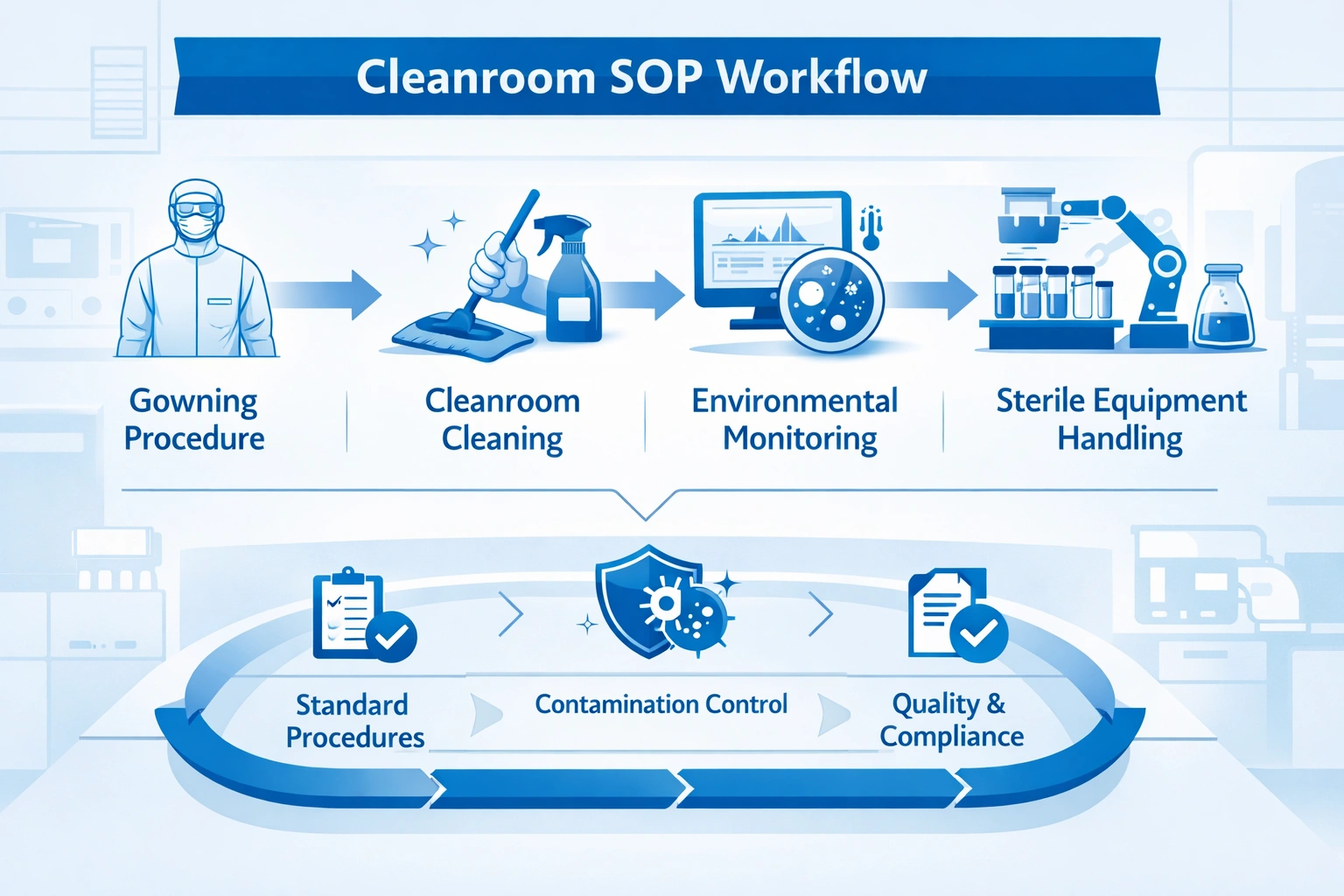
Typical SOP categories
- Gowning and personnel entry
- Cleaning and disinfection
- Environmental monitoring
- Material and equipment transfer
- Batch area clearance
- Deviation and incident response
Pharmaceutical Cleanroom SOP Guide
A practical guide to cleanroom standard operating procedures, contamination control, sterile cleaning workflows, environmental monitoring, GMP documentation, and future AI-driven quality systems.

In pharmaceutical and biotechnology manufacturing, cleanroom standard operating procedures are not just compliance documents. They are the operational backbone for contamination control, sterile behavior, validated cleaning, material transfer, and environmental consistency.
The strongest cleanroom SOP systems help manufacturers reduce deviation risk, improve audit readiness, standardize operator behavior, and protect product quality in ISO 5, ISO 7, and GMP-controlled spaces.
If you are building or upgrading a pharmaceutical cleaning program, it is also useful to review related guidance on cleanroom mopping SOPs, pharmaceutical cleanroom mop cleaning SOPs, and EU GMP Annex 1 cleanroom SOP documentation.
A cleanroom SOP is a documented procedure that defines how a specific activity must be performed inside a controlled environment. These activities usually include gowning, room entry, equipment handling, cleaning and disinfection, material transfer, line clearance, and environmental monitoring.
In pharmaceutical manufacturing, a well-written SOP does more than describe a task. It defines sequence, frequency, materials, approved tools, operator responsibilities, escalation rules, and documentation requirements.
Teams that are still structuring their documentation framework may also find it helpful to review cleanroom qualification and validation SOP templates, FDA cGMP cleanroom validation checklists, and risk-based cleanroom environmental monitoring SOP guidance.
Most contamination issues do not begin with dramatic equipment failure. They begin with inconsistency: an incomplete wipe-down, an incorrect mop path, poor gown discipline, unverified transfer steps, or environmental data that was not acted on in time.
That is why cleanroom SOPs matter. They turn contamination control from individual habit into repeatable system behavior.
For teams focused on Annex 1 readiness, aseptic zones, and documentation control, related reading includes cleanroom microbial contamination control for isolator systems, microbial contamination control in cleanroom isolator systems, and cleanroom decontamination and sterilization SOPs for batch processing.
A practical cleanroom SOP framework usually follows a defined operational chain. Although every facility has its own validation requirements, the core structure is often similar.
This workflow becomes much stronger when paired with a validated product system. For example, many pharmaceutical teams compare disposable vs reusable cleanroom mops, evaluate gamma-irradiated vs autoclavable cleanroom mops, and study low-lint cleanroom mop material comparisons before finalizing SOP-approved tools.
Cleaning is one of the most critical parts of any cleanroom SOP program. The procedure must specify not only what to clean, but how to clean it, with what material, at what frequency, and with what documented method.

Requires validated wipes, defined wipe pattern, and appropriate disinfectant contact time.
Requires a controlled mop path, approved mop head, and clear dirty-to-clean direction logic.
Sterile mops, low-lint wipes, and compatible bucket systems support repeatable contamination control.
If your team is selecting or upgrading cleaning tools, useful internal references include pharmaceutical cleanroom mop requirements, cleanroom mop implementation guides, cleanroom mop bucket systems, and pharmaceutical cleanroom mops for GMP Annex 1 environments.
Supplier qualification also matters. See how to qualify a pharmaceutical cleanroom mop supplier, cleanroom mop validation documents and COA, and batch traceability for pharmaceutical cleanroom mops.
Contamination does not come from one single source. It usually develops through an interaction between personnel, surfaces, airflow, tools, transferred materials, and incomplete execution.

Facilities building contamination control strategy should also review preventing cleanroom contamination risks, lint-free cleanroom wipes for critical zone cleaning, and biotech cleanroom mop applications.
The next stage of cleanroom SOP development is not less documentation. It is smarter control. More pharmaceutical manufacturers are now exploring digital tools that connect SOP execution with real-time data, contamination analytics, and predictive monitoring.

Even in advanced facilities, the foundation still remains the same: a clear SOP, trained personnel, validated materials, and a repeatable contamination control method.
Cleanroom SOPs should be operationally precise, not vague or generic.
Use validated wipes, sterile mops, and compatible systems that support repeatability.
Environmental results should trigger action, not just record keeping.
Operator consistency is one of the strongest contamination control variables.
A cleanroom SOP defines how controlled tasks must be performed to maintain consistency, reduce contamination risk, and support compliance in pharmaceutical or biotech production.
It should include approved materials, cleaning sequence, area frequency, disinfectant type, contact time, tool selection, documentation steps, and responsibilities.
Sterile mops help reduce contamination risk in critical cleaning processes by supporting validated cleaning methods and consistent low-lint performance.
SOPs should be reviewed on a scheduled basis and also updated whenever process changes, deviations, audit findings, or new contamination risks require revision.
No. AI can improve monitoring and decision support, but it does not replace the need for clear written procedures, validated materials, and trained human execution.
Midposi supports pharmaceutical and cleanroom teams with sterile mops, low-lint wipes, and contamination control consumables designed for controlled environments.


Nous vous contacterons dans un délai d'un jour ouvrable, veuillez faire attention à l'e-mail avec le suffixe "*@midposi.com".