A technical evaluation of Gamma Irradiation versus Autoclave sterilization for Annex 1 compliant cleanroom environments.

Under EU GMP Annex 1, mopping systems form a critical part of the Kontaminationskontrollstrategie (CCS). The selected sterilization method directly affects audit exposure, validation workload, and transfer risk for pharmaceutical facilities.
As explained in our pharmaceutical cleanroom mop supplier selection guide , sterility alone is not sufficient — material stability, packaging integrity, and documentation traceability must also be demonstrated.
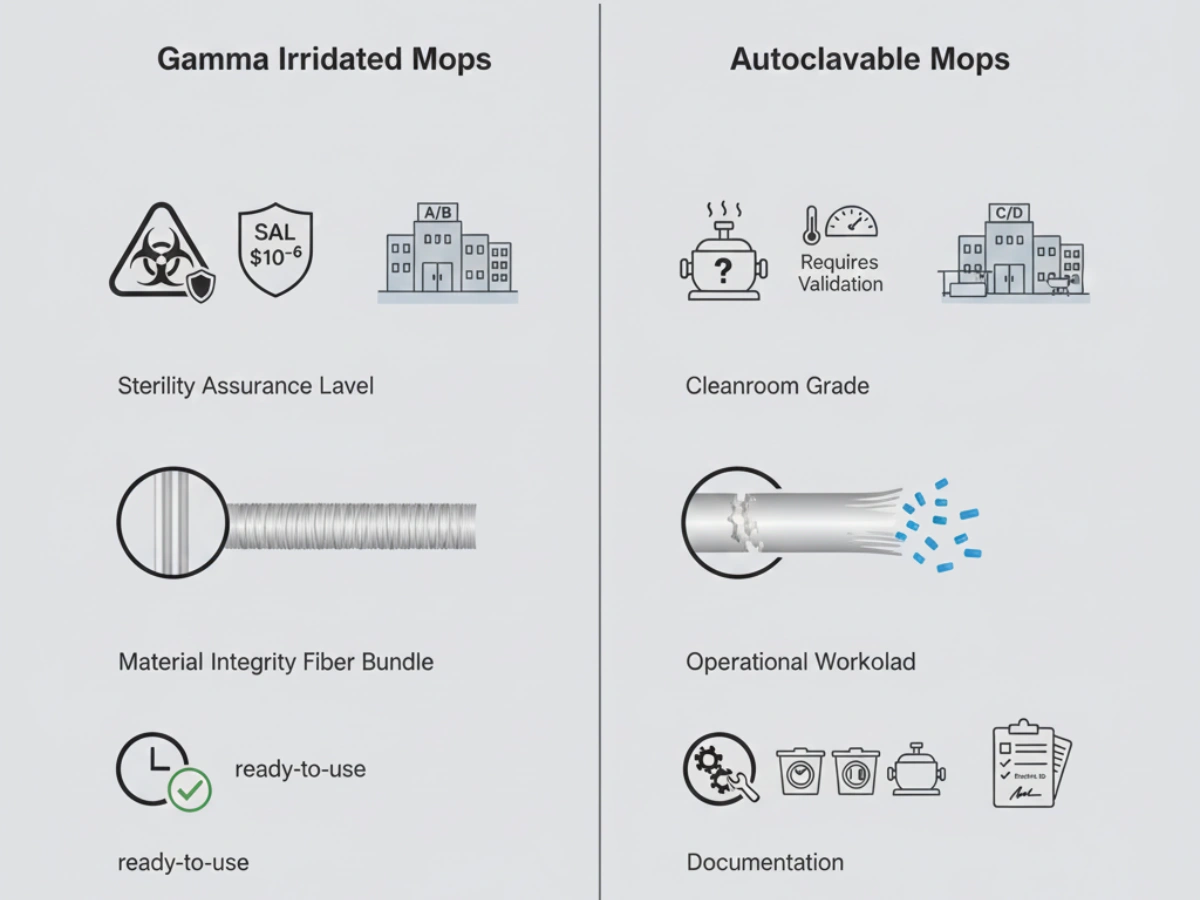
Gamma irradiation is a terminal "cold" sterilization process. Its penetrative nature ensures uniform sterilization throughout dense mop structures. Each batch is validated to SAL 10⁻⁶ and supplied with a batch-specific COI.

Autoclave sterilization relies on saturated steam (121 °C). Effective autoclaving requires dense mop heads to undergo validated vacuum cycles to eliminate trapped air and prevent cold spots.

Facilities must validate the maximum allowable cycles before particle shedding exceeds GMP limits.
| Besonderheit | Gamma Irradiated Mop | Autoclavable Mop |
|---|---|---|
| Sterility Assurance | Validated SAL 10⁻⁶ per batch | Cycle-dependent; In-house validation |
| Grade Suitability | Optimized for Grade A/B | Typically Grade C/D |
| Materielle Integrität | High (Single-use, no wear) | Degrades with each cycle |
This risk is most visible during material transfer into Grade A/B areas. For a detailed explanation, refer to our dedicated guide on double-bagged sterile cleanroom mop packaging and transfer logic .
From a QA and operations perspective, sterilization method selection should align with cleanroom grade, internal validation capability, and supplier documentation maturity.
For validation-driven facilities, we support technical review with batch documentation, sterilization summaries, and evaluation samples.
Request Technical Data or Samples

Wir werden uns innerhalb eines Werktages mit Ihnen in Verbindung setzen. Bitte beachten Sie die E-Mail mit dem Suffix „*@midposi.com“.