Why Cleanroom Cleaning Is a Core Contamination Control Step
Particles, residues, fibers, and microorganisms accumulate on cleanroom surfaces through personnel movement, material transfer, airflow disturbance, and routine operations. If these residues are not removed through a controlled cleaning process, they become persistent contamination sources that can affect product quality, environmental monitoring trends, and audit readiness.
That is why GMP cleaning SOPs must do more than assign a frequency. They must define cleaning method, disinfectant selection, operator technique, validation logic, and documentation. A good SOP also connects directly with ਵਾਤਾਵਰਣ ਦੀ ਨਿਗਰਾਨੀ, ਕਰਮਚਾਰੀ ਸਿਖਲਾਈ, gowning discipline, ਅਤੇ ਹਵਾ ਦਾ ਪ੍ਰਵਾਹ ਪ੍ਰਦਰਸ਼ਨ.
| Cleaning Objective | ਇਹ ਮਾਇਨੇ ਕਿਉਂ ਰੱਖਦਾ ਹੈ | ਕਮਜ਼ੋਰ ਹੋਣ 'ਤੇ ਜੋਖਮ |
|---|---|---|
| Particle removal | Reduces surface-to-air and surface-to-product contamination transfer | ISO class instability and visible residue |
| Microbial reduction | Controls viable contamination on contact surfaces and equipment | EM excursions and product contamination risk |
| Residue control | Prevents disinfectant buildup and material incompatibility | Chemical residue, staining, sticky surfaces |
| Technique standardization | Improves repeatability across operators and shifts | Variable cleaning performance |
| ਪ੍ਰਮਾਣਿਕਤਾ | Proves that cleaning is effective, not just performed | Weak GMP defensibility |
Cleaning and Disinfection Strategy by Grade
Cleaning programs should reflect room classification, exposure risk, intervention frequency, and microbial control needs. A Grade A zone used in aseptic processing requires a much more aggressive and validated strategy than a Grade D support space.
| Grade | Risk Level | Cleaning Frequency | Sanitization / Disinfection |
|---|---|---|---|
| ISO 5 (Grade A) | Critical aseptic | Every operation / high frequency surface control | Every operation |
| ISO 7 (Grade B) | Background aseptic | ਰੋਜ਼ਾਨਾ | ਰੋਜ਼ਾਨਾ |
| ISO 8 (Grade C) | Controlled area | Daily to weekly based on use | Weekly or defined rotation |
| ISO 9 (Grade D) | Support area | Daily routine cleaning | Weekly or risk-based |
GMP-ਤਿਆਰ ਕਲੀਨਰੂਮ ਸਹਾਇਤਾ ਦੀ ਲੋੜ ਹੈ?
Explore Midposi solutions designed for pharmaceutical and controlled environments, including cleanroom garments and contamination-control product support.
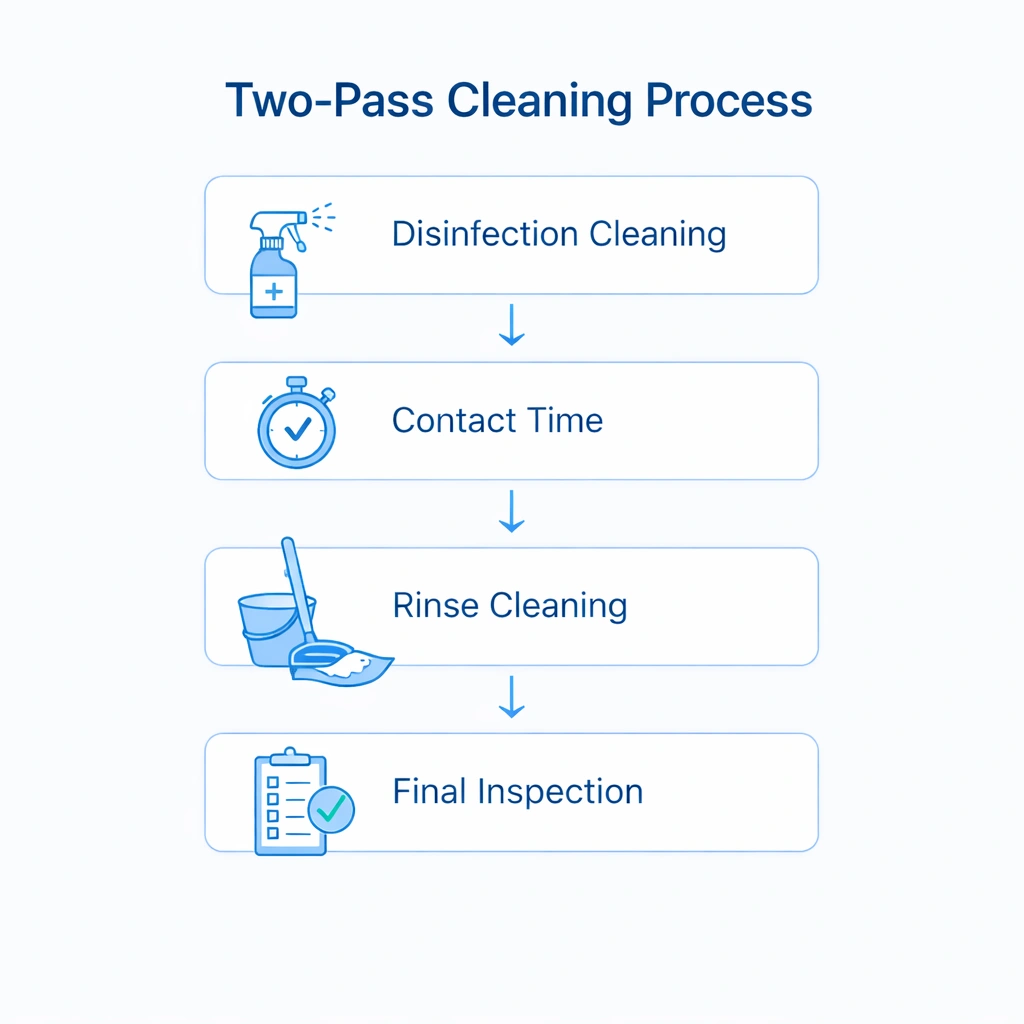
Two-Pass Cleaning Method for GMP Cleanrooms
In higher-grade environments, a two-pass method is often preferred because it separates chemical application from residue removal. This reduces the risk of incomplete disinfection, sticky surface buildup, and uneven finishing.
Pass 1: Disinfection Cleaning
- Apply validated disinfectant to the wipe or mop system
- Use unidirectional or figure-8 motion based on SOP
- Maintain defined contact time
- Cover the area systematically without random overlap
Pass 2: Residue Removal / Final Clean
- Use sterile WFI or approved rinse medium where required
- Remove residual chemistry from the surface
- Reduce the risk of buildup on equipment and floors
- Allow the area to dry and confirm clean finish
Visual Explanation: Two-Pass Cleaning SOP
A validated cleaning workflow should clearly show sequence, chemistry use, contact time, rinse logic, and final verification steps.

Disinfectant Selection and Rotation Strategy
A disinfectant program should be validated for efficacy, material compatibility, residue profile, and operational fit. One of the most common weaknesses in cleanroom programs is over-reliance on a single chemistry without a structured rotation strategy.
| Disinfectant Consideration | ਇਹ ਮਾਇਨੇ ਕਿਉਂ ਰੱਖਦਾ ਹੈ |
|---|---|
| Efficacy profile | Must be suitable for expected microbial challenges |
| Contact time | Short contact times may reduce real-world effectiveness |
| Residue behavior | Some chemistries leave visible or invisible surface residue |
| Material compatibility | Critical for floors, stainless surfaces, windows, and equipment |
| Rotation strategy | Helps strengthen microbial control over time |
Common program weaknesses include using only IPA, failing to verify contact time in practice, and not linking disinfectant selection to validation data.
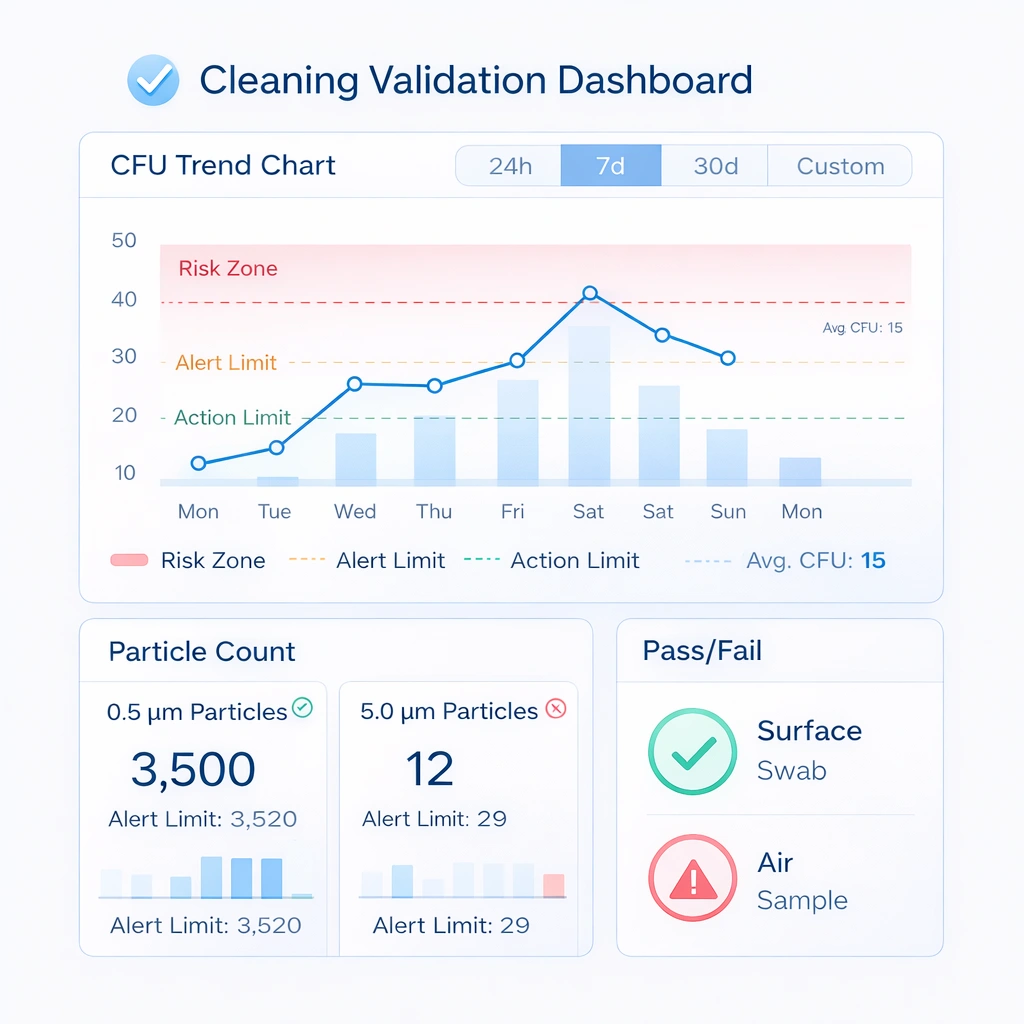
Cleaning Validation Framework
Cleaning is only defensible in GMP when effectiveness is verified. This is where many programs fail: the room may be cleaned on schedule, but there is no structured proof that the cleaning activity actually reduced contamination risk.
MIDPOSI Cleaning Validation Framework
- Visual inspection for residue, streaking, and visible contamination
- Surface microbiological testing using defined sampling points
- Environmental monitoring confirmation where relevant
- Trend review across shifts, rooms, and operators
Visual Explanation: Cleaning Validation Dashboard
A validation dashboard should combine microbial results, post-cleaning particle data, pass/fail status, and recurring trend signals for QA review.
| Validation Method | ਮਕਸਦ | Why It Adds Value |
|---|---|---|
| ਵਿਜ਼ੂਅਲ ਨਿਰੀਖਣ | Immediate surface confirmation | Fast, practical first-line check |
| Surface sampling | Microbial control verification | Confirms whether contamination has been reduced |
| Environmental monitoring review | System-level effectiveness | Shows whether cleaning supports wider contamination control |
Common Cleanroom Cleaning Failures
Most cleaning failures are not caused by missing SOPs. They are caused by weak execution, poor standardization, or lack of validation.
Visual Explanation: Correct vs Incorrect Cleaning Practice
A clear side-by-side view helps operators understand how controlled, unidirectional technique differs from poor posture, random wiping, and incomplete surface coverage.
| Common Failure | Why It Happens | Resulting Risk |
|---|---|---|
| Inconsistent technique | Operators are not trained to one repeatable method | Uneven cleaning performance |
| Wrong disinfectant use | Incorrect dilution, contact time, or rotation | Reduced microbial effectiveness |
| No validation | Cleaning is recorded but not verified | Weak GMP defensibility |
| Poor documentation | Logs lack time, batch, area, or operator traceability | Audit findings and weak investigation support |
| No link to EM | Cleaning program is not reviewed against monitoring trends | Recurring excursions without effective correction |
Documentation and Training Requirements
A GMP cleaning system should never rely on memory or habit. It should be fully supported by documented SOPs, cleaning logs, disinfectant preparation records, validation records, and training documentation.
Documentation should typically include:
- Cleaning logs by area, date, time, and operator
- Disinfectant preparation and expiry records
- Validation results and investigation follow-up
- Operator training and requalification records
Training should cover not only chemistry and frequency, but also movement discipline, wipe or mop technique, residue awareness, and escalation rules when validation fails.
Build a Stronger Cleanroom Cleaning Program with Midposi
Explore GMP-focused cleanroom solutions designed to support contamination control, audit readiness, and more consistent cleanroom operations in pharmaceutical and controlled manufacturing environments.
ਅਕਸਰ ਪੁੱਛੇ ਜਾਂਦੇ ਸਵਾਲ
What is the difference between cleaning and disinfection in cleanrooms?
Cleaning removes particles, residues, and visible contamination. Disinfection reduces microbial burden using validated chemistry. In higher-risk environments, both are required as part of one controlled contamination-control process.
Why is a two-pass cleaning method used in GMP cleanrooms?
A two-pass method separates disinfectant application from final residue removal. This improves consistency, reduces buildup, and supports cleaner surface finish in higher-grade environments.
How often should a cleanroom be cleaned?
Frequency depends on room grade, activity level, and contamination risk. Higher-grade and critical process zones generally require more frequent and more tightly controlled cleaning than support areas.
How do you validate cleanroom cleaning effectiveness?
Typical validation includes visual inspection, defined surface sampling, and review of related environmental monitoring results. Together, these provide stronger evidence than logging alone.
Can one disinfectant be used for all cleanroom situations?
Usually no. Disinfectant strategy should consider efficacy, contact time, residue profile, material compatibility, and whether a rotation approach is required for the environment.
Why do cleaning programs fail even when SOPs exist?
Common reasons include inconsistent operator technique, poor training, weak documentation, incorrect disinfectant use, and lack of validation or trend review.
How does cleaning connect to environmental monitoring?
Recurring EM excursions often indicate that cleaning frequency, chemistry, technique, or validation may not be sufficient. EM data is one of the most useful ways to assess whether a cleaning program is working.
What do auditors usually expect to see in a cleaning SOP program?
Auditors typically expect clear procedures, defined frequencies, validated disinfectant use, operator training records, cleaning logs, and evidence that cleaning effectiveness is reviewed and verified.



