Temiz Oda Kirliliği Kontrol Protokollerini Anlamak
Cleanroom contamination control protocols define how a facility prevents, detects, responds to, and documents contamination risks. In GMP and ISO-controlled environments, a protocol must connect personnel behavior, cleaning SOPs, mop system selection, environmental monitoring, validation records, and QA review.
Quick Answer
A cleanroom contamination control protocol is a documented system that defines contamination risks, cleaning procedures, personnel controls, material transfer rules, mop system requirements, monitoring methods, deviation response, and records needed for QA review and audit readiness.
1. What Should a Cleanroom Contamination Control Protocol Define?
A cleanroom contamination control protocol is not just a cleaning instruction. It is a documented control framework that explains how contamination risks are identified, controlled, monitored, documented, and reviewed. For GMP and ISO-controlled cleanrooms, the protocol should be practical for operators and strong enough for QA, CQV, supplier qualification, and regulatory audit discussions.
Prevention
Defines how personnel, materials, cleaning tools, room zoning, and mop systems prevent contamination before it spreads.
Doğrulama
Defines how environmental monitoring, cleaning records, and QA review confirm that controls are working.
Response
Defines how deviations, excursions, failed cleaning checks, or monitoring alerts are investigated and corrected.
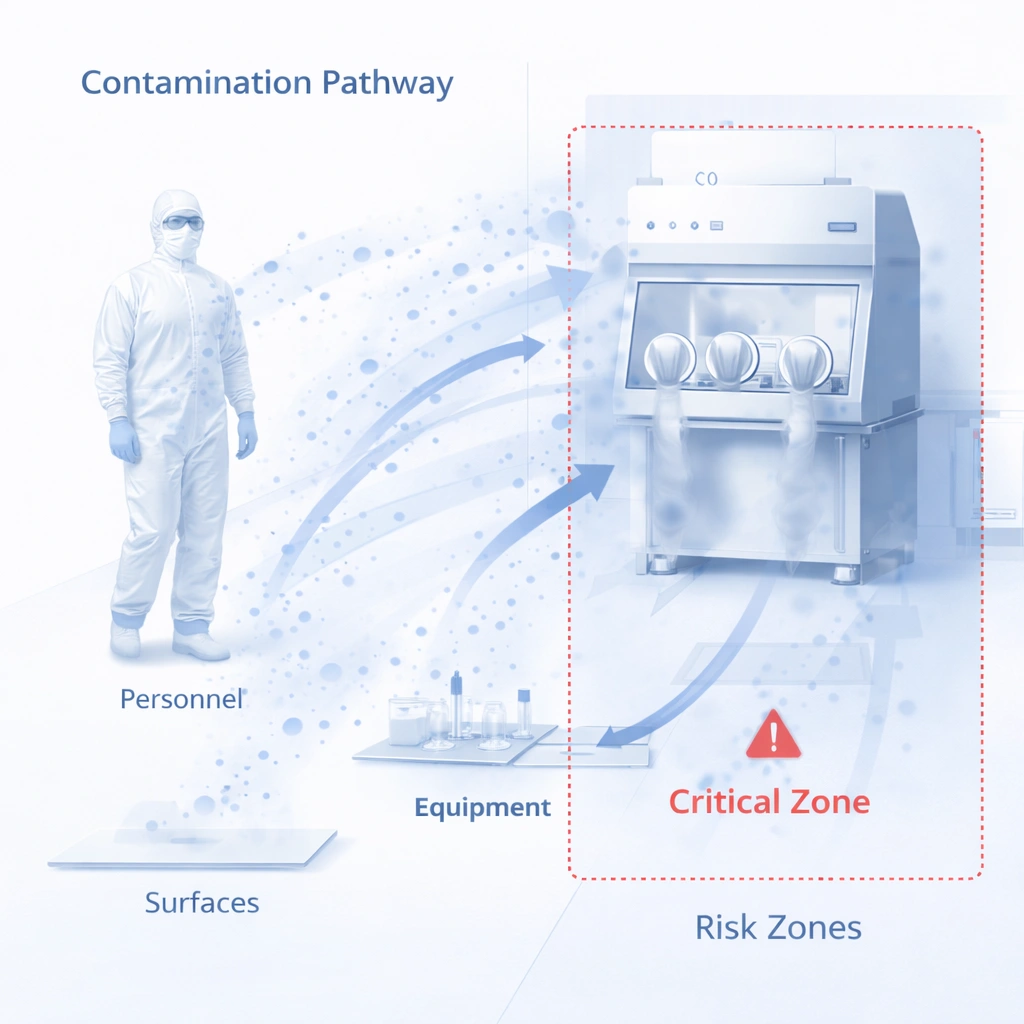
2. Match the Protocol to Cleanroom Classification and Process Risk
A contamination control protocol should be risk-based. A GMP Grade A/B area, aseptic support room, ISO 5 zone, ISO 7 production room, or ISO 8 support area should not use the same level of cleaning control, mop system requirement, sterile requirement, or documentation package.
| Protocol Area | Risk Question | Protocol Requirement | Suggested Internal Resource |
|---|---|---|---|
| Room classification | What ISO / GMP grade does the area operate under? | Define cleaning frequency, gowning level, and monitoring intensity by room grade. | ISO 14644 Sınıflandırma Kılavuzu |
| Process risk | Is the process aseptic, sterile-support, or general controlled production? | Define whether sterile mop systems, stronger transfer controls, or additional QA review are needed. | EU GMP Annex 1 SOP Documentation |
| Cleaning scope | Are floors, walls, ceilings, carts, or equipment exteriors included? | Define tool type, mop head, frame, handle, and surface-specific workflow. | Temiz Oda Paspaslama SOP'si |
| Denetim hazırlığı | Can the facility prove the protocol was followed? | Maintain SOPs, cleaning logs, training records, product specifications, and monitoring records. | How to Prepare for GMP Cleanroom Audit |
3. Include Cleaning SOPs and Cleanroom Mop System Controls
The cleaning section of a contamination control protocol should define how surfaces are cleaned and which tools are approved. In many cleanrooms, mop systems are used for floors, walls, ceilings, and large surfaces, so the protocol must define mop head material, sterile status, replacement frequency, chemical compatibility, and documentation requirements.

- Approved mop system: model, material, sterile / non-sterile status, and intended cleanroom grade.
- Surface scope: floor, wall, ceiling, equipment exterior, or transfer area.
- Mopping pattern: direction, overlap, zone sequence, and pass control.
- Mop head change rule: area size, soil level, room grade, or batch change requirement.
- Tool compatibility: frame, handle, bucket, wringer, and disinfectant compatibility.
- Records: operator, date, room, cleaning agent, mop lot, and QA review if required.
4. Connect the Protocol to Environmental Monitoring
A contamination control protocol is incomplete without verification. Environmental monitoring helps confirm whether personnel behavior, cleaning workflow, mop system selection, transfer controls, and room operations are maintaining the intended level of control.
The protocol should specify:
- Monitoring locations and sampling frequency
- Alert and action levels
- Surface, air, and personnel monitoring logic
- Review frequency for trend data
- How cleaning records are compared with monitoring results
- When deviations, investigations, or CAPA are triggered
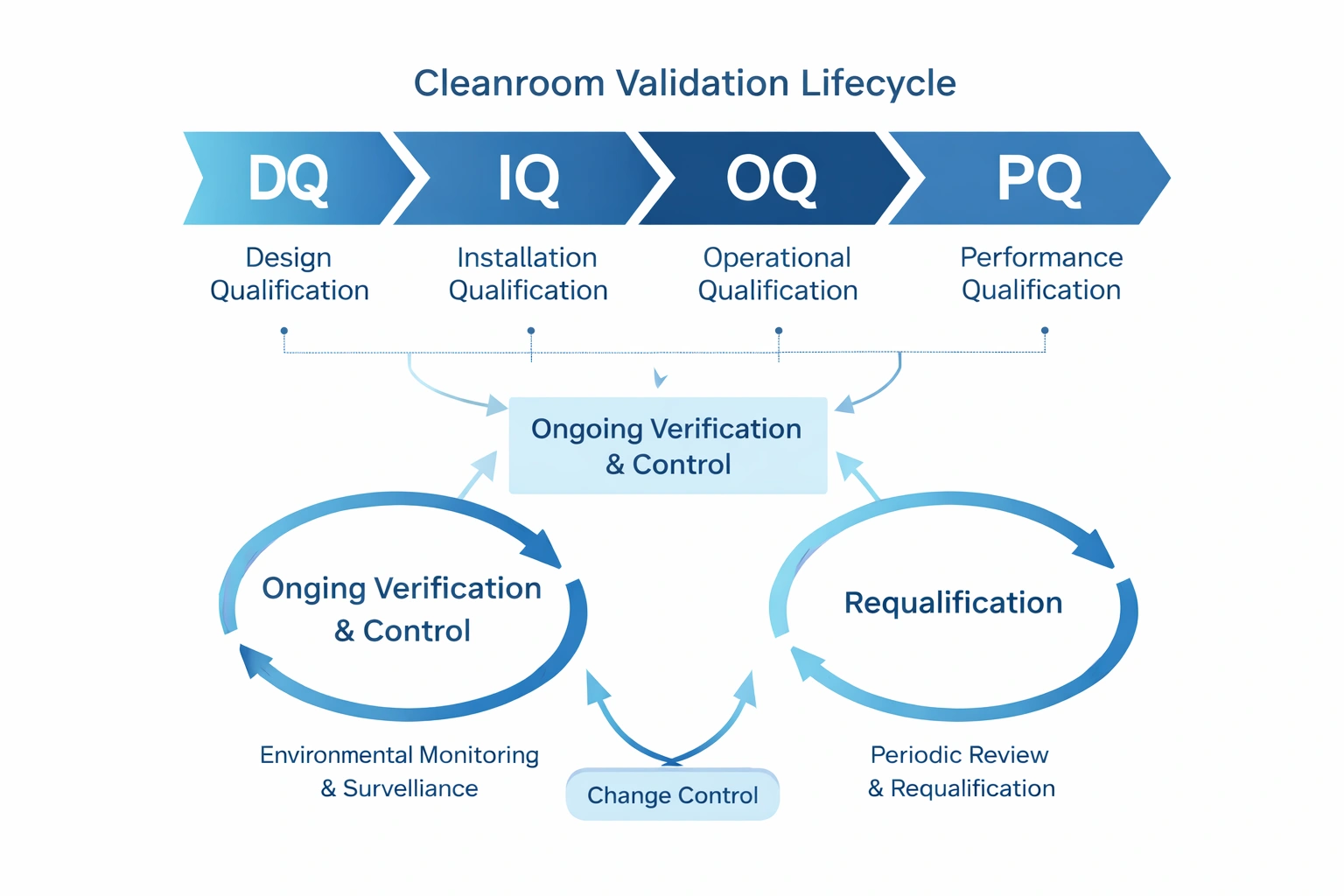
5. Align Protocols with QA, Validation, and Supplier Qualification
Cleanroom contamination control protocols should be aligned with QA and validation expectations. If a facility uses a mop system, garment system, sterile consumable, or critical cleaning tool, the supplier and product documentation should support qualification and audit review.
| QA / Validation Element | Protocol Question | Records to Maintain |
|---|---|---|
| Supplier qualification | Is the supplier suitable for GMP cleanroom consumables? | Supplier information, product specification, quality documents, audit support records. |
| Product qualification | Is the mop system suitable for the intended cleanroom grade and workflow? | Specification sheet, material data, sterile/non-sterile status, COA reference. |
| Cleaning validation support | Does the tool support the cleaning method defined in the SOP? | Cleaning records, SOP checklist, environmental monitoring trend, deviation history. |
| Denetim izi | Can the facility show the protocol was followed consistently? | Training records, cleaning logs, review signatures, CAPA records, batch traceability. |
Useful QA and validation pages include Cleanroom IQ/OQ/PQ Qualification Explained, Temiz Oda Doğrulama Yaşam Döngüsü Yönetimi, Ve Temiz Oda Kalifikasyon Doğrulaması SOP Şablonu.
6. Define Deviation Response, CAPA, and Protocol Improvement
A cleanroom contamination control protocol should also explain what happens when control is lost or trending becomes unfavorable. This includes environmental monitoring excursions, repeated cleaning failures, improper gowning, incorrect mop use, unapproved material transfer, or missing cleaning records.
Deviation response should include:
- Immediate containment or cleaning response
- Initial assessment of affected area, product, process, or batch
- Root cause review: personnel, material, mop system, workflow, disinfectant, or equipment
- Corrective action: retraining, SOP update, tool replacement, supplier review, or workflow change
- Preventive action: trend review, monitoring adjustment, or protocol revision
- QA closure with documented evidence
7. Cleanroom Contamination Control Protocol Checklist
The following checklist can help QA, validation, and cleanroom operations teams review whether a protocol is complete.
| Protocol Section | Required Content | Evidence / Link |
|---|---|---|
| Kapsam | Room grade, process area, surface type, and cleaning responsibility | Room classification and area list |
| Contamination risks | Personnel, material, surface, tool, airflow, and transfer risks | Risk assessment or contamination pathway map |
| Cleaning method | SOP, cleaning sequence, disinfectant, mop system, replacement rules | Cleaning SOP and mopping SOP |
| İzleme | Sampling locations, frequency, alert/action levels, trend review | Environmental monitoring plan |
| Doğrulama | Supplier qualification, product suitability, IQ/OQ/PQ or lifecycle review where applicable | QA / validation records |
| Sapma yanıtı | Investigation, CAPA, retraining, SOP update, and closure | Sapma ve DÖF kayıtları |
A strong contamination control protocol is not only a document. It is the operating logic that connects people, cleanroom tools, cleaning workflows, monitoring data, and QA evidence.
Conclusion: Protocols Turn Cleanroom Cleaning into a Controlled System
Understanding cleanroom contamination control protocols helps facilities move beyond routine cleaning and build a documented, risk-based, audit-ready control system. The protocol should define contamination sources, cleaning methods, personnel controls, mop system requirements, environmental monitoring, validation records, and deviation response.
For cleanroom mop systems, this means the product must fit the room grade, cleaning workflow, sterile requirement, documentation needs, and QA expectations. When the protocol and product selection are aligned, contamination control becomes easier to train, verify, and defend during audits.
FAQ: Cleanroom Contamination Control Protocols
What is a cleanroom contamination control protocol?
It is a documented system that defines how contamination risks are prevented, monitored, investigated, corrected, and recorded in a cleanroom environment.
What should be included in a contamination control protocol?
It should include scope, room classification, contamination risks, personnel controls, cleaning SOPs, mop system requirements, environmental monitoring, deviation response, CAPA, and documentation records.
How do cleanroom mop systems fit into the protocol?
Mop systems should be defined by material, sterile status, surface use, replacement frequency, frame and handle compatibility, disinfectant compatibility, and documentation support.
Why does QA need mop system documentation?
QA needs documentation to verify product suitability, supplier qualification, lot traceability, sterile status, cleaning records, and audit readiness.
How often should protocols be reviewed?
Protocols should be reviewed when there are monitoring excursions, audit findings, process changes, new cleaning tools, new room classifications, repeated deviations, or scheduled quality system reviews.
Need Mop System Documentation for Cleanroom Protocols?
MIDPOSI can help you review low-lint cleanroom mop systems, sterile and non-sterile configurations, documentation support, and sample options for GMP and ISO-controlled cleanrooms.



