A strong cleanroom MOPS framework helps pharmaceutical and controlled-environment teams standardize gowning, operations, contamination control, environmental monitoring, deviation handling, and continuous improvement.
In regulated cleanrooms, written procedures are not just documentation. They define how people enter, behave, monitor, clean, investigate, and improve within controlled spaces. When MOPS are incomplete, inconsistent, or poorly implemented, the result is not just operational inefficiency. It can directly affect contamination control, audit readiness, training effectiveness, and GMP compliance.
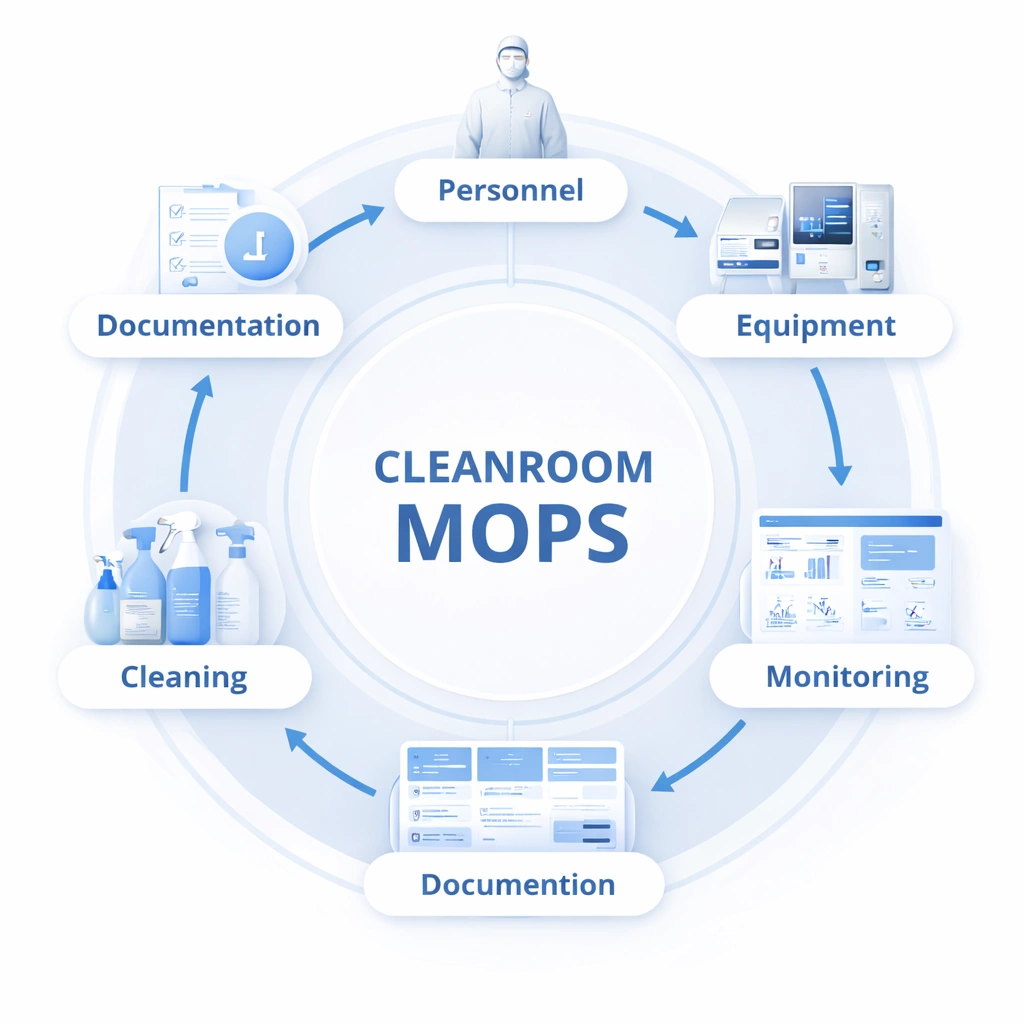
Cleanroom MOPS are structured operating procedures used to control behavior, workflow, hygiene, monitoring, deviation response, and documentation in cleanroom environments. In pharmaceutical and other regulated facilities, an effective MOPS system supports GMP compliance by translating cleanroom requirements into repeatable actions, training standards, records, and corrective workflows.
The best cleanroom MOPS frameworks cover not only what should be done, but also why, by whom, under what conditions, how deviations are handled, and how the system is reviewed and improved over time.
In pharmaceutical manufacturing and other controlled environments, the cleanroom is only as reliable as the procedures that govern it. Your original draft correctly frames cleanroom MOPS as the operational core of daily cleanroom control rather than just a documentation requirement. :contentReference[oaicite:1]{index=1}
A well-designed MOPS system helps reduce variation in how personnel move, gown, clean, monitor, intervene, and react to abnormal events. It also creates a shared operational language between QA, production, engineering, microbiology, and training teams.
A practical cleanroom MOPS system should cover more than one narrow activity. It should connect people, process, environment, records, and corrective action into one coherent operating framework. Your uploaded content points to personnel behavior, equipment use, cleaning, disinfection, and environmental monitoring as the main procedural domains. :contentReference[oaicite:2]{index=2}
Entry, gowning, hygiene, movement restrictions, intervention behavior, and training qualification.
Task execution, equipment handling, cleaning steps, documentation discipline, and escalation paths.
Environmental monitoring, deviation review, audit response, trend evaluation, and CAPA follow-up.
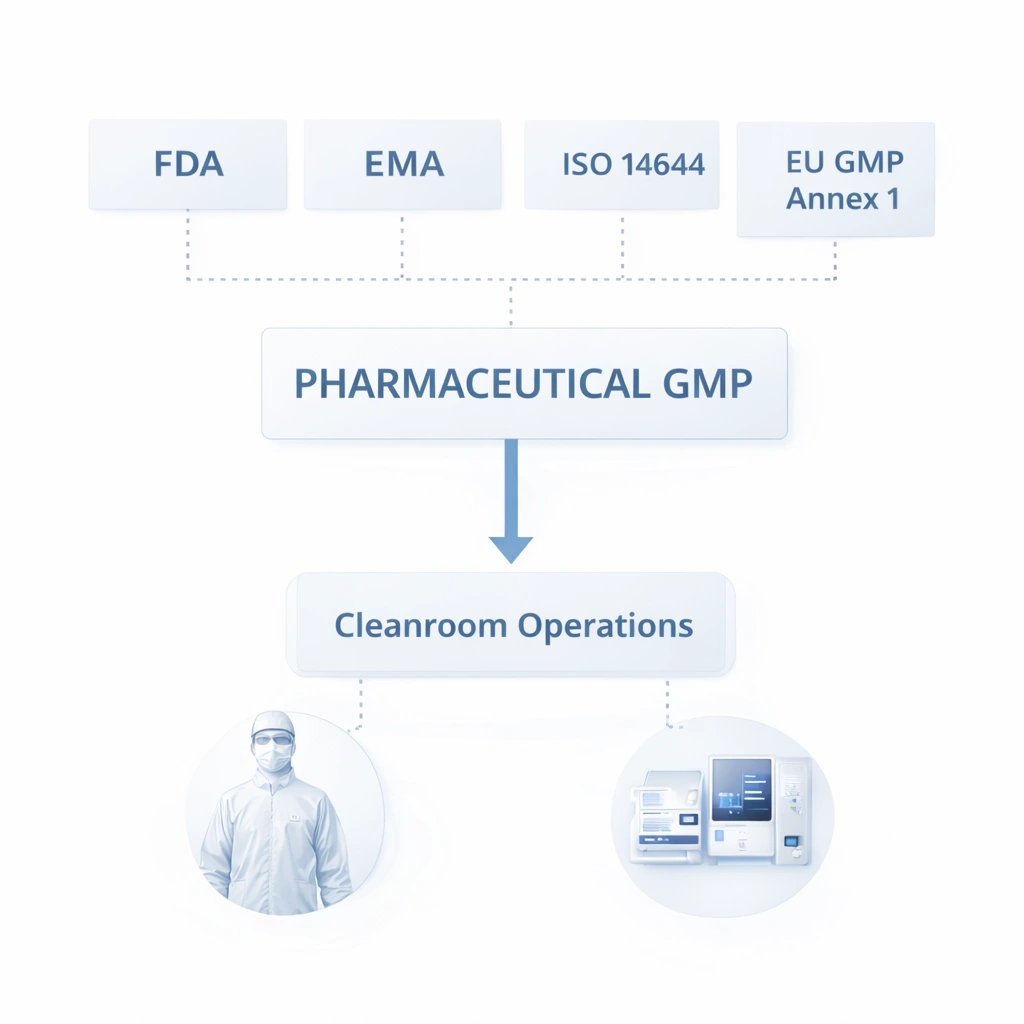
Cleanroom MOPS should align with the broader expectations of GMP, ISO-based environmental control, and site-specific quality systems. Regulatory bodies do not only expect written procedures to exist. They expect those procedures to be appropriate, current, trained, followed, documented, and reviewed.
| ਖੇਤਰ | MOPS Relevance | ਇਹ ਮਾਇਨੇ ਕਿਉਂ ਰੱਖਦਾ ਹੈ |
|---|---|---|
| Personnel Behavior | Defines gowning, movement, hygiene, and interventions | Reduces contamination introduced by human activity |
| Cleaning and Disinfection | Standardizes what, when, how, and by whom | Improves consistency and traceability of cleaning control |
| ਵਾਤਾਵਰਣ ਦੀ ਨਿਗਰਾਨੀ | Defines sampling locations, frequency, action triggers, and response | Supports data-based contamination control decisions |
| Deviation Handling | Clarifies escalation, investigation, and documentation | Prevents confusion during abnormal events |
| CAPA and Review | Links investigation to procedural improvement | Supports continuous improvement and audit readiness |
The strongest procedures are designed for real execution, not just approval. Your source draft highlights key design elements such as scope, purpose, responsibilities, operating steps, quality standards, and deviation handling. :contentReference[oaicite:3]{index=3}
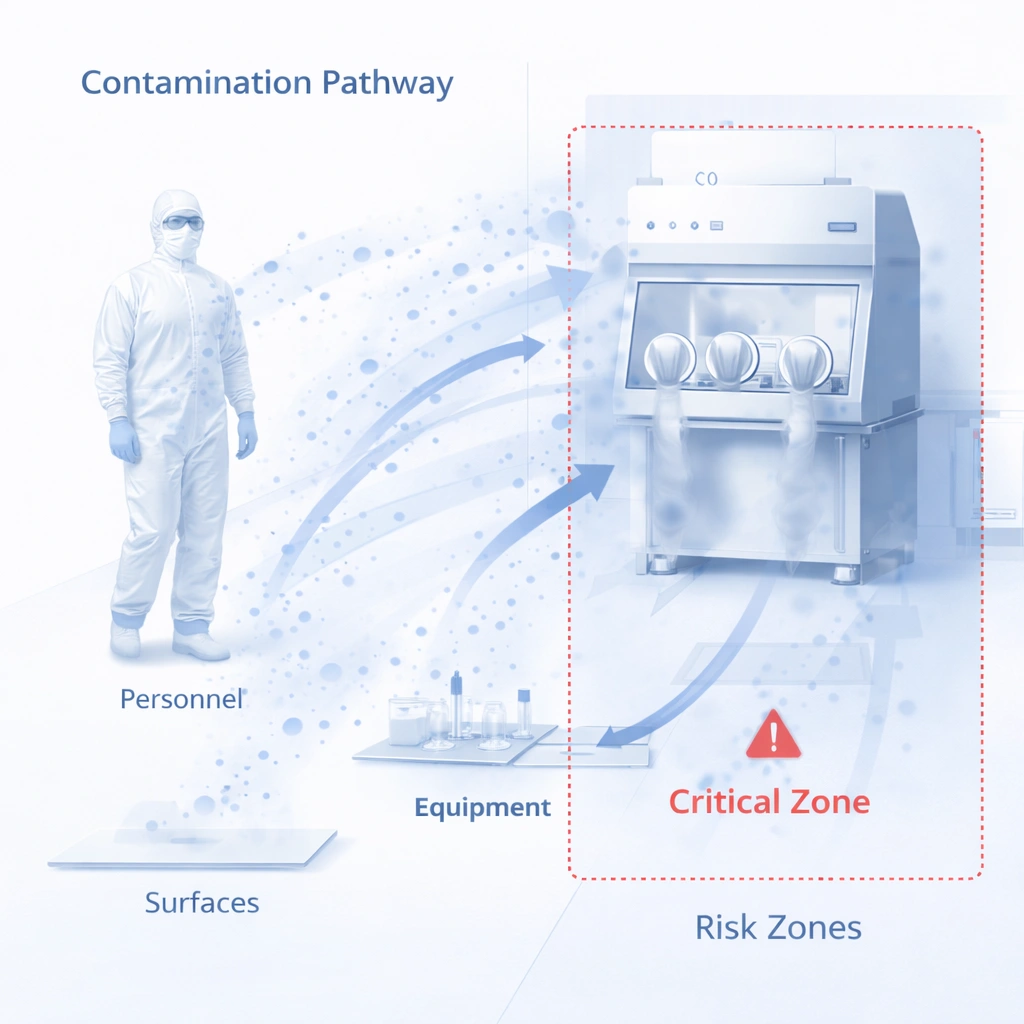
Gowning is one of the clearest examples of why cleanroom MOPS matter. Even strong facility design cannot compensate for inconsistent entry behavior. A strong gowning MOPS should define garment sequence, hand hygiene expectations, entry checks, prohibited actions, and re-entry logic.

Cleanroom MOPS should also define how routine operations are performed under contamination-control expectations. This includes material handling, cleaning sequences, behavior around critical zones, monitoring steps, data review, and response thresholds.
MOPS should specify when and where monitoring is performed, how results are documented, who reviews them, and how action-level events are escalated. This helps ensure that monitoring is used as an operational control rather than just a compliance report.
Standardized procedures reduce operator-to-operator variation and help align production behavior with cleaning, monitoring, and intervention control.

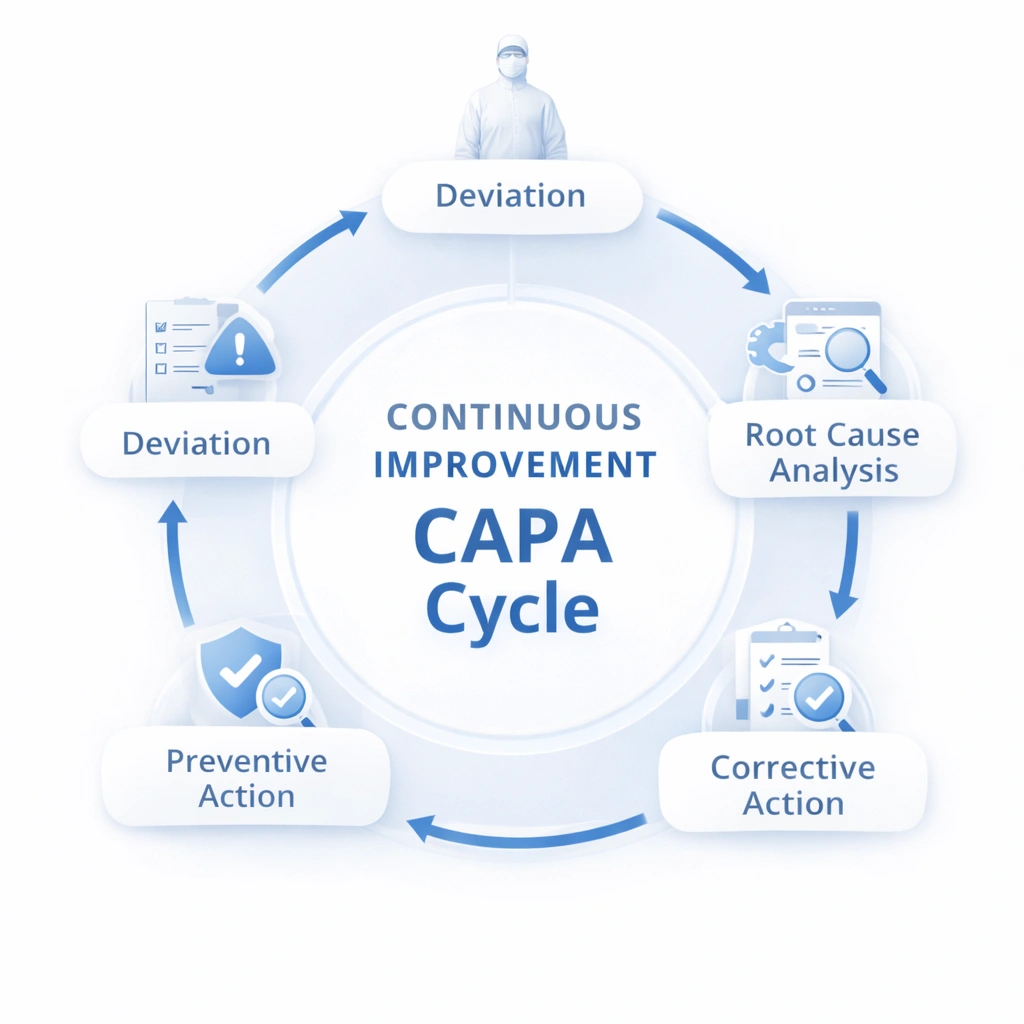
No cleanroom MOPS system is complete if it only describes normal operations. It must also define what happens when conditions are not normal. Your original content correctly highlights deviation handling and CAPA as essential elements of effective cleanroom procedure systems. :contentReference[oaicite:4]{index=4}
Define how personnel report, classify, contain, and document abnormal events affecting cleanroom control.
Link observations, records, monitoring results, and root-cause analysis to determine what failed and why.
Translate findings into corrective and preventive actions, then revise MOPS, training, or controls as needed.
Define the procedure scope, users, risks, records, escalation points, and success criteria for each cleanroom activity.
Train personnel on both the procedural steps and the contamination-control logic behind them.
Review EM trends, deviations, internal audits, CAPA outcomes, and feedback to improve the MOPS framework over time.
Cleanroom MOPS are structured operating procedures used to control personnel behavior, cleaning, disinfection, monitoring, workflow, documentation, and deviation handling in controlled environments.
They help standardize how critical tasks are performed, reduce contamination risk, improve training consistency, and support documentation and audit readiness.
A practical system should include personnel entry procedures, gowning rules, cleaning and disinfection steps, monitoring procedures, deviation handling, and CAPA-linked review.
Effectiveness is typically reviewed through training qualification, audit results, environmental monitoring trends, deviation analysis, and CAPA closure performance.
Review frequency depends on site procedure and risk, but procedures should be revisited after significant process changes, audit findings, repeated deviations, or quality system updates.
ਮਿਡਪੋਸੀ ਨਿਯੰਤ੍ਰਿਤ ਖਰੀਦਦਾਰਾਂ ਨੂੰ ਕਲੀਨ ਰੂਮ ਖਪਤਯੋਗ ਚੀਜ਼ਾਂ ਅਤੇ ਨਿਯੰਤਰਿਤ ਵਾਤਾਵਰਣਾਂ ਲਈ ਦਸਤਾਵੇਜ਼-ਮੁਖੀ ਸੰਚਾਰ ਦੇ ਨਾਲ ਸਮਰਥਨ ਕਰਦਾ ਹੈ, ਜਿਸ ਵਿੱਚ ਯੋਗਤਾ ਸਹਾਇਤਾ, ਸਮੱਗਰੀ ਸਮੀਖਿਆ, ਅਤੇ ਗੰਦਗੀ-ਨਿਯੰਤਰਣ ਚਰਚਾਵਾਂ ਸ਼ਾਮਲ ਹਨ।


ਅਸੀਂ ਤੁਹਾਡੇ ਨਾਲ 1 ਕੰਮਕਾਜੀ ਦਿਨ ਦੇ ਅੰਦਰ ਸੰਪਰਕ ਕਰਾਂਗੇ, ਕਿਰਪਾ ਕਰਕੇ ਪਿਛੇਤਰ ਵਾਲੀ ਈਮੇਲ 'ਤੇ ਧਿਆਨ ਦਿਓ “*@midposi.com”.