Cleanroom Mop for Medical Device Manufacturing: ISO Cleaning and Particle Control
MIDPOSI cleanroom mop systems support medical device manufacturing facilities where particle control, low-lint materials, packaging cleanliness, surface cleaning, supplier qualification, and documentation are essential for ISO-classified cleanroom operations.
AI Search Summary
Medical device cleanrooms require low-lint mop systems that help control particles, fibers, residues, and cross-area contamination during device assembly, inspection, and packaging. Buyers should review mop material, sterile or non-sterile requirements, disposable replacement workflow, packaging format, lot traceability, and supplier documentation before selecting a cleanroom mop system.

Why Medical Device Cleanrooms Need Dedicated Mop Systems
Medical device cleanrooms are designed to control particles, fibers, residues, and contamination risks that may affect device assembly, inspection, packaging, and product quality. A regular janitorial mop may release lint, retain residues, or carry contaminants between controlled areas.
A cleanroom mop for medical device manufacturing should be evaluated as part of a controlled cleaning system: mop cover, frame, handle, bucket workflow, replacement frequency, packaging, traceability, and documentation support.
粒子制御
Low-lint mop materials help reduce the risk of particles settling on device surfaces, components, and work areas.
Packaging Cleanliness
Fibers near sterile barrier packaging, trays, films, or seals may create avoidable quality concerns.
Supplier Review
Medical device buyers often need TDS, material information, packaging details, lot traceability, and sample approval records.
Medical Device Cleanroom Cleaning Risks
In medical device manufacturing, contamination risk is not limited to visible dust. Small particles, fibers, chemical residues, and uncontrolled cleaning tools may affect product assembly, packaging cleanliness, inspection results, and internal quality review.
Device Surface Particles
Particles may settle on molded parts, implantable components, tubing, diagnostic parts, or assembly surfaces.
Fiber Contamination
Loose fibers may interfere with packaging cleanliness, sterile barrier materials, and visual inspection workflows.
Residue Transfer
Incompatible mop materials may leave residue or interact poorly with the facility’s approved cleaning agents.
Cross-Area Movement
Cleaning tools used across multiple zones may carry contamination from support areas into higher-control rooms.
Inconsistent Replacement
Unclear mop change rules may create variation between operators, shifts, rooms, or cleaning frequencies.
Weak Documentation
Missing specifications, packaging details, or traceability information can delay supplier qualification and QA approval.
Medical Device Cleanroom Mop Selection Matrix
Use the table below to match cleanroom mop selection with common medical device manufacturing areas.
| Medical Device Area | Main Cleaning Risk | Recommended Mop Focus |
|---|---|---|
| Device Assembly Room | Particles on device surfaces and components | Low-lint polyester mop or validated microfiber mop system |
| Packaging Cleanroom | Fibers near sterile barrier materials, films, trays, or seals | Disposable low-lint mop covers with controlled replacement workflow |
| Inspection Area | Surface residue, glare, dust, and fine particle pickup | Microfiber or low-lint mop selected according to SOP and surface type |
| Gowning Area | Traffic-related contamination and transfer from personnel movement | Frequent mop cover replacement and zone-based cleaning workflow |
| Warehouse / Support Area | Floor particles entering controlled spaces | Cost-efficient disposable or reusable cleanroom-compatible mop option |
Cleanroom Mops for Medical Device Packaging Areas
Packaging cleanrooms require special attention because fibers, dust, and residues may affect sterile barrier packaging, seal areas, pouches, trays, labels, and final device presentation. A mop system used in packaging areas should support controlled floor cleaning without becoming a source of loose fibers or residues.
Packaging Area Concerns
- Fibers near sterile barrier seals
- Particles on packaging film or trays
- Residues from incompatible cleaning tools
- Cross-area transfer from gowning or material staging zones
- Inconsistent cleaning frequency between shifts
Recommended Mop Focus
- Low-lint polyester or qualified microfiber mop covers
- Disposable replacement workflow for higher-control areas
- Defined mop change frequency
- Clear packaging and lot traceability
- Sample testing before bulk approval

Cleanroom Mop Material Selection for Medical Device Manufacturing
The best mop material depends on the surface type, cleanroom classification, product risk, cleaning agent, replacement workflow, and internal SOP. Medical device buyers should compare materials by lint control, absorbency, residue pickup, chemical compatibility, and supplier documentation.

| Material / Format | 最適な用途 | Buyer Review Focus |
|---|---|---|
| ポリエステル製クリーンルームモップ | Stable low-lint cleaning in assembly, packaging, and support areas | Material consistency, packaging, mop weight, and lint-control expectations |
| マイクロファイバークリーンルームモップ | Fine particle pickup, surface contact, and residue removal | Fiber shedding, processing cleanliness, chemical compatibility, and SOP suitability |
| Disposable Mop Covers | Controlled replacement, packaging areas, and multi-zone cleaning workflows | Lot traceability, packaging format, change frequency, and cost per cleaning task |
| Sterile Mop Pads | Specific workflows where sterile presentation is required by product risk or SOP | Sterility information, sterile packaging, lot tracking, and QA approval requirements |
Disposable vs Reusable Mop Systems for Medical Device Cleanrooms
Medical device manufacturers often prioritize repeatability and practical process control. Disposable mop covers can simplify replacement control and reduce laundering-related variables. Reusable mops may be suitable when the facility has controlled laundering, drying, inspection, storage, and reuse limits.
Disposable Mop Advantages
- Defined replacement after room, zone, or cleaning task
- Reduced laundering validation burden
- Helpful for packaging cleanrooms and higher-control areas
- Easier lot-based inventory planning
- Consistent unused surface for each defined cleaning workflow
Reusable Mop Considerations
- Requires validated washing and drying process
- Requires inspection before reuse
- Needs storage control after laundering
- Lifecycle and reuse limits should be defined
- Total cost should include labor, laundering, and documentation
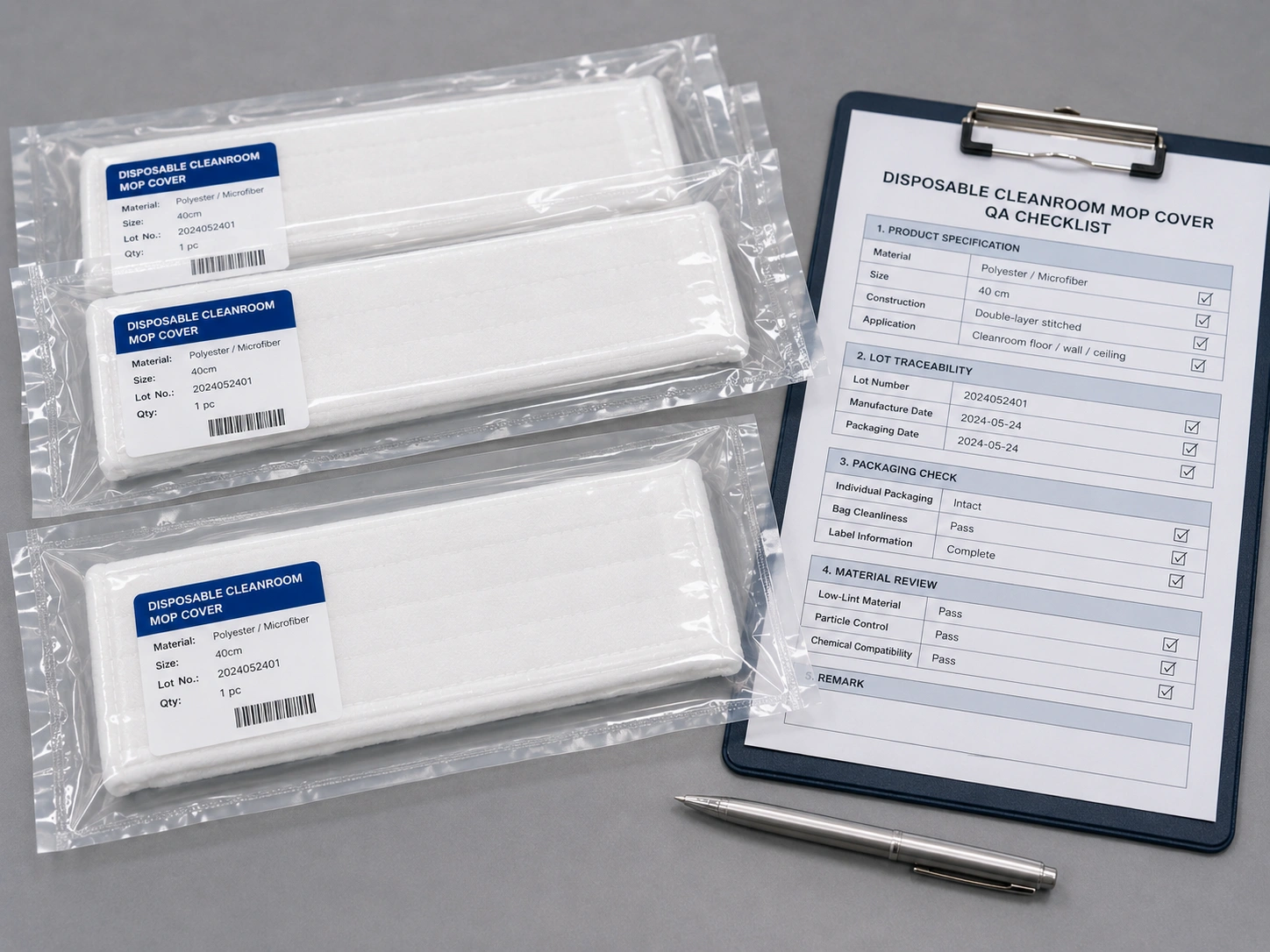
Do Medical Device Cleanrooms Need Sterile Mop Pads?
Not every medical device cleanroom requires sterile mop pads. Some workflows, such as implantable device manufacturing, sterile barrier packaging support, or higher-risk controlled areas, may require sterile or highly controlled packaging. Many ISO cleanrooms use non-sterile low-lint mop systems for routine cleaning when the facility SOP allows it.

Cleaning Workflow and SOP Integration
Cleanroom mop performance depends not only on the mop material, but also on how the tool is used inside a defined SOP. Medical device cleanrooms should define cleaning direction, zoning, mop cover replacement frequency, wet mopping control, bucket workflow, and documentation practices.
- Defined cleaning direction: Clean from more controlled areas toward lower-control areas where applicable.
- Area zoning: Assign mop systems by room, zone, or production area to reduce cross-area transfer.
- Mop cover replacement: Define when mop covers are changed by area, square footage, shift, or cleaning task.
- Wet mopping control: Use suitable bucket and wringer systems to control solution application.
- Cleaning logs: Record cleaning activity and retain product or lot information where required.
ISO 13485 Supplier Qualification Support
Medical device manufacturers operating under ISO 13485 quality systems often require supplier documentation, traceability, sample approval records, stable repeat supply, and clear product specifications. A cleanroom mop supplier should support the buyer’s supplier qualification process with practical documentation and consistent communication.
Product Information
Size, material, construction, sterile/non-sterile status, packaging, and compatible cleanroom use areas.
Traceability Support
Lot or batch information to support incoming inspection, repeat orders, and internal supplier review.
Sample Approval
Sample testing can help confirm mop fit, absorbency, packaging, handling, and compatibility before bulk purchase.
購入者が要求する必要がある文書
Medical device buyers should evaluate cleanroom mops by more than price and appearance. Documentation helps purchasing, QA, production, and cleanroom operations teams decide whether the mop system fits the facility’s cleaning program.
| Document / Information | なぜそれが重要なのか | Buyer Review Purpose |
|---|---|---|
| Technical Data Sheet | Shows size, material, construction, packaging, and recommended use | Initial product evaluation and comparison |
| Material Information | Explains polyester, microfiber, or other mop construction | Low-lint and chemical compatibility review |
| Packaging Details | Shows inner packaging, carton data, sterile or non-sterile packaging format | Storage, cleanroom entry, and handling review |
| Lot Traceability | Connects product shipments to lot or batch information | Incoming inspection and repeat-order control |
| Sample Approval Record | Confirms the tested product matches the future bulk order | Prevents sample-to-order mismatch |
| Supplier Capability Information | Shows B2B support, MOQ, lead time, packaging, and documentation availability | Supplier qualification and procurement planning |

Reviewed by MIDPOSI Cleanroom Contamination Control Team
This guide is prepared for B2B buyers, cleanroom supervisors, QA teams, procurement teams, and medical device manufacturing facilities evaluating cleanroom mop systems for controlled cleaning workflows.
ミドポシ supports cleanroom consumables sourcing, including cleanroom mop systems, low-lint mop covers, disposable cleanroom mops, sterile and non-sterile mop options, cleanroom garments, and documentation support for controlled environments.
MIDPOSI Recommendation for Medical Device Buyers
For medical device manufacturing, MIDPOSI recommends starting with a low-lint mop system that matches the facility’s ISO cleanroom classification, packaging area risk, cleaning surface, replacement workflow, and supplier documentation needs.
For Assembly Areas
Use stable low-lint mop materials that support particle control and repeatable cleaning.
For Packaging Areas
Consider disposable low-lint mop covers to support clean replacement and reduce fiber carryover risk.
For QA-Driven Buyers
Request specifications, packaging details, lot information, and sample support before bulk order approval.
よくある質問
What type of mop is used in medical device cleanrooms?
Medical device cleanrooms generally use flat, low-lint cleanroom mops made from polyester, microfiber, or other cleanroom-compatible materials. The mop should support particle control, surface cleaning, packaging cleanliness, and the facility’s internal SOP.
Do medical device cleanrooms require sterile mop pads?
Not always. Some high-risk workflows may require sterile mop pads, but many ISO cleanrooms in medical device manufacturing use non-sterile low-lint mop systems for routine cleaning. The final choice depends on product risk, cleanroom classification, and internal SOP.
Are disposable cleanroom mops suitable for medical device manufacturing?
Yes. Disposable cleanroom mops are often suitable because they help simplify replacement control, reduce laundering variables, and support consistent cleaning workflows across rooms, shifts, and packaging areas.
What mop material is best for medical device cleanrooms?
Polyester is often selected for stable low-lint performance, while microfiber may be selected for stronger surface contact and fine particle pickup. The best choice depends on the cleaning surface, approved disinfectants, SOP, and QA requirements.
How do cleanroom mops help control particles?
Cleanroom mops help control particles by using low-lint materials, controlled mop construction, suitable frames, and defined replacement workflows. They help reduce the risk that the cleaning tool itself becomes a source of contamination.
What documents should medical device buyers request?
Buyers should request a technical data sheet, material information, packaging details, lot traceability, cleanroom-use information, sample approval records, and supplier capability information where required.
Can cleanroom mops be used in packaging cleanrooms?
Yes. Low-lint cleanroom mops can be used in medical device packaging cleanrooms to help control floor particles, fibers, residues, and contamination risks near sterile barrier materials, trays, films, and packaging zones.
Need Cleanroom Mop Systems for Medical Device Manufacturing?
Contact MIDPOSI to discuss your ISO cleanroom grade, device assembly area, packaging workflow, mop material preference, documentation needs, sample plan, MOQ, and B2B supply requirements.



