A technical reference for QA managers and validation engineers on the integration of consumable traceability into the site Contamination Control Strategy (CCS).

In a GMP-regulated environment, the cleanroom mop is no longer viewed as a generic utility but as a critical controlled input. Batch traceability cleanroom mop systems serve three primary functions:
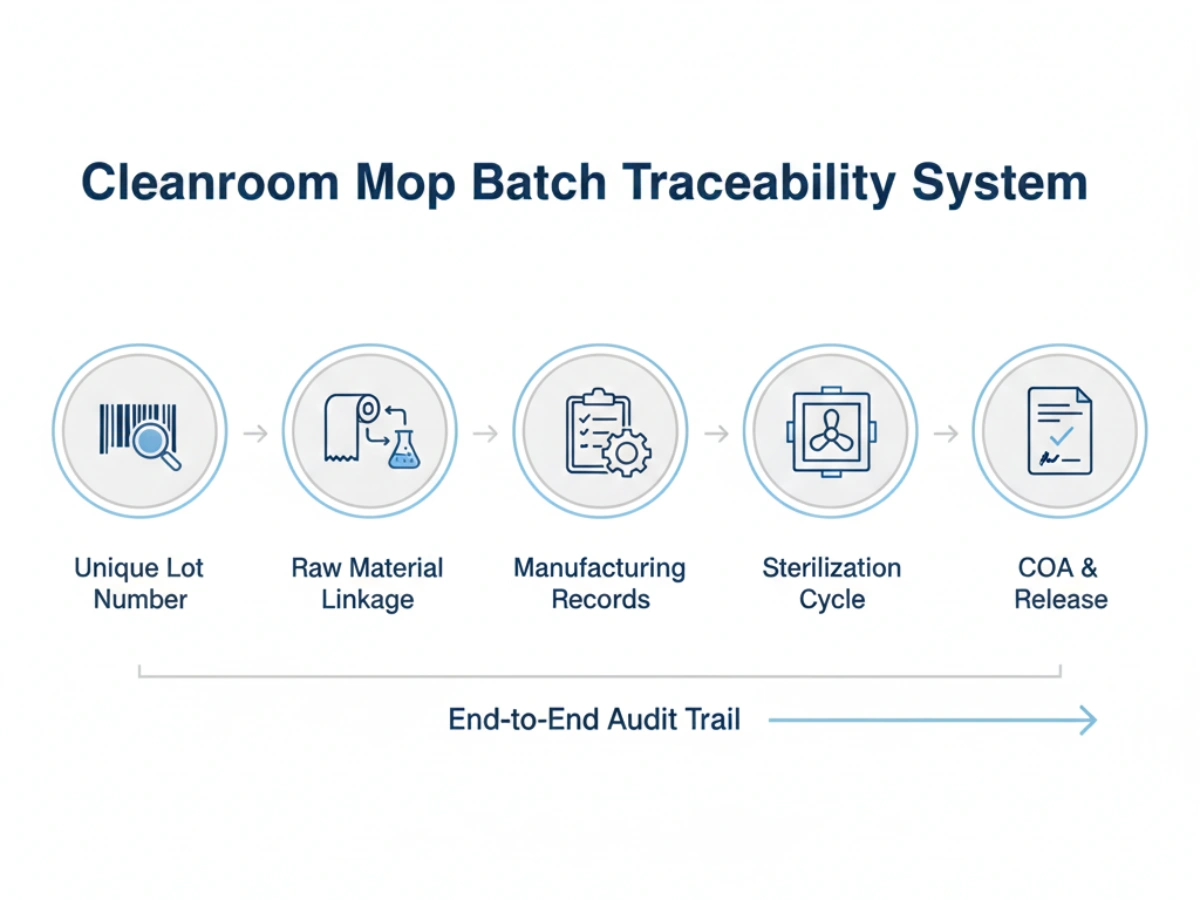
Product-level traceability identifies a SKU; batch-level traceability identifies the specific manufacturing event. For cleanroom mops, this requires a "genealogy" that links the finished product back to the specific lot of polyester yarn or microfiber fabric used, the cleanroom laundry shift, and the specific sterilization cycle.
When mops are treated as controlled inputs, every individual package is a data point. This data allows QA teams to verify that the product in their hand is identical in performance and purity to the product validated during the initial facility startup or PQ (Performance Qualification).
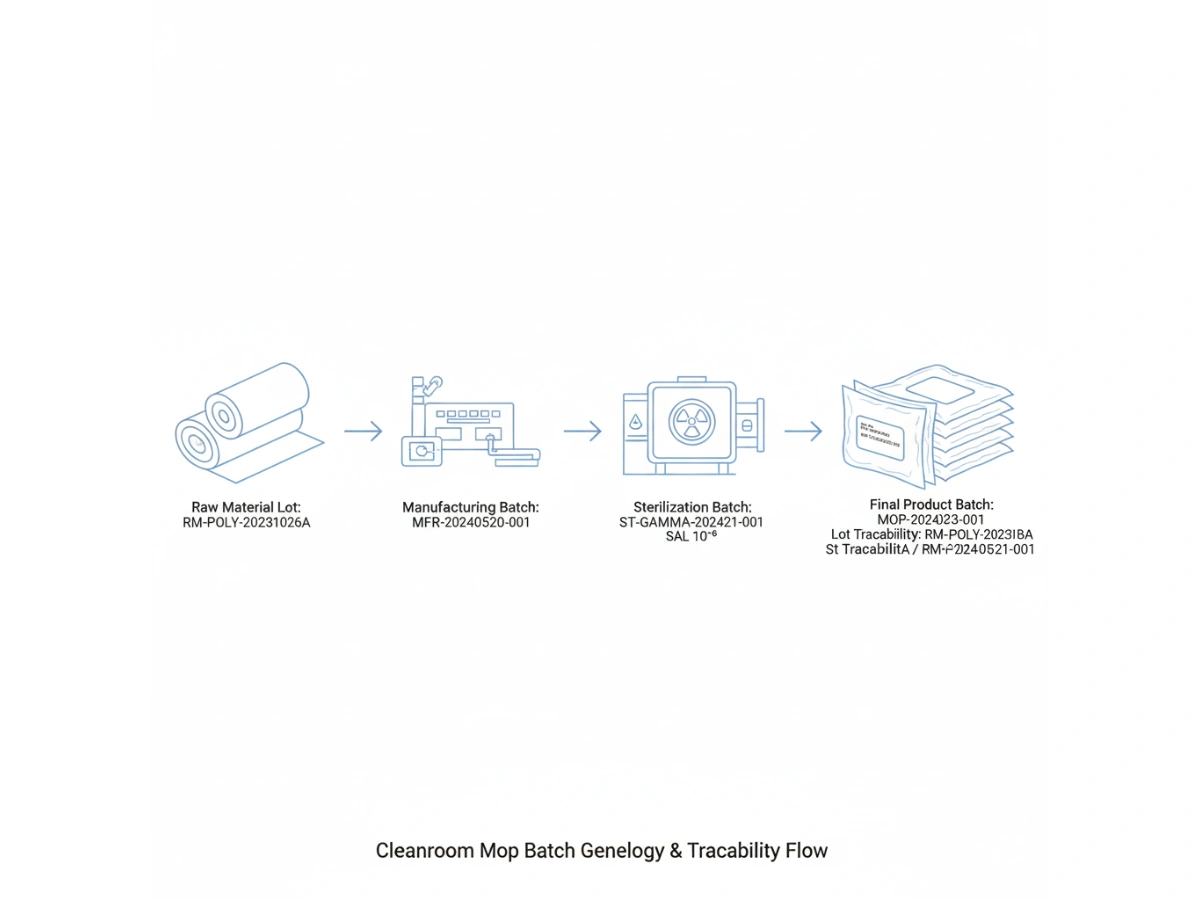
Modern GMP principles require that all materials coming into contact with sensitive surfaces be fully accounted for. During an inspection, an auditor may select a date of production and ask for the lot traceability of the cleaning materials used on that day.
Expectations focus on the "Linkage of Records." An auditor will look for a seamless transition from the facility's cleaning logs to the バッチレベルのドキュメント provided by the supplier. If a mop batch cannot be linked to a Certificate of Analysis (COA) or a sterilization record, the sterility and cleanliness of the entire cleanroom suite may be called into question.

A robust traceability system for cleanroom consumables GMP compliance must include:
Supply stability is often threatened by "drift"—small, undocumented changes in manufacturing that accumulate over years. Batch traceability prevents this by establishing a baseline for ロット間の一貫性.
環境モニタリング (EM) のスパイクを伴う CAPA (是正および予防措置) が発生した場合、QA はトレーサビリティを利用して、モップの特定のバッチがそのイベントに寄与したかどうかを判断できます。これがなければ、施設はすべての在庫を廃棄し、生産を停止せざるを得なくなり、大きな運営リスクが生じる可能性があります。トレーサビリティは、施設全体の潜在的な危機を局所的な物質の検疫に変えます。

あ pharmaceutical mop manufacturer / OEM acts as an extension of the pharma site’s quality system. Their responsibilities include:
For a qualified manufacturer, the ability to produce these records on demand is the hallmark of GMP maturity.
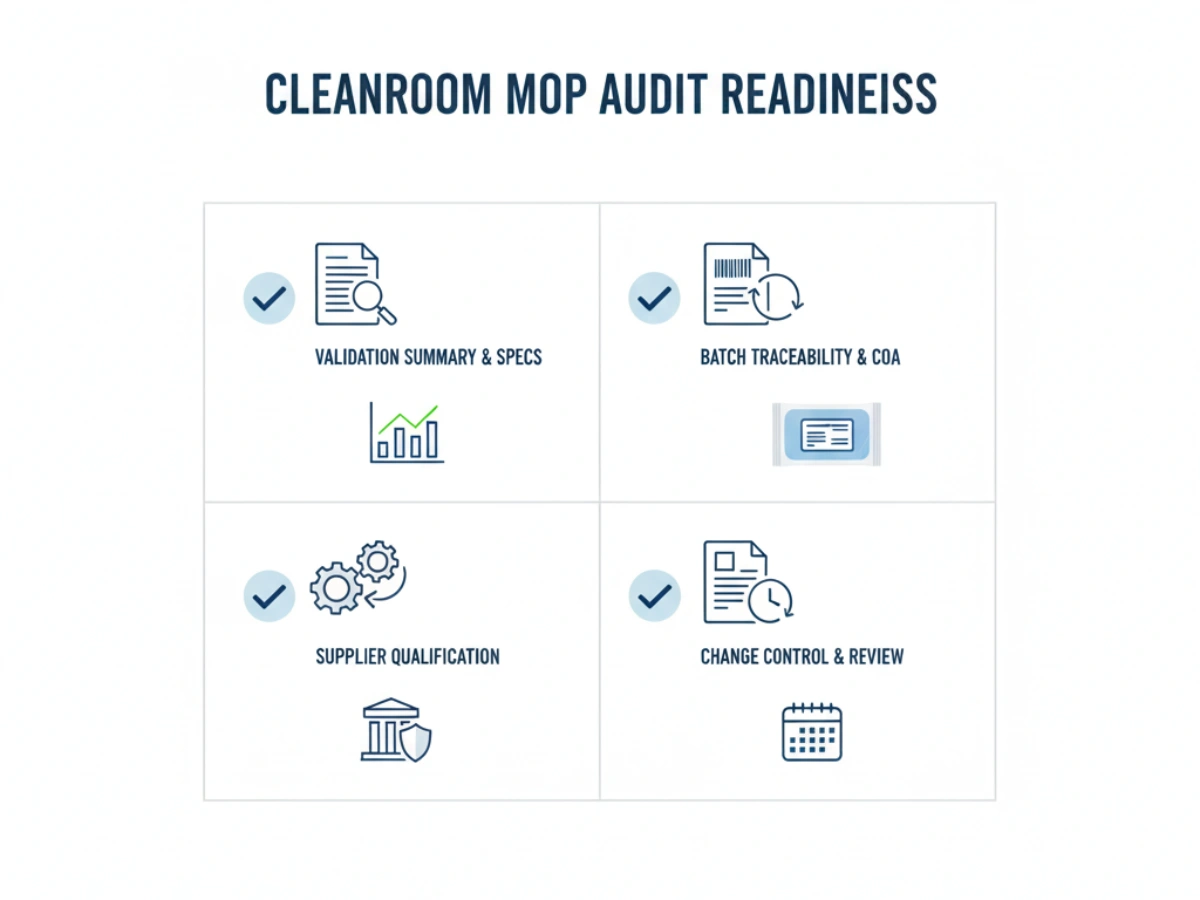
その間 OEM supplier qualification, QA teams should utilize the following checklist:
| Checkpoint | 要件 |
|---|---|
| Batch Definition | Is the batch size clearly defined and manageable? |
| Sample Retention | Does the supplier keep retain samples for every lot? |
| Audit Access | Can the supplier provide full raw material logs within 48 hours? |
| Physical Labeling | Is the batch number resistant to cleanroom disinfectants (IPA)? |
Batch traceability is not merely a documentation exercise; it is the backbone of a defensible quality system. In the context of Grade A and B environments, the ability to track every fiber and sterilization cycle ensures that the cleaning process remains a controlled variable rather than an unknown risk.
成熟した製薬 QA チームは、バッチ記録に完全な透明性を示しているサプライヤーを優先し、患者の安全と規制遵守における長期的なパートナーと見なしています。
監査ナラティブを強化するために、ロット系図、バッチ固有の COA、滅菌連携、および変更管理の準備状況を尋ねます。
ドキュメントパッケージをリクエストする

1営業日以内にご連絡いたします。末尾が次のメールに注意してください 「*@midposi.com」.