Temiz Oda Paspası Nedir?
A cleanroom mop is a specialized floor-cleaning tool engineered to meet the contamination-control demands of controlled environments—pharmaceutical manufacturing suites, biotechnology labs, medical device assembly areas, and semiconductor fabs. Unlike conventional janitorial mops, which are designed primarily for dirt removal and cost efficiency, cleanroom mops are contamination-control devices first and cleaning tools second.
The defining characteristic islow particle generation. Where a standard cotton or rayon mop may shed thousands of fibers and particles with every stroke, a cleanroom mop is constructed from tightly woven, edge-sealed materials (typically microfiber, polyester knit, or non-shedding foam) that generate minimal airborne or surface contamination during use. These mops are tested and qualified to operate within ISO-classified cleanrooms (ISO Class 5 through Class 8) without violating particle count limits or introducing microbial contamination that could compromise sterile manufacturing operations or trigger regulatory findings.
For pharmaceutical and GMP facilities, the cleanroom mop is part of a validated cleaning and disinfection program. It must be compatible with aggressive sporicidal agents (isopropyl alcohol, hydrogen peroxide, quaternary ammonium compounds, sodium hypochlorite), capable of withstanding repeated sterilization cycles where required (autoclaving for Grade A/B areas), and supported by documentation that demonstrates particle shedding performance, chemical resistance, and sterility assurance. In regulatory terms, the cleanroom mop is not a commodity purchase—it is a critical piece of contamination-control infrastructure that must be selected, validated, and maintained according to your facility’s Contamination Control Strategy (CCS) and quality management system.

Figure 1: Aseptic manufacturing environment where cleanroom mops are critical contamination-control devices. Personnel in full sterile gowning demonstrate the high-stakes environment where even minor particle generation from cleaning equipment can compromise product sterility and trigger regulatory findings.
Why Cleanroom Mops Are Different from Normal Mops
The gap between a standard janitorial mop and a cleanroom mop is not a matter of quality grade—it is a difference in design philosophy and performance requirements.
Parçacık üretimi: Conventional mops (cotton, rayon, blended fibers) are optimized for absorbency and scrubbing power. Their fibers are often loosely twisted, cut, or looped, and edges may be unfinished. During use, mechanical friction causes fiber breakage and particle shedding. In a typical office or hospital corridor, this creates no issue. In an ISO Class 7 aseptic processing support area (particle limit: 352,000 particles/m³ at ≥0.5 µm), a shedding mop can single-handedly violate classification limits and trigger an out-of-specification (OOS) event.
Cleanroom mops use continuous-filament polyester, tightly knitted microfiber, or sealed foam structures withheat-sealed or ultrasonically welded edges to prevent unraveling. Particle generation rates are quantified during qualification testing per ISO 14644-14 (assessment of equipment suitability by airborne particle concentration). Acceptable cleanroom mops typically generate fewer than 100 particles ≥0.5 µm per stroke under controlled test conditions, compared to thousands for conventional mops.
Kimyasal uyumluluk: Standard mops are designed for use with neutral pH cleaners, dilute disinfectants, and occasional bleach. Pharmaceutical cleanrooms demand compatibility with high-concentration isopropyl alcohol (70% IPA), hydrogen peroxide (3–35%), sporicidal agents, and contact times measured in minutes rather than seconds. Materials that perform well with household cleaners may degrade, discolor, or shed particles when exposed repeatedly to these aggressive chemistries. Cleanroom mop materials are selected and tested for chemical resistance, dimensional stability, and maintained low-linting performance across hundreds of cleaning cycles and sterilization events.
Sterilization capability: In EU GMP Annex 1 Grade A and Grade B environments (aseptic processing zones, filling lines under unidirectional airflow), all equipment and materials entering the area must be sterilized. Standard mops cannot withstand autoclaving at 121°C or exposure to vaporized hydrogen peroxide without melting, shrinking, or compromising structural integrity. Cleanroom mops intended for these areas are engineered from autoclavable polymers (certain polyesters, polypropylene blends) that survive repeated sterilization cycles without performance loss, or supplied as pre-sterilized, validated single-use systems with sterility assurance documentation.
Traceability and documentation: Cleanroom mops are supplied with certificates of conformance, material safety data sheets, extractables and leachables data (where required), and particle generation test reports. Batch records and lot traceability support regulatory audits and root-cause investigations. Standard mops come with a price tag and little else.

Figure 2: Cleanroom-grade microfiber mop head showing tightly woven, continuous-filament fiber construction. The dense fiber structure provides high absorbency and particle capture while maintaining low linting performance across 100+ sterilization cycles—characteristics that distinguish cleanroom mops from conventional janitorial equipment.
Key Features Required for Pharma & GMP Facilities
When specifying cleanroom mops for pharmaceutical and GMP operations, four core features determine whether a product will support or undermine your contamination control program.
Low-linting & non-shedding structure
Particle generation is the primary qualification criterion. EU GMP Annex 1 and FDA aseptic processing guidance require that materials and equipment used in classified areas minimize particle shedding. ISO 14644-14 provides the test framework: mop heads are subjected to simulated-use conditions (wet/dry cycles, mechanical agitation, contact with representative surfaces) while airborne particle counters measure release at ≥0.5 µm and ≥5 µm.
Acceptable cleanroom mops demonstrate particle generation rates well below the classification limits of their intended use area. For ISO Class 5 (3,520 particles/m³ at ≥0.5 µm), mops must generate virtually zero additional particles during use. For Class 7 or Class 8 support areas, slightly higher generation may be tolerable, but any mop that contributes a measurable particle burden risks pushing the environment out of specification during routine operations or bracketing studies.
Construction details matter:continuous-filament fibers (no cut ends to fray),sealed edges (heat-sealed, ultrasonically welded, or enclosed in a border binding),tight knit or weave density (prevents fiber pull-out), andcontrolled laundering or sterilization protocols that do not degrade the material over the product’s qualified use life. Vendors should provide particle generation test data from independent labs or internal quality systems that reference ISO 14644-14 methodology.

Figure 3: Cleanroom mop anatomy showing key contamination-control features. Cross-section reveals continuous-filament fiber structure (no cut ends to fray), heat-sealed edges (preventing unraveling), tight weave density (minimizing fiber pull-out), and sealed handle interface. These design elements work together to achieve particle generation rates below ISO 14644-14 qualification thresholds.
Compatibility with disinfectants (IPA / Quat / Bleach / H₂O₂)
Pharmaceutical cleaning programs rotate or layer multiple disinfectant chemistries to achieve broad-spectrum microbial kill and prevent resistance development. Your mop must survive this chemical gauntlet without performance loss.
Isopropyl alcohol (IPA, 70%): The most common pharmaceutical disinfectant. Many polymers tolerate IPA well, but repeated exposure can cause some materials to swell, stiffen, or lose tensile strength. Microfiber blends and polyester knits generally perform well; certain foam substrates may degrade.
Kuaterner amonyum bileşikleri (kuatlar): Widely used for routine disinfection. Generally well-tolerated by cleanroom mop materials. Residue management is important—quats can leave films that attract particles or interfere with subsequent sporicidal treatments.
Sodium hypochlorite (bleach): Effective sporicidal agent but aggressive. Concentrations above 0.5% can cause color fading, fiber weakening, and accelerated wear in some polyester and microfiber constructions. If bleach is part of your CCS, specify mops validated for bleach compatibility and monitor for performance degradation over the qualified use period.
Hydrogen peroxide (H₂O₂, 3–35%): Increasingly popular for sporicidal bio-decontamination, especially in isolators and RABS. Peroxide is an oxidizer; materials must be tested for resistance to oxidative degradation, color stability, and maintained structural integrity. Some foam-core mops and certain microfiber blends show excellent H₂O₂ compatibility; others do not.
Request chemical compatibility matrices from vendors and validate in-house under your actual use conditions (concentrations, contact times, temperatures, number of cycles). Include visual inspection (color change, fiber damage), mechanical testing (tensile strength retention), and particle generation re-testing after simulated aging.
Autoclavable vs Disposable
Your choice between reusable (autoclavable) and single-use (disposable) mop systems hinges on area classification, throughput, validation complexity, and cost structure.
Autoclavable (reusable) systems: Designed for repeated sterilization at 121°C (or higher) in steam autoclaves. Constructed from polymers that maintain dimensional stability, mechanical strength, and low-linting performance across 50–200+ autoclave cycles (depending on material and vendor qualification). These systems require validated autoclave cycles (temperature, pressure, time, load configuration), documented post-sterilization hold times and sterility assurance, and routine inspection for wear (fiber damage, edge degradation, discoloration). Reusable systems offer lower per-use cost and reduced waste generation but demand robust laundry/sterilization infrastructure, operator training, and periodic requalification.
Disposable (single-use) systems: Supplied pre-sterilized (gamma irradiation or ethylene oxide) in sealed, validated packaging. Used once and discarded. These eliminate laundering complexity, cross-contamination risk between batches or product lines, and the need for in-house autoclave validation. Disposable mops are preferred in high-risk aseptic areas (Grade A filling lines, isolator environments) where any reuse risk is unacceptable, and in facilities with limited sterilization capacity. Trade-offs include higher per-use cost and increased waste volume (though many vendors now offer recycling programs for used cleanroom consumables).
Annex 1 does not mandate one approach over the other; your Contamination Control Strategy should document the rationale, include risk assessments for both options, and define the validation and monitoring requirements for your chosen system.
Mop handle & bucket pairing
Mops do not operate in isolation. The complete cleaning system—mop head, handle, frame, and bucket—must be designed, validated, and maintained as an integrated contamination-control assembly.
Handles and frames: Should be constructed from smooth, non-porous materials (stainless steel, electropolished aluminum, autoclavable polymers) that do not shed particles, resist corrosion from repeated disinfectant exposure, and can be sterilized (for Grade A/B use) or thoroughly disinfected (for Grade C/D use). Telescoping handles and adjustable frames introduce additional crevices and joints that can harbor contamination; fixed-length, sealed-joint designs are preferred in high-grade areas. Ergonomics matter—gowned operators working in positive-pressure suits or isolators need lightweight, balanced handles that minimize fatigue and reduce the risk of uncontrolled movements that generate particle bursts.
Buckets and wringer systems: Must be autoclavable or supplied as sterile disposables for high-grade areas. Bucket material (often stainless steel or autoclavable polypropylene) should resist chemical attack and allow visual confirmation of solution clarity (indicating absence of particulate contamination or microbial growth). Two-bucket systems (one for clean solution, one for waste) prevent cross-contamination and are standard practice in GMP cleanrooms. Some facilities use pre-saturated mop systems (mop heads pre-wetted with disinfectant, sealed in sterile pouches) to eliminate bucket handling entirely in Grade A/B zones.
Color coding and segregation: Implement color-coded systems (e.g., blue for production areas, red for waste handling zones, green for personnel gowning areas) to prevent cross-contamination between classified and non-classified spaces. Color coding must extend to mop heads, handles, frames, and buckets, with documented procedures and operator training to ensure compliance.

Figure 4: Validated mopping procedure in a pharmaceutical cleanroom. Personnel in full protective coverall, hood, and gloves demonstrate proper technique with color-coded equipment and two-bucket system. This integrated approach—qualified mops, validated procedures, trained operators—forms the foundation of effective contamination control in GMP facilities.
Types of Cleanroom Mops (Microfiber / Polyester / Foam-Core / Disposable)
Cleanroom mops fall into four primary categories, each optimized for different contamination-control requirements, chemical exposures, and operational workflows.
Microfiber mops
Microfiber mops use ultra-fine synthetic fibers (typically polyester/polyamide blends) with diameters below 1 denier (roughly 10 micrometers). The high surface-area-to-weight ratio delivers exceptional absorbency and particle capture. Microfiber’s split-fiber construction creates microscopic channels that trap particles mechanically rather than relying solely on chemical cleaning action, making these mops effective for both wet mopping (with disinfectants) and damp dusting (residual particle removal between disinfection cycles).
Advantages: Superior liquid retention (often 6–8 times the mop’s dry weight), excellent particle capture efficiency, durability across 100–200+ launder/autoclave cycles (when properly maintained), and compatibility with most pharmaceutical disinfectants including IPA, quats, and hydrogen peroxide. Microfiber performs well in ISO Class 6–8 environments and is a workhorse for routine cleaning in aseptic processing support areas, gowning rooms, and material airlocks.
Sınırlamalar: Some microfiber blends show reduced performance or fiber damage after repeated exposure to high-concentration bleach (>1%). Microfiber requires validated laundering protocols—improper washing (high heat, fabric softeners, or contaminated laundry equipment) can introduce particles or residues that compromise cleanroom performance. Pre-saturated or disposable microfiber options are available for facilities that prefer to avoid laundry complexity.
Polyester knit mops
Polyester knit mops are constructed from continuous-filament 100% polyester yarns, typically in a tubular knit or flat-weave configuration. Edges are heat-sealed or ultrasonically welded to prevent fraying. These mops emphasize durability, dimensional stability, and chemical resistance.
Advantages: Excellent compatibility with aggressive disinfectants, including sustained exposure to bleach, hydrogen peroxide, and phenolic compounds. Polyester knit withstands autoclaving across 200+ cycles with minimal loss of tensile strength or dimensional change. Low lint generation and high abrasion resistance make polyester knit suitable for ISO Class 5–7 environments, including areas with frequent disinfectant rotation and heavy-use schedules (multiple shifts, high-throughput manufacturing lines). Many vendors offer polyester knit mops validated to ISO 14644-14 with particle generation data supporting use in Grade B and Grade C environments.
Sınırlamalar: Slightly lower absorbency than microfiber (typically 4–6 times dry weight). Polyester knit may feel stiffer than microfiber, which can affect operator preference and maneuverability in tight spaces. Cost per unit is often higher than microfiber, though total cost of ownership may be lower due to extended use life.
Foam-core mops
Foam-core (or sponge-core) mops feature a central foam substrate encased in a non-woven or knit outer layer. The foam core provides structural rigidity and fluid distribution, while the outer layer handles particle capture and surface contact.
Advantages: Uniform fluid distribution across the mop head, reducing streaking and ensuring consistent disinfectant contact. Foam-core designs excel in applications where controlled, even wetting is critical—such as applying sporicidal agents to isolator interiors or cleaning validated surfaces where residue patterns must be minimized. Some foam-core mops are specifically formulated for hydrogen peroxide compatibility and are used in automated bio-decontamination systems (e.g., isolator foggers followed by manual wipe-down with foam-core mops).
Sınırlamalar: Foam substrates vary widely in chemical resistance; not all formulations tolerate repeated bleach or peroxide exposure. Foam can degrade (crumbling, discoloration, loss of structural integrity) over time, especially under aggressive chemical regimens or high autoclave temperatures. Particle generation testing is critical—some foam-core mops generate acceptable particle levels, while others shed foam fragments or fibers from the outer layer. Always request vendor validation data and conduct in-house qualification before deploying foam-core mops in ISO Class 7 or higher environments.
Disposable (single-use) mops
Disposable mops are supplied pre-sterilized, individually packaged, and intended for one-time use. Materials range from non-woven polyester/polypropylene blends to sealed-edge microfiber or foam-core constructions identical to their reusable counterparts, but packaged and sterilized for single use.
Advantages: Eliminate cross-contamination risk between production batches, eliminate the need for validated laundering or in-house sterilization, simplify logistics and inventory management (especially for multi-site organizations), and provide audit-ready documentation (sterility certificates, lot traceability, expiration dating). Disposable mops are the default choice for high-risk Grade A operations, small-batch or clinical-trial manufacturing (where batch-to-batch segregation is paramount), and facilities undergoing cleaning validation or investigating contamination events (disposables remove laundry and reprocessing as variables).
Sınırlamalar: Higher per-use cost (often 3–10× the cost per use of reusable mops, depending on volume and vendor). Increased waste generation raises environmental and disposal costs, though some vendors offer take-back programs and recycling services. Facilities must manage inventory expiration dates and storage conditions to maintain sterility assurance.
Seçim rehberliği: For routine, high-volume cleaning in ISO Class 6–8 environments with robust laundry infrastructure, reusable microfiber or polyester knit mops offer the best cost-performance balance. For Grade A/B aseptic cores, isolators, or operations with limited sterilization capacity, disposable mops reduce risk and operational complexity. Hybrid approaches—reusable mops for Grade C/D support areas, disposables for Grade A/B cores—are common and pragmatic.
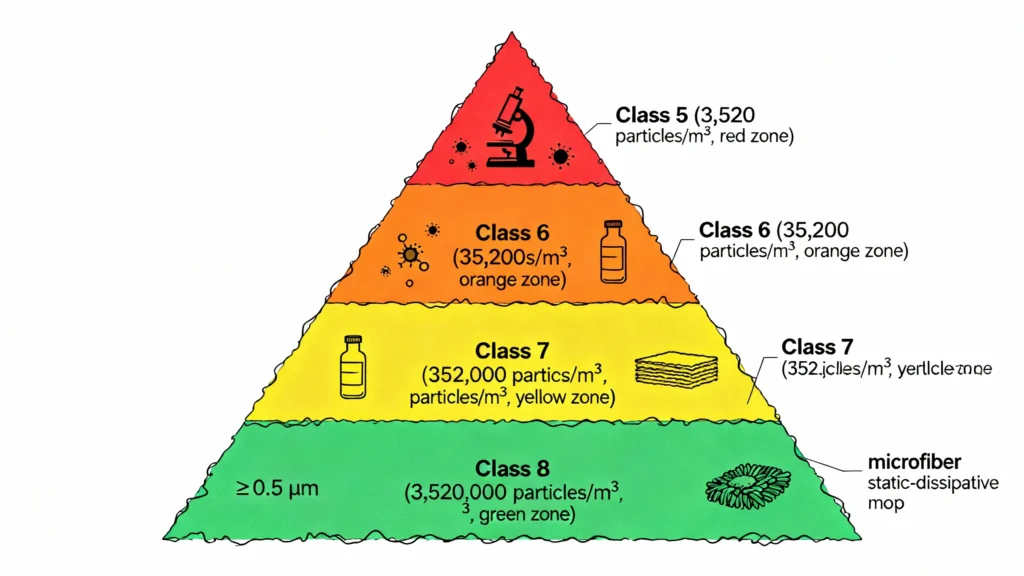
Figure 5: Material comparison matrix for cleanroom mop selection. Each material type offers distinct performance characteristics: microfiber excels in absorbency and particle capture; polyester knit provides superior chemical resistance and durability; foam-core delivers uniform fluid distribution; disposables eliminate cross-contamination risk. Selection should align with area classification, disinfectant program, and operational workflow requirements.
How Cleanroom Mops Help Maintain ISO & GMP Uyumluluğu
Cleanroom mops are not incidental to regulatory compliance—they are integral components of the control systems that keep your facility in specification and audit-ready.
Particle control and ISO classification maintenance: ISO 14644-1 classifies cleanrooms by measured airborne particle concentration. Your facility’s environmental monitoring program continuously tracks particle counts at designated locations, and any excursion above classification limits triggers investigations, corrective actions, and potential production holds. Introducing a non-qualified, particle-shedding mop into a classified area can generate enough particles to push the environment out of spec, especially during high-activity periods (shift changes, material transfers, cleaning operations). Qualified cleanroom mops, tested per ISO 14644-14, provide documented evidence that your cleaning equipment does not contribute to particle burden. This documentation supports your Contamination Control Strategy (CCS) and demonstrates due diligence during inspections.
Microbial contamination control: EU GMP Annex 1 mandates routine microbiological monitoring and sets colony-forming unit (CFU) limits for each cleanroom grade (Grade A: <1 CFU per contact plate; Grade D: ≤100 CFU/m³ active air sample). Mops must not introduce microbial contamination. For Grade A/B environments, sterile mops (autoclaved reusables or pre-sterilized disposables) are required. For Grade C/D areas, mops must be disinfected and managed to prevent microbial growth during storage. A contaminated mop—whether from inadequate sterilization, improper storage, or use beyond its validated shelf life—can seed microbial contamination across floors, walls, and equipment, leading to failed environmental monitoring results, product holds, and regulatory observations.
Cleaning validation and residue management: Annex 1 and FDA CGMP regulations require validated cleaning procedures for equipment and facility surfaces. Your cleaning validation protocol must demonstrate that cleaning agents and methods consistently reduce microbial and particulate contamination to acceptable levels, and that residues from cleaning agents do not adversely affect product quality. The mop is a critical variable in this validation: its absorbency, particle capture efficiency, and compatibility with disinfectants directly affect cleaning efficacy. Mop material must not leach substances that interfere with disinfectant activity or leave residues that attract particles. Validation studies often include worst-case testing with the actual mops, disinfectants, and surface materials used in production, providing documented evidence that the complete cleaning system meets acceptance criteria.
Audit readiness and traceability: During FDA inspections, EMA audits, or third-party certifications, inspectors examine your CCS, cleaning procedures, equipment qualification records, and environmental monitoring trends. They expect to see documented rationale for equipment selection, vendor certificates of conformance, particle generation test data, autoclave validation (for reusable systems), sterility assurance documentation (for disposables), and evidence of operator training. Cleanroom mops with full traceability (lot numbers, material certifications, sterilization records) provide the documentation trail that closes out inspection observations and demonstrates systematic contamination control.
Regulatory expectations across regions: FDA’s 2004 Aseptic Processing Guidance emphasizes environmental control, personnel training, and validated cleaning for aseptic manufacturing. EU GMP Annex 1 (2022 revision) requires a facility-wide CCS, explicit controls for cleaning and disinfection, and qualification of materials and equipment used in classified areas. PIC/S (Pharmaceutical Inspection Co-operation Scheme) and WHO GMP guidelines align closely with these expectations. In all cases, the cleanroom mop is part of the control system that must be designed, validated, and maintained to regulatory standards. Failure to qualify cleaning equipment has been cited in FDA 483 observations and EMA deficiency letters, often linked to environmental monitoring excursions or contamination events during product campaigns.
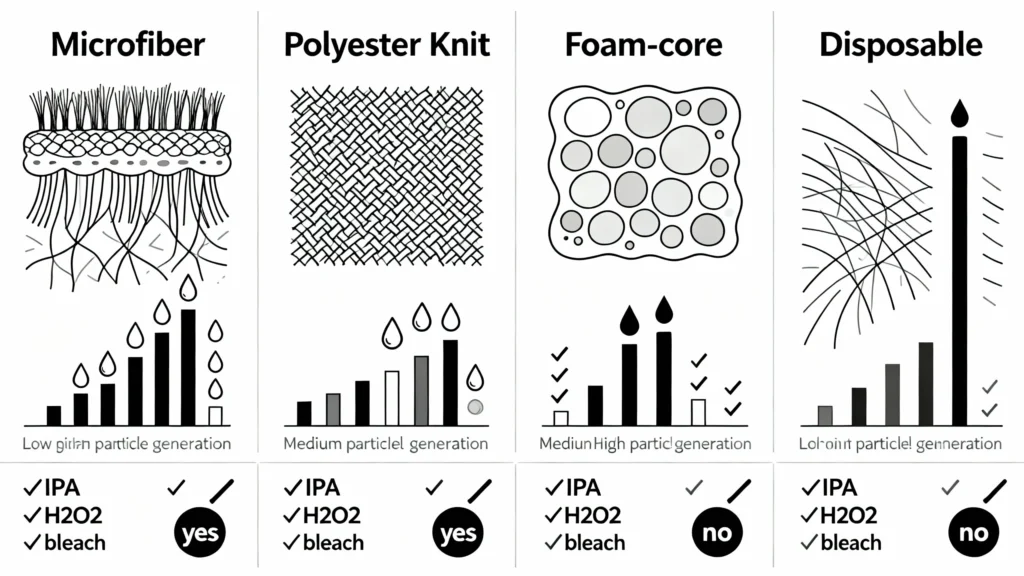
Figure 6: ISO 14644-1 cleanroom classification and corresponding mop requirements. Higher-grade environments (Class 5, Grade A/B) demand sterile mops with near-zero particle generation, while lower-grade support areas (Class 7-8, Grade C/D) can accept validated reusable systems. Particle count limits shown at ≥0.5 µm per cubic meter; these thresholds drive equipment qualification criteria and validation protocols.
How to Choose the Right Mop for Your Facility
Selecting cleanroom mops is a multi-variable decision that balances contamination risk, operational complexity, regulatory requirements, and cost structure. Use this framework to guide your specification process.
1. Define area classification and contamination riskStart with your cleanroom classification map. ISO Class 5 / EU GMP Grade A and B environments (aseptic cores, filling lines, lyophilization loading) demand the highest level of contamination control: sterile mops (autoclaved reusables or pre-sterilized disposables), materials validated to generate <10 particles ≥0.5 µm per stroke, and full traceability. ISO Class 6–7 / Grade C areas (aseptic processing support zones, gowning rooms, material prep) can often use reusable microfiber or polyester knit mops with validated laundering and disinfection protocols. ISO Class 8 / Grade D areas (final packaging, non-sterile compounding) may accept less stringent specifications, but mops must still demonstrate low linting and compatibility with facility disinfectants.
2. Map your disinfectant programList all cleaning and disinfection agents used in each area: IPA concentrations, quat formulations, bleach protocols, hydrogen peroxide schedules. Cross-reference with vendor chemical compatibility matrices. If your CCS includes aggressive oxidizers (bleach >0.5%, H₂O₂ >10%) or requires daily sporicidal treatment, prioritize polyester knit or foam-core mops with validated chemical resistance data. If you rotate disinfectants to prevent microbial resistance, confirm that your chosen mop material tolerates all agents in the rotation without cumulative degradation.
3. Evaluate reusable vs disposable economics and infrastructureFor Grade A/B areas, disposable mops eliminate sterilization complexity and cross-contamination risk, but cost 3–10× more per use. Calculate annual consumption (number of areas × cleaning frequency × mop heads per cleaning event) and compare total cost of ownership: reusable mops require laundering infrastructure, autoclave capacity, validation studies, and operator training, but offer lower per-use cost at scale. For multi-product facilities or clinical trial manufacturers where batch segregation is critical, disposables may justify the premium through risk reduction and simplified logistics.
4. Assess operational workflows and ergonomicsConsider operator experience. Gowned personnel working in Grade A environments (full sterile suits, positive-pressure hoods) need lightweight, balanced mop systems that minimize fatigue and reduce the risk of uncontrolled movements. Telescoping handles introduce crevices; fixed-length handles sealed at joints are preferred. If your facility uses pre-saturated mop systems or sterile pouches, confirm that packaging integrates smoothly with gowning and material transfer procedures (e.g., outer pouch removed in airlock, inner sterile pouch opened inside classified area).
5. Demand documentation and qualification dataRequest from vendors: particle generation test reports (per ISO 14644-14), chemical compatibility studies (exposure concentrations, number of cycles, visual and mechanical testing), autoclave validation data (cycles to failure, dimensional stability), sterility assurance documentation (for disposables: sterilization method, D-value data, sterility test results), material certifications (FDA Drug Master File numbers, EU Pharmacopeia compliance), and extractables/leachables testing (if mops contact product-contact surfaces or are used in isolators where residues could migrate). Reputable vendors provide this documentation as standard; if a vendor cannot supply it, look elsewhere.
6. Pilot and validate before full deploymentRun pilot studies in representative areas before committing to facility-wide adoption. Include: environmental monitoring (particle counts before, during, and after mopping; microbial surface sampling post-cleaning), operator feedback (ease of use, perceived effectiveness, ergonomics), visual inspection of mops after multiple use cycles (fiber integrity, edge condition, discoloration), and integration with existing workflows (compatibility with current buckets/handles, fit with gowning procedures, storage and disposal logistics). Document findings in your CCS and use pilot data to support vendor selection and regulatory justification.

Figure 7: Complete mop system pairing by cleanroom grade. Grade A/B aseptic cores require sterile disposables with pre-saturated pouches and stainless-steel components; Grade C support zones use autoclavable microfiber with two-bucket systems; Grade D packaging areas can accept standard reusable configurations with validated disinfection protocols. Color-coding across all components prevents cross-contamination between classified and non-classified spaces.
MIDPOSI Cleanroom Mop Solutions
MIDPOSI specializes in contamination-control consumables for pharmaceutical, biotechnology, and medical device manufacturing. Our cleanroom mop portfolio is engineered to meet the demanding requirements of ISO-classified environments and GMP operations, with a focus on validated performance, regulatory documentation, and reliable supply-chain support.
Product range: MIDPOSI offers microfiber, 100% polyester knit, and foam-core mop heads in both reusable (autoclavable) and pre-sterilized disposable configurations. Our reusable mops are validated for 150+ autoclave cycles at 121°C with maintained particle generation performance and dimensional stability. Disposable mops are gamma-sterilized and individually packaged with lot-specific sterility certificates and expiration dating.
Validated performance: All MIDPOSI cleanroom mops undergo particle generation testing per ISO 14644-14, with test reports provided for each product line. Typical particle generation rates are <50 particles ≥0.5 µm per stroke for microfiber and polyester knit mops, supporting use in ISO Class 5–8 environments. Chemical compatibility has been validated across 70% IPA, 3–10% hydrogen peroxide, quaternary ammonium compounds (up to 2,000 ppm), and sodium hypochlorite (up to 1%), with visual, mechanical, and particle generation testing after 100 simulated-use cycles.
Complete system solutions: MIDPOSI provides integrated mop systems including autoclavable stainless-steel handles with sealed joints, color-coded mop frames (preventing cross-contamination between areas), and autoclavable or disposable bucket systems. Pre-saturated mop options (mop heads pre-wetted with 70% IPA, sealed in double-pouch sterile packaging) streamline workflows in Grade A/B environments and eliminate the need for on-site solution preparation.
Regulatory documentation: Every MIDPOSI product shipment includes certificates of conformance, material safety data sheets, particle generation test summaries, and (for disposables) sterility assurance documentation traceable to gamma irradiation dose records. Our quality management system is ISO 13485-certified, and select products are registered with FDA Drug Master Files to support customer regulatory filings.
Technical support and validation assistance: MIDPOSI’s technical team works with facility QA and engineering groups to support pilot studies, cleaning validation protocols, and regulatory justification documentation. We provide sample kits for in-house evaluation, on-site training for cleaning personnel, and customized product configurations (size, material, sterilization method) to meet specific facility needs.
For more information on MIDPOSI cleanroom mop solutions, request a product catalog and technical specification package through our website or contact your regional representative.









