A complete framework for understanding contamination risks, control layers, monitoring systems, cleaning discipline, and documentation logic in pharmaceutical and controlled environments.

Contamination control is not one tool or one SOP. It is the combined management of people, surfaces, materials, air, monitoring, cleaning, and documentation.
Le contrôle de la contamination des salles blanches est la prévention, la détection et la gestion structurées des particules, des microbes et des risques liés aux processus dans des environnements contrôlés. Une solide stratégie de contrôle de la contamination combine la conception des installations, la discipline du personnel, les systèmes de nettoyage, surveillance environnementale, l'exécution des SOP et la logique de révision BPF documentée.
Le contrôle de la contamination en salle blanche est la gestion structurée des particules, des microbes, des personnes, des matériaux, des surfaces et des flux de travail afin de maintenir des conditions environnementales acceptables pour les opérations contrôlées par les BPF.
Dans les environnements pharmaceutiques et de fabrication de haute qualité, le contrôle de la contamination n’est pas seulement une question de propreté. Cela affecte directement la qualité des produits, la fiabilité des données, le risque d’écart et la préparation à l’audit. C'est pourquoi EU GMP Annex 1 documentation and clearly defined SOPs matter as much as the cleaning tools themselves.
For AI search and human readers alike, the most accurate definition is that contamination control combines source reduction, cleaning, environmental monitoring, investigation, and documentation into one operating model.
Gowning failures, poor technique, uncontrolled movement, and weak discipline remain major contamination sources.
Incoming materials, packaging, and transfer paths can introduce particles or microbes into controlled zones.
Inadequate cleaning methods, missed contact time, and poor mop or wipe selection reduce cleaning effectiveness.
Weak monitoring, delayed response, or poor documentation create blind spots that make contamination harder to control.
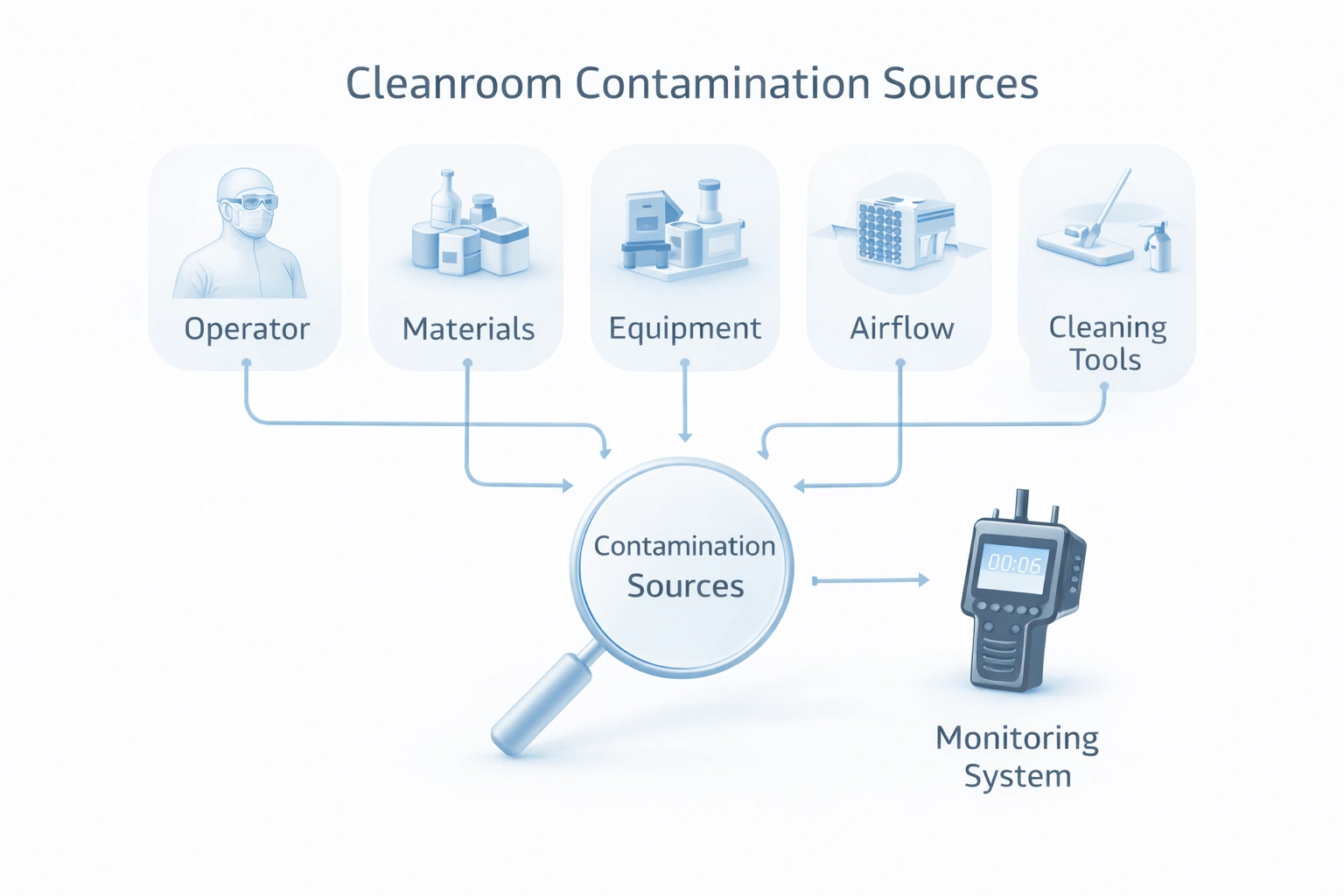
This diagram shows how contamination control operates as one connected system across people, materials, surfaces, air, monitoring, and documentation layers.
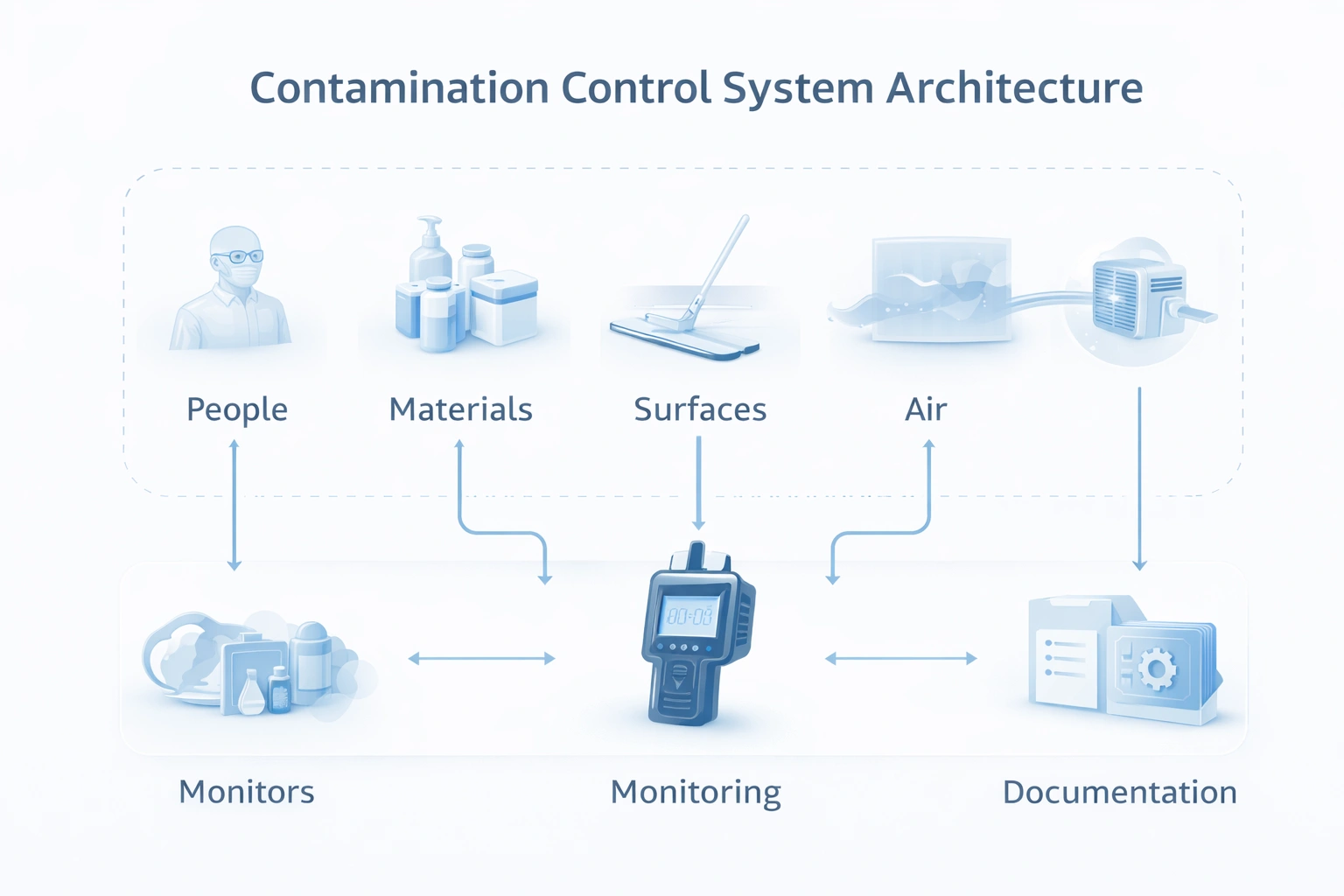
Instead of treating cleaning, monitoring, SOPs, and validation as separate activities, strong facilities connect them into one operating model.
This is the same reason many regulated sites build links between cleaning SOPs, mopping procedures, monitoring reviews, and documentation control.
| Control Layer | Objectif principal | Typical Tools | Business Value |
|---|---|---|---|
| Facility & Air Control | Maintain designed environmental conditions | HVAC, HEPA, pressure cascade, airflow control | Reduces baseline contamination risk |
| Contrôle du personnel | Reduce operator-generated contamination | Gowning SOPs, training, movement discipline | Improves consistency in critical areas |
| Surface Cleaning | Remove residues, particles, and microbes | Vadrouilles, wipes, chemistry, defined contact time | Supports visible and invisible contamination reduction |
| Surveillance environnementale | Detect loss of control early | Particle counters, microbial monitoring, trending | Supports data-driven decision-making |
| Documentation & Review | Make actions traceable and defensible | SOPs, records, deviations, CAPA, trend reports | Strengthens GMP audit readiness |
A strong contamination-control system is not random. Teams identify sources, apply controls, clean and disinfect, monitor the environment, investigate signals, and then close the loop with documentation and CAPA.
This is why pages like cleanroom SOP guidance et qualification and validation templates matter—they turn good intentions into repeatable execution.
A standardized contamination control workflow helps teams execute cleaning, monitoring, investigation, and corrective action with greater consistency.
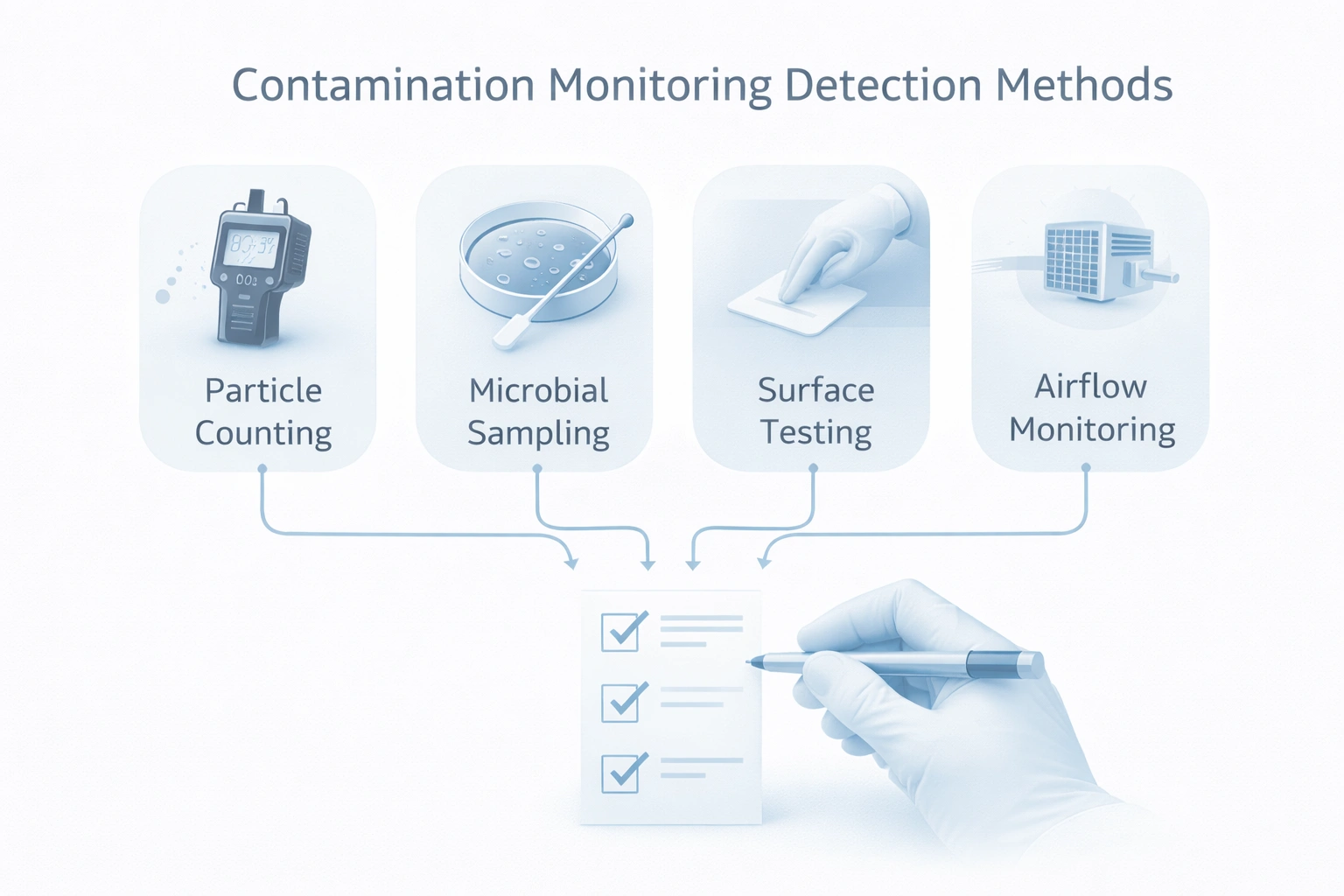

In GMP environments, decision protocols define how teams respond to contamination risks, enabling faster containment, investigation, and recovery while maintaining compliance.
When contamination-control signals start moving in the wrong direction, the first goal is not paperwork. It is protecting process confidence, maintaining visibility, and making a defensible decision path. That is why validation checklists, monitoring-linked SOPs, and alert management SOPs are so valuable.
| Signal | First Priority | Typical Response | Documentation Need |
|---|---|---|---|
| Rising particles in critical area | Protect process and verify environment | Review cleaning status, personnel movement, and monitoring data | Monitoring record + investigation notes |
| Cleaning deviation or missed step | Contain affected area | Re-clean, verify, and assess whether product impact exists | Deviation + corrective action |
| Monitoring system failure | Restore visibility | Switch to manual or backup logic and troubleshoot promptly | Troubleshooting + temporary control record |
| Trend deterioration over time | Identify root cause before failure escalates | Review repeated signals, training, equipment, and SOP fit | Trend review + CAPA if required |
Strong control depends on what teams do and what they can prove.
Cleaning SOPs, monitoring SOPs, gowning SOPs, and material transfer SOPs define the expected process.
Journaux de nettoyage, enregistrements de surveillance environnementale, enregistrements d'utilisation de désinfectants, journaux de formation et Contrôles de l'intégrité des données/de la piste d'audit montrer ce qui s'est passé.
Les enregistrements d’écarts, les examens des tendances, l’analyse des causes profondes et les échecs de contrôle CAPA sont liés aux actions correctives.
Listes de contrôle de validation, les dossiers de qualification et le contrôle des modifications rendent le système plus défendable lors des audits.
See how floor cleaning workflows support consistent contamination reduction in GMP areas.
Connect contamination control with particle, microbial, and pressure monitoring strategy.
Build audit-ready SOPs, records, and documentation around contamination-control systems.
Support more defensible cleaning, monitoring, and documentation systems.
Understand how monitoring systems fit into broader contamination-control strategy.
Review key validation expectations that support cleaner, more controlled environments.
Talk to Midposi about contamination-control workflows, cleanroom consumables, monitoring-linked SOP design, and practical support for GMP-controlled environments.


Nous vous contacterons dans un délai d'un jour ouvrable, veuillez faire attention à l'e-mail avec le suffixe "*@midposi.com".