Choosing cleanroom mops for ISO 5–8 environments is not about finding the “best” product in isolation. The right choice depends on contamination-control requirements, room classification, disinfectant protocol, sterilization method, operator workflow and total cost of ownership.
The wrong mop can contribute to environmental monitoring excursions, cleaning inconsistency and audit questions. Over-specifying can also waste budget on features that are not necessary for lower-risk areas. This guide compares leading cleanroom mop options and explains how buyers can build a defensible shortlist for pharmaceutical, biotech, medical device and semiconductor cleanrooms.
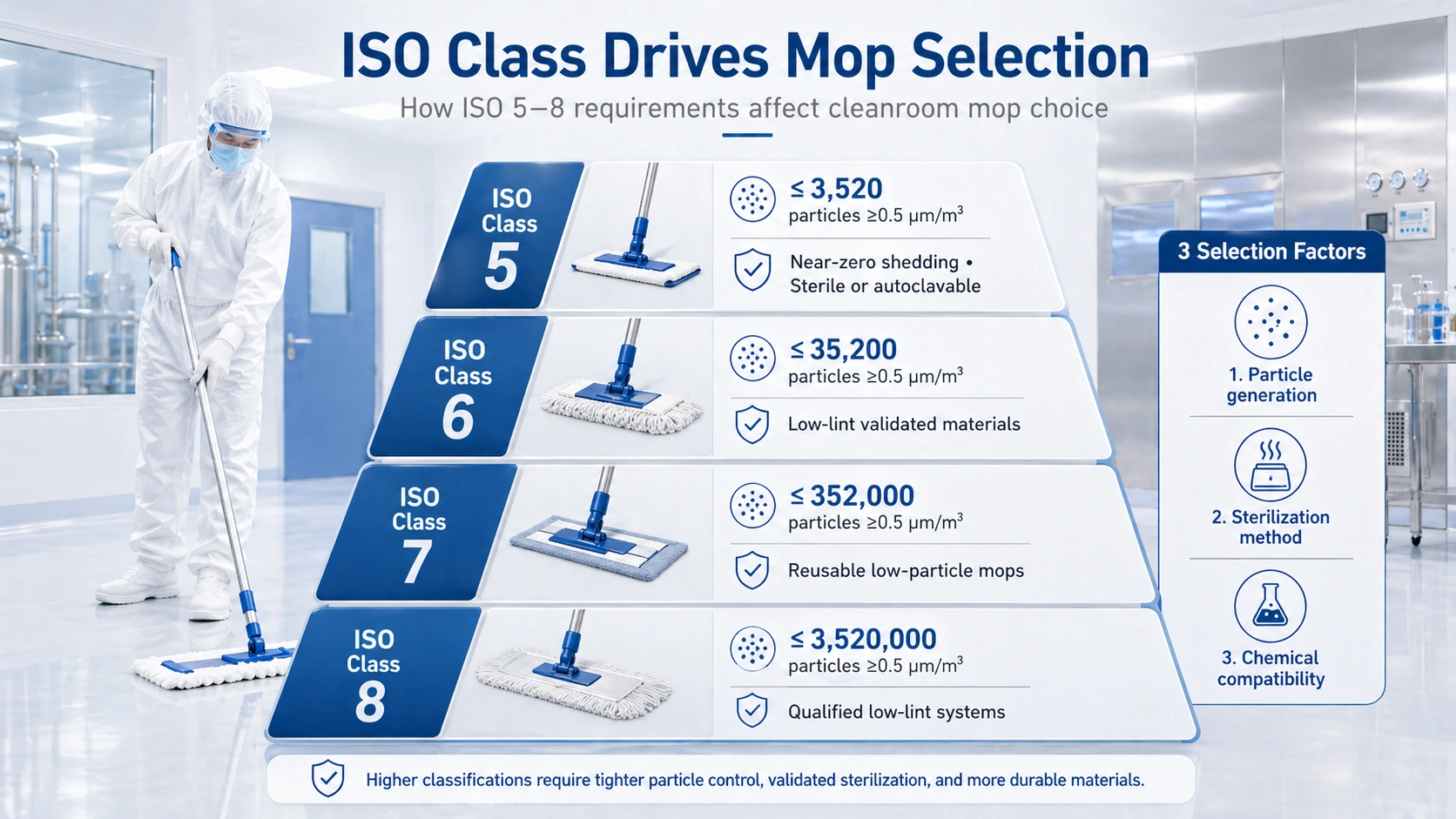
Start with the strictest room class in your facility, then confirm whether the mop must be sterile, autoclavable, reusable, disposable, pre-saturated or compatible with aggressive disinfectants. The mop head, handle, frame and bucket workflow should be reviewed together as a complete cleaning system.
ISO classification defines the particle-control expectations of a cleanroom. The stricter the cleanroom class, the lower the tolerance for particle generation during routine cleaning. A conventional janitorial mop may be acceptable in an office, but it is not suitable for classified environments where particles, fibers and residue can affect product quality or environmental monitoring results.
In cleanroom mop selection, ISO class drives three practical decisions: how low-lint the mop material must be, whether the mop must be sterile or autoclavable, and how much chemical resistance is needed for the facility’s disinfectant program.
A cleanroom mop should not become a particle source during use. Buyers should review whether the mop head uses low-lint fabric, controlled edges, durable yarn structure and cleanroom-compatible packaging. For high-control areas, particle generation documentation and supplier quality records become important parts of vendor evaluation.
Higher-risk pharmaceutical and biotech areas may require sterile or sterilizable cleaning tools. This can mean gamma-sterilized disposable mop heads, autoclavable reusable mop heads, or complete systems that include compatible صاف ڪمرو ايمپ هينڊلز ۽ cleanroom mop frames.
Cleanroom mop materials must tolerate the disinfectants used in the facility. Microfiber, polyester knit, foam-core and string-style polyester all behave differently under repeated exposure to IPA, hydrogen peroxide, sodium hypochlorite and other cleaning agents.
For a component-level comparison, review MIDPOSI’s microfiber cleanroom mop, cleanroom flat mop system, mop frames ۽ mop handles as a complete cleaning assembly.

This guide evaluates cleanroom mop systems across five weighted criteria. These criteria are designed for procurement, QA, facility and contamination-control teams that need to compare products in a structured way.
Procurement tip: do not compare mop heads alone. A qualified cleanroom mop program should include mop heads, handles, frames, bucket workflow, disinfectant compatibility and documentation.

Contec-style foam and microfiber systems are often selected when documentation, sterility assurance and audit readiness matter more than the lowest per-use cost. For pharmaceutical aseptic suites, biologics facilities and clinical trial manufacturing, a turnkey sterile consumable system can reduce the burden of in-house reprocessing validation.
The main limitation is cost. Pre-sterilized or high-documentation mop heads can be more expensive than reusable polyester alternatives. They are best justified where batch segregation, lot traceability and sterility documentation carry direct compliance value.
Large-format polyester pad mop systems are practical for high-volume cleanrooms where coverage efficiency matters. They are especially useful for gowning rooms, airlocks, material staging areas and controlled production zones where facilities already have a defined cleaning and reprocessing workflow.
Double-sided flat mop systems are attractive because they increase usable cleaning surface area. When one side becomes soiled, the operator can use the other side before changing the head. This can reduce downtime and improve coverage efficiency in large cleanrooms.
Foam sponge mop systems can be useful when frequent change-out is part of the SOP. Their absorbency and lower entry cost make them attractive for startup facilities, validation studies, support rooms or operations where simple inventory and frequent replacement are preferred.
String-style polyester mop systems can handle high liquid volumes and irregular surfaces. They are often considered in electronics, semiconductor and large-area controlled environments where absorbency and drying behavior are important. For facilities that prefer easier standardization, a cleanroom flat mop system may provide a more controlled workflow.

Material selection is one of the most important decisions in a cleanroom mop program. Microfiber may provide strong pickup and good surface contact. Knitted polyester can offer durability and chemical resistance. Foam-core mop heads can help with even fluid release. String-style polyester can support high absorbency and fast drying.
| Mop Material | طاقتون | Common Use | Buyer Caution |
|---|---|---|---|
| مائڪرو فائبر | Low lint, strong pickup, good surface contact | ISO 5–7 cleaning, precision wiping and controlled floor cleaning | Check chemical compatibility and laundering behavior. |
| Knitted Polyester | Durable, chemical resistant, suitable for repeatable cleaning programs | ISO 3–8 support and production areas | Confirm edge construction and cycle life. |
| Foam Core | Even fluid release and good wet contact | Regulated pharma, disinfectant application and sterile-friendly workflows | Review compatibility with oxidizers and repeated sterilization. |
| String-Style Polyester | High absorbency and fast drying | Electronics, semiconductor and large-area mopping | Requires good laundering and drying control. |
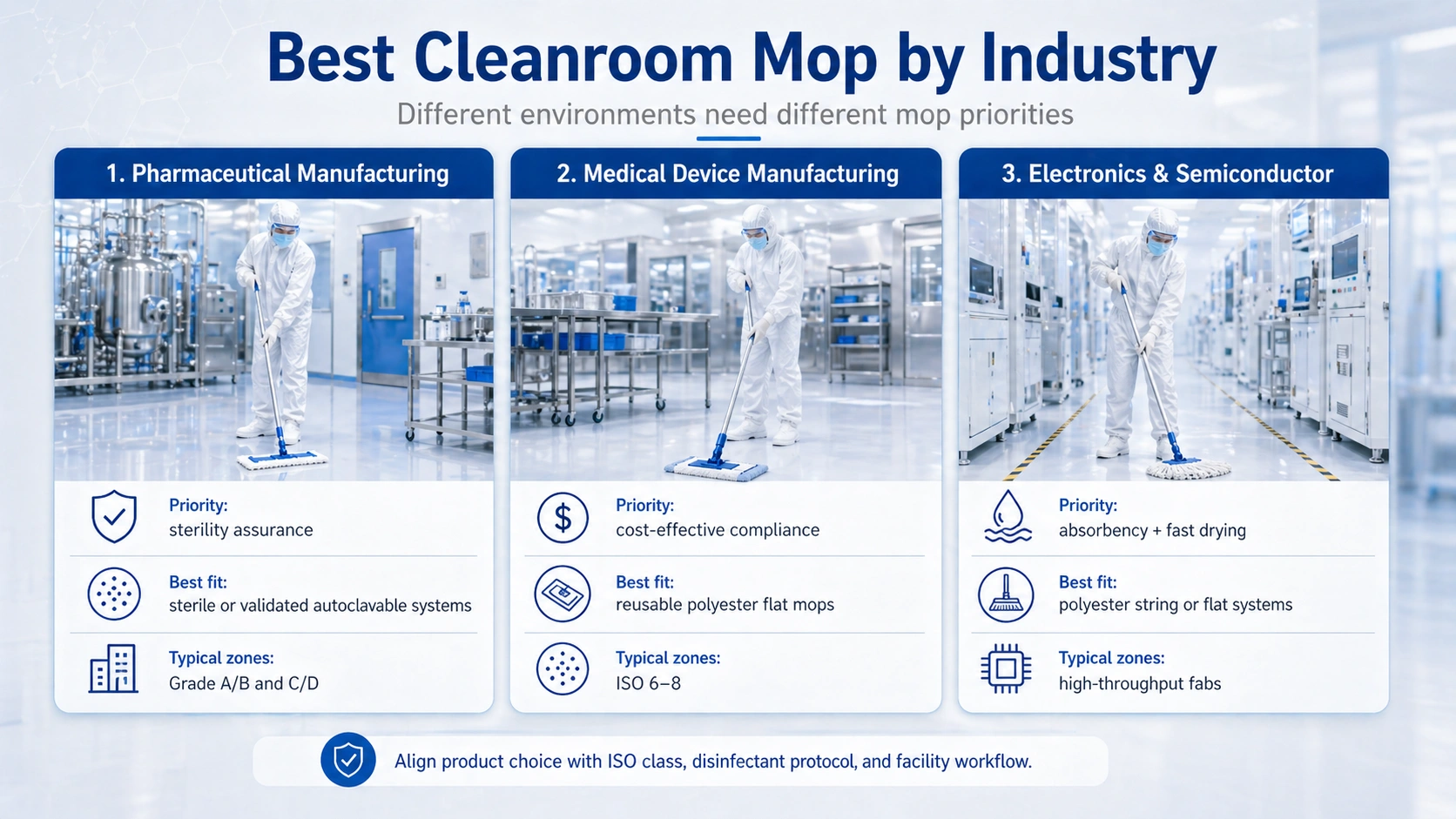
Pharmaceutical cleanrooms often need the strongest documentation package. Sterility assurance, batch traceability, CoA, change control and mop compatibility with cleaning SOPs can be more important than headline unit price. For detailed pharmaceutical applications, see pharmaceutical cleanroom mop solutions, GMP Annex 1 cleanroom mop guidance ۽ pharmaceutical cleanroom mop cleaning SOP guidance.
Medical device facilities often need a balance between compliance, repeatable cleaning and cost control. Reusable flat mop systems can work well when the facility has defined reprocessing, storage and change-out procedures. Component compatibility matters, especially for cleanroom mop frames ۽ صاف ڪمرو ايمپ هينڊلز.
Semiconductor and electronics cleanrooms often prioritize particle control, fast drying and efficient coverage. In high-throughput fabs, mop absorbency and operator speed can affect downtime between shifts. For this application, see semiconductor cleanroom mop solutions.
Biotech facilities often operate across multiple cleanroom grades and process types. Hybrid mop programs are common: sterile or high-documentation mops for critical areas and reusable systems for support zones. Learn more from MIDPOSI’s biotech cleanroom mop guide.
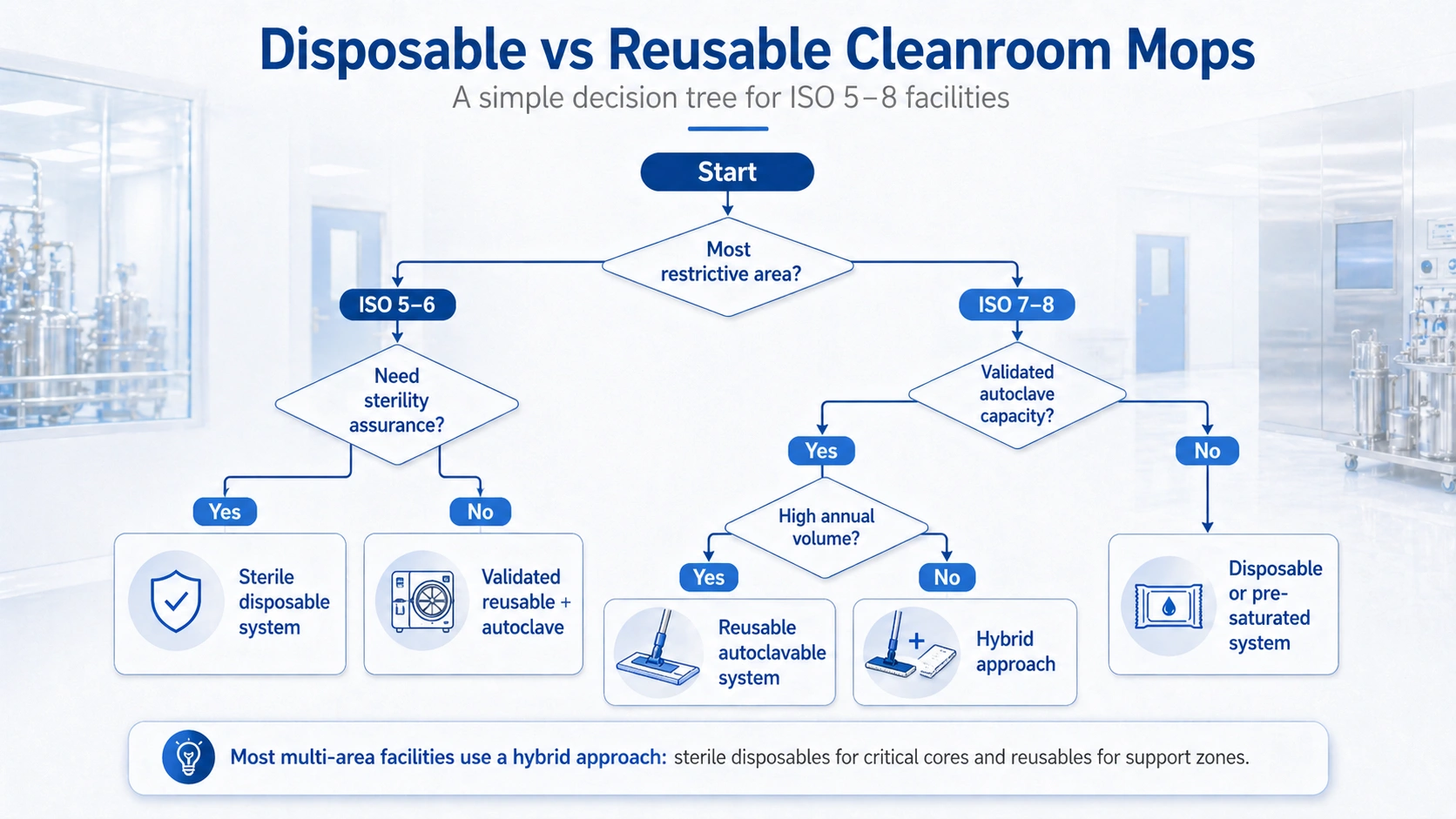
Disposable or sterile single-use mops are often selected when the facility wants to reduce reprocessing complexity, prevent cross-use risk or simplify batch segregation. They are particularly relevant for high-risk pharmaceutical or biotech areas where sterility assurance and lot documentation are important.
Reusable mop systems make sense when the facility already has validated laundry, autoclave or controlled reprocessing capacity. They can reduce long-term cost in high-volume areas, especially when reusable mop heads have a documented cycle life and are paired with compatible frames and handles.
Most multi-area facilities do not need one mop strategy for every zone. A practical approach is to use sterile or disposable mops in critical areas and reusable mop systems in support areas. For more detail on sterilization choices, review gamma-irradiated vs autoclavable cleanroom mops ۽ double-bagged sterile cleanroom mops.
Mops do not work in isolation. The complete cleanroom mop system includes the mop head, frame, handle, bucket, wringer, color coding and storage process. If these components are not compatible, operators may struggle to follow the SOP consistently.
Fixed or sealed mop handles can reduce crevices compared with poorly designed telescoping handles. Frames should match the mop head attachment method and be compatible with cleaning, disinfection or sterilization requirements. For system planning, see صاف ڪمرو فليٽ موپ سسٽم, cleanroom mop frames ۽ صاف ڪمرو ايمپ هينڊلز.
For pharmaceutical procurement teams, MIDPOSI also provides dedicated guidance on ڪيئن قابليت حاصل ڪجي هڪ دواسازي ڪلين روم موپ سپلائر, cleanroom mop validation documents and CoA ۽ batch traceability for pharmaceutical cleanroom mops.
Continue exploring MIDPOSI’s cleanroom mop resource cluster for system components, industry applications, material selection, validation documents and procurement guidance.
MIDPOSI supplies cleanroom mop systems for pharmaceutical, biotechnology, medical device, electronics and controlled manufacturing environments. Our product range includes microfiber mop heads, flat mop systems, mop frames, mop handles and cleanroom-compatible components designed for controlled cleaning workflows.
For regulated operations, MIDPOSI can support procurement teams with product recommendations, material comparison, mop system configuration and documentation for supplier review. If your facility needs a complete product set, review the MIDPOSI cleanroom garments and mops catalog or visit the product solutions page.
ISO 5 areas usually require a mop system with very low particle generation, strong documentation and either sterile supply or validated sterilization. The best choice depends on whether the facility prefers disposable sterile mops or autoclavable reusable systems.
Reusable mops can offer lower long-term cost when the facility has validated reprocessing capacity. Disposable or sterile mops can reduce reprocessing complexity and may be preferred for high-risk or batch-segregated areas.
Microfiber, knitted polyester, foam-core and string-style polyester each have different strengths. The right material depends on particle-control requirements, disinfectant compatibility, absorbency, sterilization method and cleaning surface.
No. Mop handles and frames should be selected as part of the complete mop system. Mop heads, frames, handles and bucket workflow must be compatible with the cleaning method, disinfectants, sterilization process and operator SOP.
Depending on the application, buyers may request CoA, material information, lot traceability, particle-control documentation, sterilization records, chemical compatibility information and supplier qualification documents.
Send MIDPOSI your cleanroom class, industry, disinfectant protocol, sterilization method and expected cleaning frequency. Our team can help you compare mop heads, frames, handles and complete cleaning system options for your facility.


اسان توهان سان 1 ڪم ڪندڙ ڏينهن اندر رابطو ڪنداسين، مهرباني ڪري لاتعداد سان اي ميل ڏانهن ڌيان ڏيو "*@midposi.com".