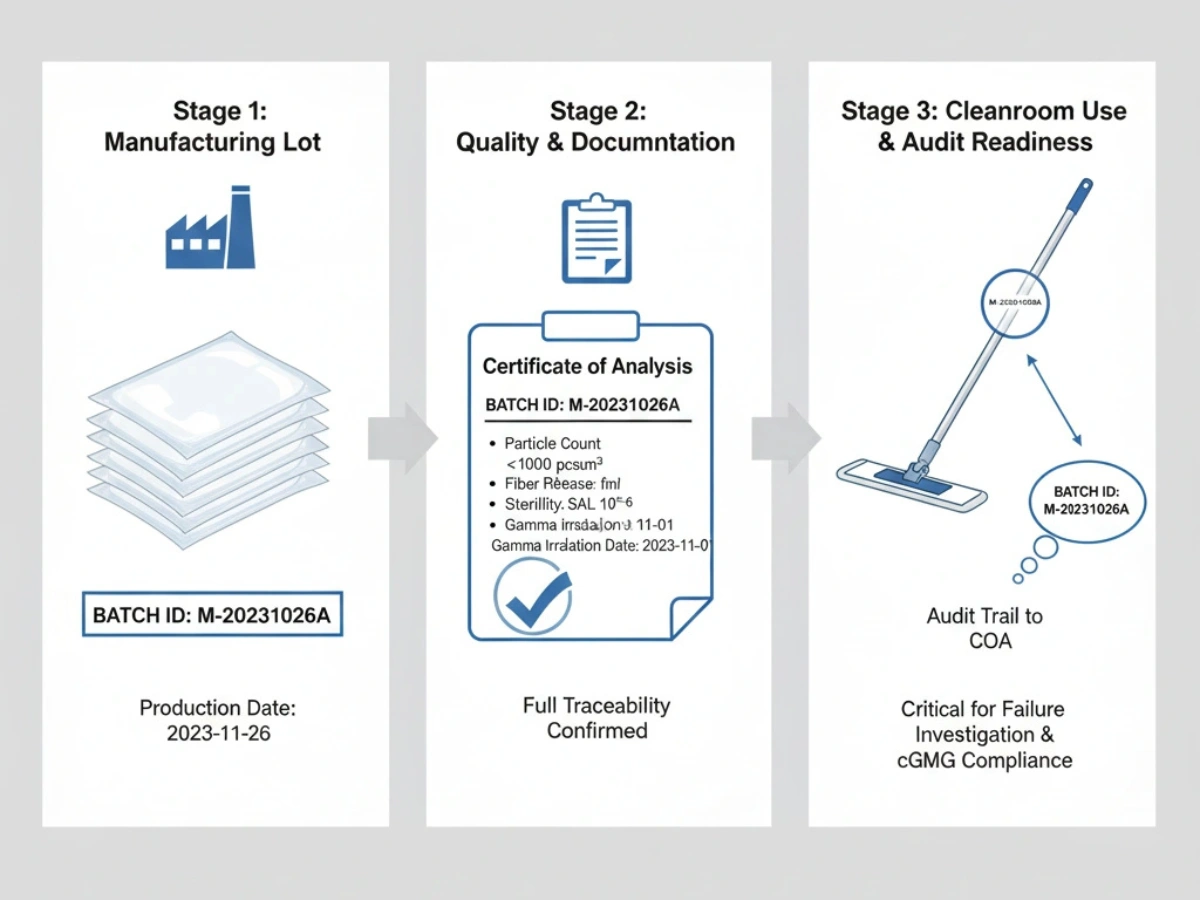
Batch traceability for cleanroom consumables is a critical GMP requirement that enables rapid identification, isolation, and recall of defective products. While many facilities maintain basic batch records, complete traceability across the full product lifecycle—from raw materials to end-user consumption—remains difficult to implement well.
Traceability failures can lead to regulatory citations, delayed investigations, extended recalls, and weaker audit performance. A strong system should allow teams to move quickly in both directions: from a customer complaint back to raw materials, and from a raw material issue forward to all affected finished products. :contentReference[oaicite:2]{index=2}
Regulatory Requirements
Your original article correctly anchors traceability in EU GMP Annex 1, FDA cGMP, and ISO 13485. These frameworks may differ in wording, but they all expect the same practical outcome: traceable product movement, reliable records, and rapid recall capability. :contentReference[oaicite:3]{index=3}
| Rahmen | Area | Key expectation |
|---|---|---|
| Ich GMP Anhang 1 | Traceability and recall readiness | Complete traceability and rapid recall capability |
| FDA 21 CFR 211 | Distribution and batch records | Distribution records sufficient for recall and investigation |
| ISO 13485 | Documented traceability procedures | Unique identification and retrieval of relevant records |
Traceability Framework
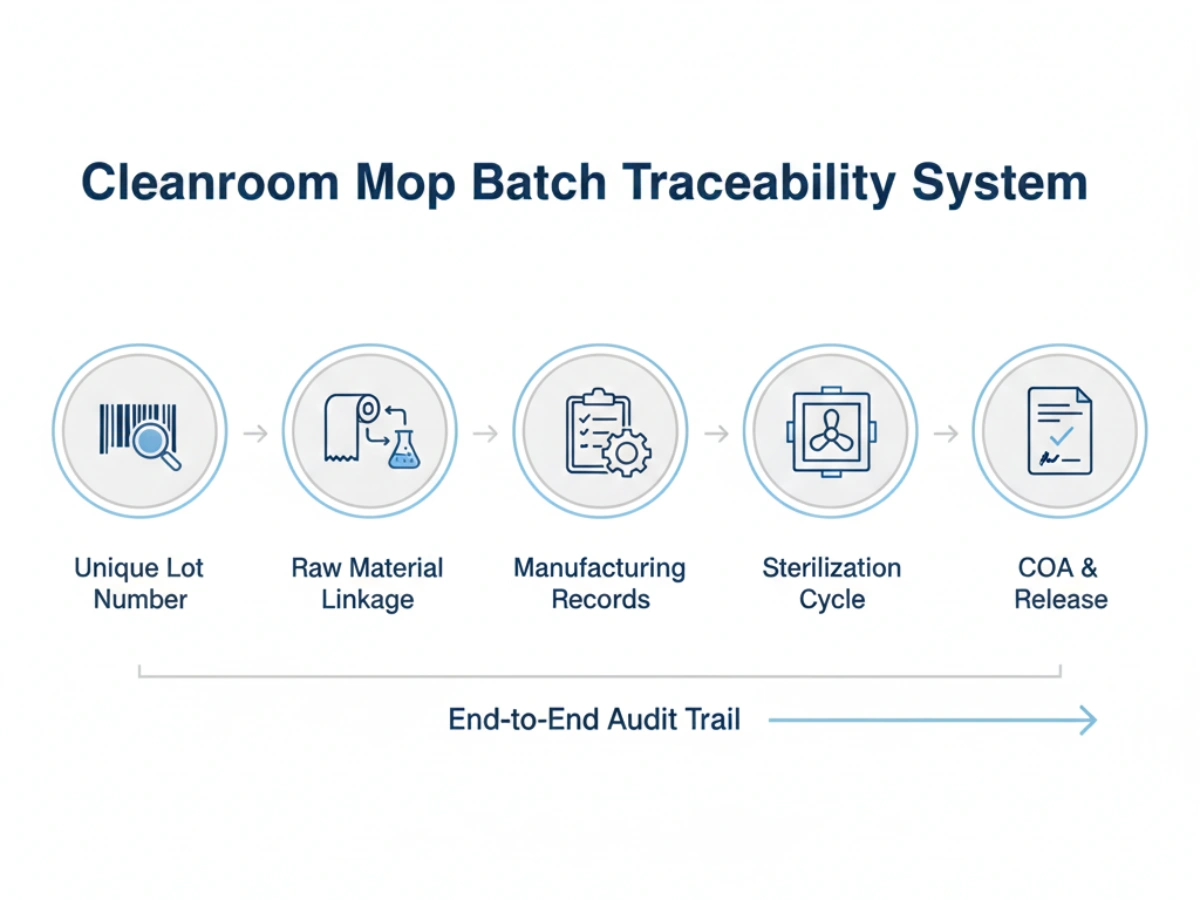
One of the strongest parts of your original article is the MIDPOSI 5-layer traceability model. This works well because it maps traceability not as a single database field, but as a linked lifecycle: raw materials, manufacturing, sterilization, distribution, and consumption. :contentReference[oaicite:4]{index=4}
| Schicht | Scope | Key data elements |
|---|---|---|
| Layer 1 | Raw materials | Supplier ID, lot number, receipt date, COA, certificate of conformity |
| Layer 2 | Manufacturing | Production batch, equipment, operators, materials used, in-process QC |
| Layer 3 | Sterilisation | Sterilization batch, method, parameters, SAL, release decision |
| Layer 4 | Distribution | Customer ID, shipment date, carrier, quantity, address |
| Layer 5 | Consumption | Usage date, operator, quantity used, disposal or return pathway |
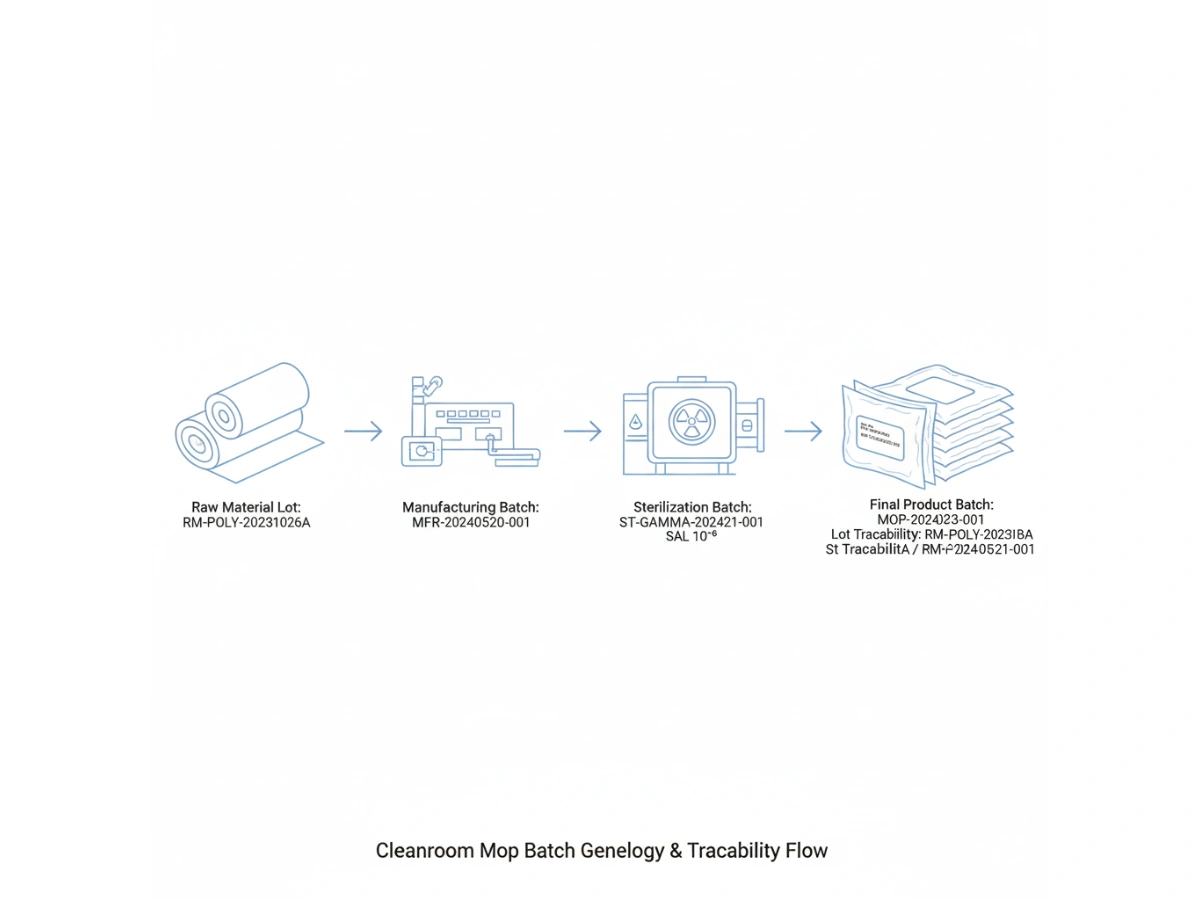
Batch Coding Systems
A traceability system depends on an understandable, unique, and consistently applied batch numbering structure. Your original article proposes a practical structure with supplier code, year, month, day, serial number, and process suffix. That is a good model because it balances readability with uniqueness. :contentReference[oaicite:5]{index=5}
| Element | Format | Example | Purpose |
|---|---|---|---|
| Supplier code | 3–8 characters | MITTELPOSI | Identifies manufacturer / source |
| Year | 4 digits | 2025 | Manufacturing year |
| Month | 2 digits | 01 | Manufacturing month |
| Day | 2 digits | 15 | Manufacturing day |
| Serial number | 3 digits | 001 | Daily production sequence |
| Suffix | 1 character | A / B / C | Process-step indicator |
Documentation Requirements
Traceability is only as good as its records. Each layer should have a documented handoff. That means incoming material records, batch records, sterilization linkage, shipment records, and end-use or disposal records should all be retrievable and mutually linked.
Minimum traceability record set
- Batch number
- Manufacturing date
- Expiration date
- Sterilization lot
- Customer recipient
- Consumption or disposal date
Documents most often missed
- Supplier lot to internal batch linkage
- Sterilization cycle linkage
- Shipment quantity reconciliation
- Consumption or return records at user level
Traceability Testing
A traceability system should not be trusted just because records exist. It should be tested. Your original article correctly calls for forward traceability, backward traceability, and recall simulation, each with a 24-hour target. :contentReference[oaicite:6]{index=6}
Forward traceability test
Start from a raw material lot and verify that all related production, sterilization, distribution, and customer records can be identified.
Backward traceability test
Start from a customer complaint, usage record, or finished batch and trace back to all contributing raw materials and process steps.
Recall simulation test
Select a batch and verify that all affected customers, quantities, and related records can be identified within the target timeframe.
Recall / Retraction Procedures
Traceability exists so that recalls and investigations can be executed fast and accurately. A strong recall system should define decision ownership, customer notification flow, retrieval actions, regulatory communication, and root-cause follow-up.
| Recall step | Expected timing | Typical owner |
|---|---|---|
| Recall initiation | Immediate | QA / Quality manager |
| Regulatory notification | Within 24 hours | QA / Regulatory |
| Customer notification | Within 24 hours | Customer service / QA |
| Product retrieval | Within 48 hours | Logistics |
| Root-cause investigation | Within 7 days | QA / Operations |
Electronic Traceability Solutions
Electronic systems can significantly improve traceability speed, retrieval accuracy, and audit readiness, but only if they are validated, access-controlled, and backed up properly. Your original article highlights barcode or RFID capture, database integration, mobile access, alerts, reporting, and audit trails as core features. :contentReference[oaicite:7]{index=7}
| System feature | Why it matters |
|---|---|
| Batch number generation | Ensures unique and controlled identification |
| Barcode / RFID scanning | Reduces manual transcription errors |
| ERP / MES integration | Connects procurement, production, and shipping data |
| Audit trail | Supports data integrity and change review |
| Backup & recovery | Protects records during system disruption |
| Role-based access | Protects traceability data from unauthorized changes |
Electronic traceability can replace paper-heavy systems, but it should never reduce visibility or make record retrieval harder during inspection.
Need Help Improving Batch Traceability for Cleanroom Consumables?
Get practical guidance on lot structure, traceability records, supplier linkage, recall testing, and documentation systems that are easier to defend during audits.
- Gap analysis for traceability readiness
- Support for batch coding and record structure
- Guidance on recall simulation and supplier-linked traceability
Häufig gestellte Fragen
What level of traceability is required for cleanroom consumables?
Best practice is full batch-level traceability from raw materials through manufacturing, sterilization, distribution, and end use, even if local regulatory minimums are narrower.
How long should traceability records be retained?
Retention depends on regulatory framework and quality system, but many companies keep records for at least 5–7 years to support full lifecycle coverage.
What is the minimum time frame for identifying all affected products in a recall?
Industry expectation is often within 24 hours for identifying affected batches, customers, and quantities.
Can electronic traceability systems replace manual records?
Yes, if the system is validated, secure, backed up, and maintains audit trail integrity. Electronic systems should improve speed and control, not reduce visibility.
What are the most common traceability gaps?
Common gaps include incomplete raw material linkage, missing sterilization lot linkage, incomplete shipment records, and no user-level consumption or disposal record.
How often should traceability be tested?
Forward and backward traceability plus recall simulation should be tested at least annually, with additional testing after major process or system changes.